Status Assessment 2022 - Lophelia pertusa reefs
| Assessment of status | Distribution | Extent (for habitats) | Condition (for habitats) | Previous OSPAR status assessment | Status (overall assessment) | |
|---|---|---|---|---|---|---|
Region | I | ←→1,2,5 | ←→1,5 | ←→1,5 | ● | Poor |
| II | ←→1,2,5 | ←→1,5 | ←→1,5 | ● | Poor | |
| III | ←→1,2,5 | ←→1,5 | ←→1,5 | ● | Poor | |
| IV | ←→1,2,5 | ←→1,5 | ←→1,5 | ● | Poor | |
| V | ←→1,2,5 | ←→1,5 | ←→1,5 | ● | Poor | |
| Assessment of threats | Habitat loss/degradation through physical damage from demersal fisheries | Habitat loss or alteration through community shifts and/or ocean acidification due to climate change | Threat or impact | |
|---|---|---|---|---|
Region | I | ←→ 1,2,5 | ? 3,5 | |
| II | ←→ 1,2,5 | ? 3,5 | ||
| III | ←→ 1,2,5 | ? 3,5 | ||
| IV | ←→ 1,2,5 | ? 3,5 | ||
| V | ←→ 1,2,5 | ? 3,5 | ||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: If in QSR 2010 then enter Regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ) based on Chapter 10 Table 10.1 and Table 10.2 . If a more recent status assessment is available, then enter ‘poor’/’good’
Status*(overall assessment)
red – poor
green – good
? – status unknown
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressure
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threats or impacts (overall assessment)
red – significant threat or impact;
green–no evidence of a significant threat or impact
Blue cells – insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
Confidence
Overall: Low to Medium confidence is placed overall on the assessment.
Distribution: High confidence that the distribution has not changed.
Condition: Low to medium confidence that the overall condition across the Regions is still declining. There is good evidence for declining condition, but it is limited to particular sites that have been studied, and broader evidence of decline is drawn from proxies such as overlap of fishing pressure with the habitat. Of cause for concern is the presence of fishing activity within Marine Protected Areas and Special Areas of Conservation.
Fishing Pressure: Medium confidence in the scale of fishing pressure currently acting on the habitat based on analysis of Global Fishing Watch data notwithstanding the various assumptions made during the analysis. However, it should be noted that while sufficient to highlight risk, the link between pressure and actual impact has not been well established.
Background Information
Year added to OSPAR List: 2003 https://www.ospar.org/documents?v=7182
- The original evaluation of Lophelia pertusa reefs against the Texel-Faial criteria referred to global importance, decline and sensitivity criteria, with information also provided on threat.
- The key criteria for their inclusion were:
- global and regional importance because of the high proportion of the known occurrences of these reefs in the North-East Atlantic;
- decline principally due to the impact of deep-water fishing;
- sensitivity based on fragility and slow growth rates, and the likely long duration required for reef regeneration if any, following damage;
- evidence of reefs impacted by deep-sea trawling.
Last status assessment: 2009. OSPAR (2009) updated the evaluation of L. pertusa reefs against the Texel-Faial criteria to globally important, ‘very sensitive’ to the effects of demersal trawling and ‘sensitive’ to the localised effects of oil and gas exploitation, ecologically ‘very important’, experiencing ‘significant’ decline and ‘currently’ under threat. https://www.ospar.org/documents?v=7182
Geographical Range and Distribution
Lophelia reefs are found in all OSPAR Regions. They typically occur below 200 m down to 1 200 m water depth but shallow occurrences in as little as 40 m have been recorded in Norwegian fjords. While the entire distribution and extent of L. pertusa reef habitat in the OSPAR area is not yet known, offshore exploration programmes, both national and as part of large EU projects such as CoralFISH and ATLAS, have since 2010 continued to increase the knowledge of the distribution of coral reefs in the OSPAR Maritime Area. For example, Arctic Waters (Region I) – Iceland, Faroes, Norway (Buhl-Mortensen et al., 2015); Bay of Biscay and Iberian Coast (Region IV) – Bay of Biscay (van den Beld et al., 2017), La Gaviera Canyon, Cantabrian Sea (Sanchez et al., 2014); and Wider Atlantic (Region V) – Galicia Bank (Somoza et al., 2014), Rockall Bank and Porcupine margin (SeaRover 2017/2019, Picton et al., 2021) (Figure 1). Within the EU CoralFISH project, Davies et al. (2017) identified 81 coral biotopes in OSPAR and Mediterranean waters including a number of Lophelia biotopes (living and dead reefs, dense framework, loosely-packed framework, rubble and isolated colonies).
The recorded range in the OSPAR Maritime Area has not expanded northward since the last assessment, however due to habitat mapping in the Barents Sea undertaken as part of the Norwegian Mareano programme many new records have been added. Elsewhere, the number of known occurrences of the habitat has increased due to on-going targeted research assessing VMEs.
It is likely however that new Lophelia reefs will continue to be discovered as unexplored seafloor is investigated.
Method of assessment: 3a
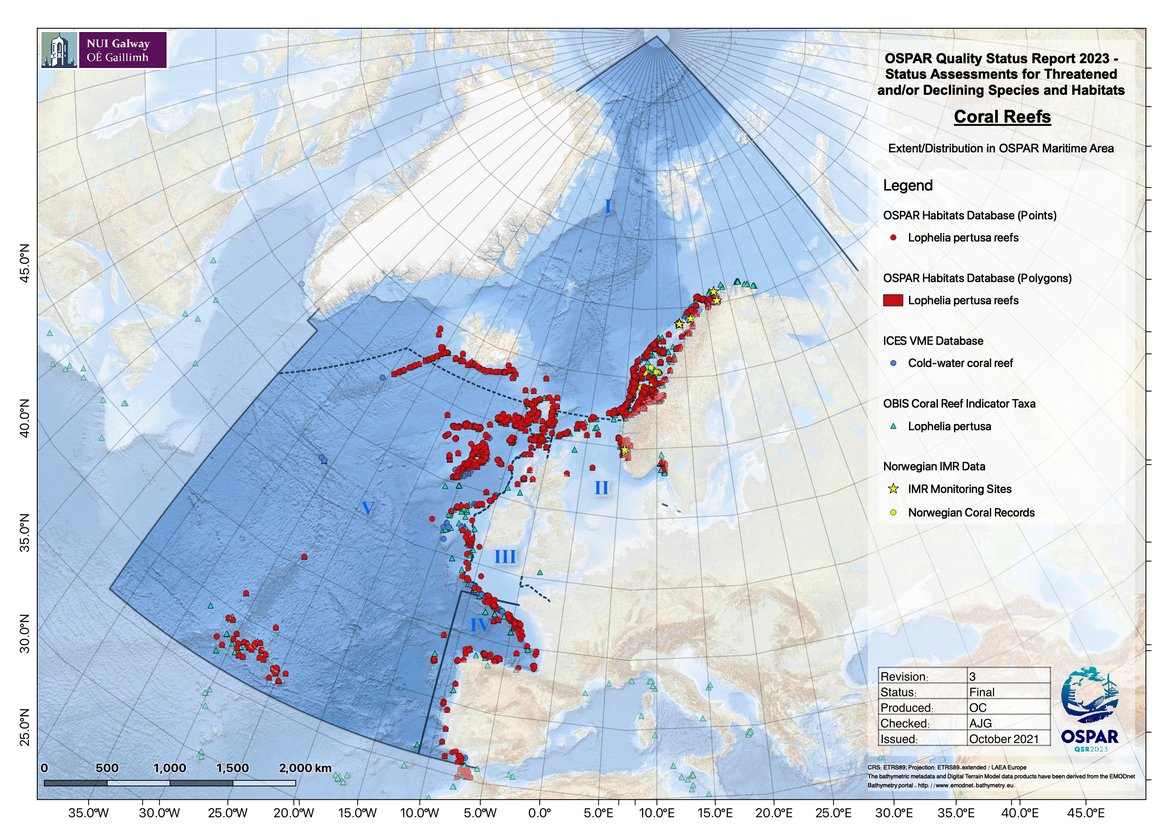
Figure 1: Distribution of known Lophelia pertusa reef in the OSPAR area.
Extent
Notwithstanding the introduction of successful management measures to reduce the impact of fishing in Norway, the extent of the habitat in OSPAR Region I is likely to still be decreasing because of fishing pressure (Figure 2). Buhl-Mortensen et al. (2019) carried out a risk assessment of the potential impact of fishing in Arctic and Sub-Arctic Waters and demonstrated that regionally, several VMEs are predicted to have experienced intermediate to high levels of fishing effort in > 50% of their optimal habitat including Lophelia reefs off Iceland. However, there is no direct evidence of impact and therefore it is not possible to state with confidence that the extent has decreased substantially.
Future prospects for habitat extent are that unprotected reefs, especially small patch reefs, are expected to continue to decline in extent due to the ongoing impacts of demersal fishing although the rate of decline may be slowing due to improved fisheries management (e.g. the 2016 EC Deep-Sea Access Regime; NEAFC VME protection measures and national initiatives such as in Norway) and over the next ten years should move from declining towards stability as pressure on reef habitats is relieved. There are indications that removing fishing pressure (e.g. in protected areas) can lead to recovery of ecosystem function in the short term and structural reef in the longer term (decades) (Buhl-Mortensen 2017; Beazley et al., 2021).
Method of assessment: 2a
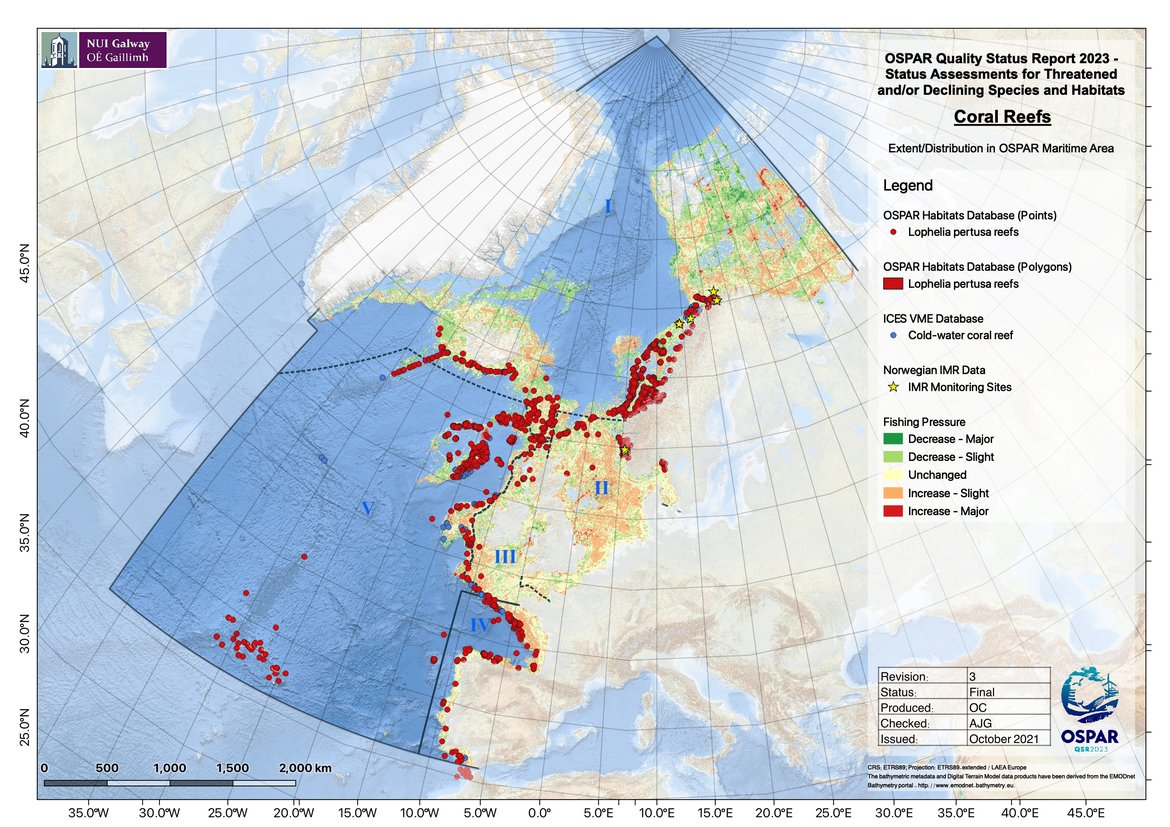
Figure 2: Distribution of coral reefs in the OSPAR region with fishing pressure.
Condition
Within OSPAR Region I and Greater North Sea (Region II), selected Lophelia pertusa reefs have been revisited regularly since 2012 as a part of an IMR (Norway) monitoring program of coral reefs. This monitoring uses a combination of functional indicators (e.g. respiration, nutrient cycling and energy stores of collected coral fragments) alongside structural indicators observed along randomly placed ROV video transects crossing the reefs (e.g. species composition, cover of live versus dead framework, siltation and tissue necrosis of corals and sponges) to assess the overall health status of the reefs. Over the nine year period, from the north (i.e. Stjernsund, Korallen, Steinavær and Hola) to the south (i.e. Nakken and Straumsneset) along the western Norwegian coast, no changes in the health parameters assessed were detected. Instead, in all monitored coral reefs (that are all located within fisheries closure areas) reefs appear to be in a stable state and healthy.
Conservation Status (2013 to 2018) of Marine Atlantic Reef habitats (Habitats Directive, Article 17) by EU Member State were last recorded as “Unfavourable-Inadequate (Ireland) and “Unfavourable-Bad” (UK) (see https://nature-art17.eionet.europa.eu/article17/). A deep-water habitat assessment performed in the ATLAS project (Kazanidis et al., 2020) indicated that the Rockall in Region V and Mingualy in Celtic Seas (Region III) are not in good status. Some evidence of damage to reefs was also observed during the SeaRover surveys 2017 to 2019 (Picton et al., 2021).
No change has been noted in Region IV but both Sanchez et al. (2016) and van den Beld et al. (2017), draw attention to possible evidence of historical fishing damage. France is embarking on a five year (2021-2026) monitoring programme: Characterisation and Ecology of Cold-water Reefs (ChEReef) which will include the installation of a coral reef observatory and in situ growth and recruitment rate assessments (Menot et al., 2021).
Threats and Impacts
The 2009 assessment noted that fishing was the only threat that has led to a documented decline in L. pertusa reefs while threats from deep-sea mining, and locally from oil and gas, activities existed. The report highlighted concerns that global increases in anthropogenic CO2 and shifting ocean currents could alter the ecology of the OSPAR Maritime Area and threaten L. pertusa reefs. A more recent overview of threats to Lophelia reefs can be found in Ragnarsson et al. (2017).
Analysis of Global Fishing Watch data from 2012 to present indicates that fishing pressure has remained relatively unchanged over that period (Figure 3).
While potential threats from oil and gas activities have decreased (with the possible exception of decommissioning leading to the removal of coral-supporting oil and gas infrastructure in the North Sea), the threat from climate change is increasing. It has been shown that meridional overturning circulation has the potential to increase acidification in the deep Atlantic Ocean with rapid shoaling of the aragonite saturation horizon particularly in the subpolar North Atlantic possible within the next three decades (Perez et al., 2018). Acidification can impact the integrity of coral reefs and cause ecosystem-scale habitat loss (Hennige et al. 2020). Some recent studies, however, suggest that Lophelia may contain sufficient genetic variability to adapt to future ocean acidification (Kurman et al., 2017) at least in some regions (Georgian et al., 2016).
Morato et al. (2020) used habitat suitability modelling to compare present-day (1951 to 2000) and projected Lophelia distributions under future (2081 to 2100; RCP8.5 or business-as-usual scenario) environmental conditions. The models predict a decrease of c. 80% in suitable habitat for Lophelia pertusa by 2100. The authors stress the importance of identifying and preserving species climate refugia when planning conservation measures to improve resilience against climate change.
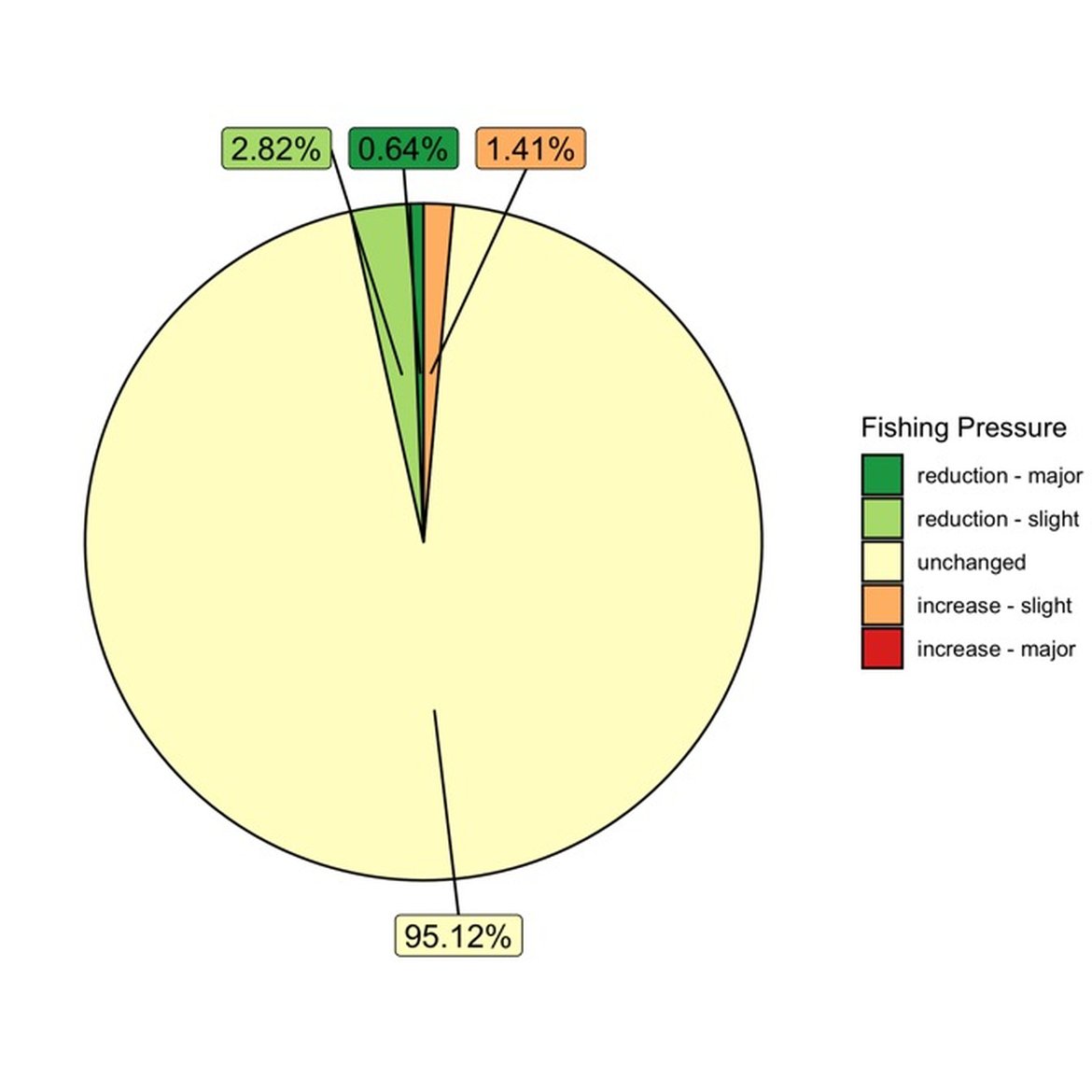
Figure 3: Overlap of Fishing Pressure and Coral Reefs based on Global Fishing Watch Data Aggregated to a Resolution of 25km² (2012 to present)
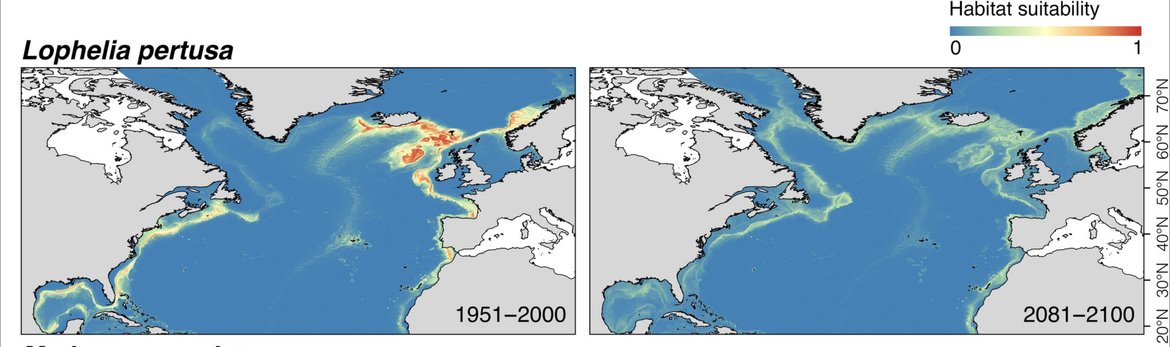
Figure 4: The predicted distribution of Lophelia pertusa under present day and under future environmental conditions (Morato et al., 2020).
Fishing pressure is likely to stabilize over the next 6 to 12 years as fisheries regulations implement a move to ecosystem based management. Ensuring that all anthropogenic pressures are removed from marine protected areas established for the conservation of Lophelia reefs is crucial to promote resilience and allow the species the space to adapt to climate change over the coming decades.
Marine protected areas and fisheries closures using a variety of legislative tools afford partial protection for Lophelia reefs (Figure 5) in the OSPAR Maritime Area. In total over 70% of Lophelia occurrences lie outside protected areas. There is however considerable regional variation with 50% of Lophelia occurrences protected in Region V and only 13% protected in Region I.
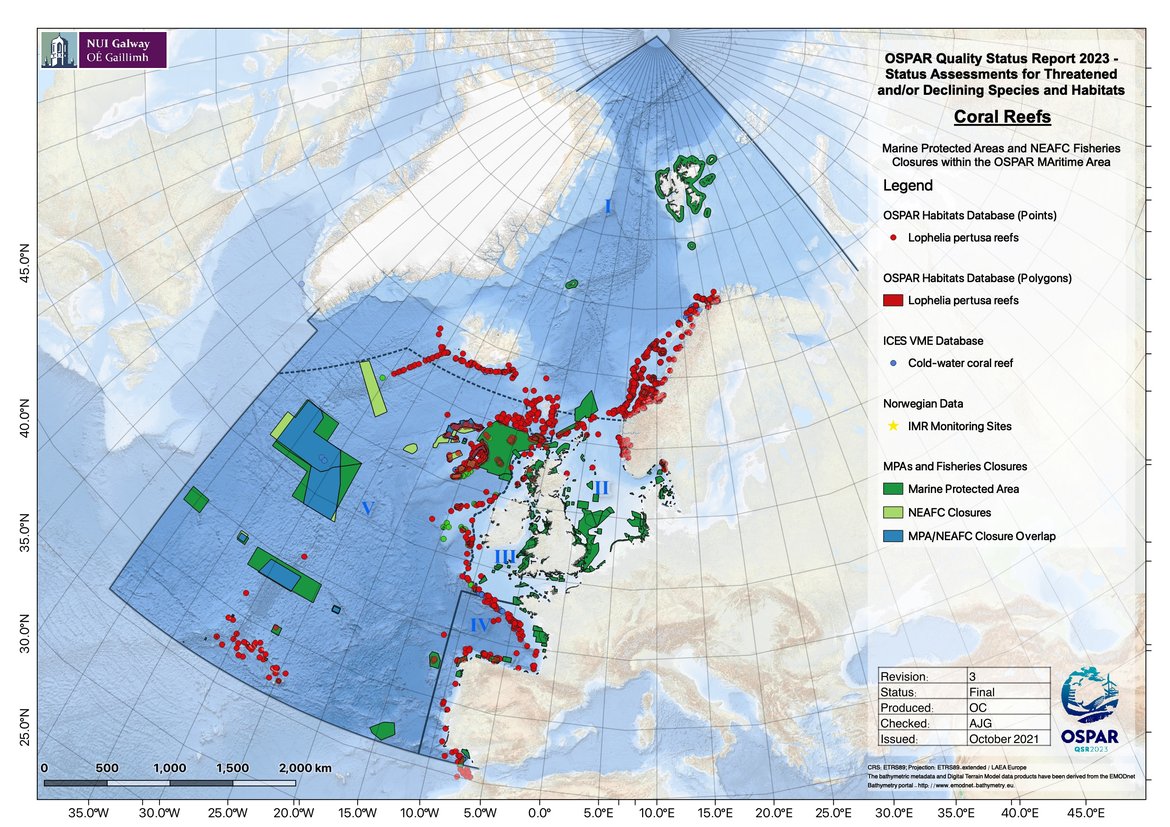
Figure 5: OSPAR marine protected areas and NEAFC fisheries closures in the OSPAR region
Of concern is evidence that bottom contact gear has been used in protected areas (Figure 7).
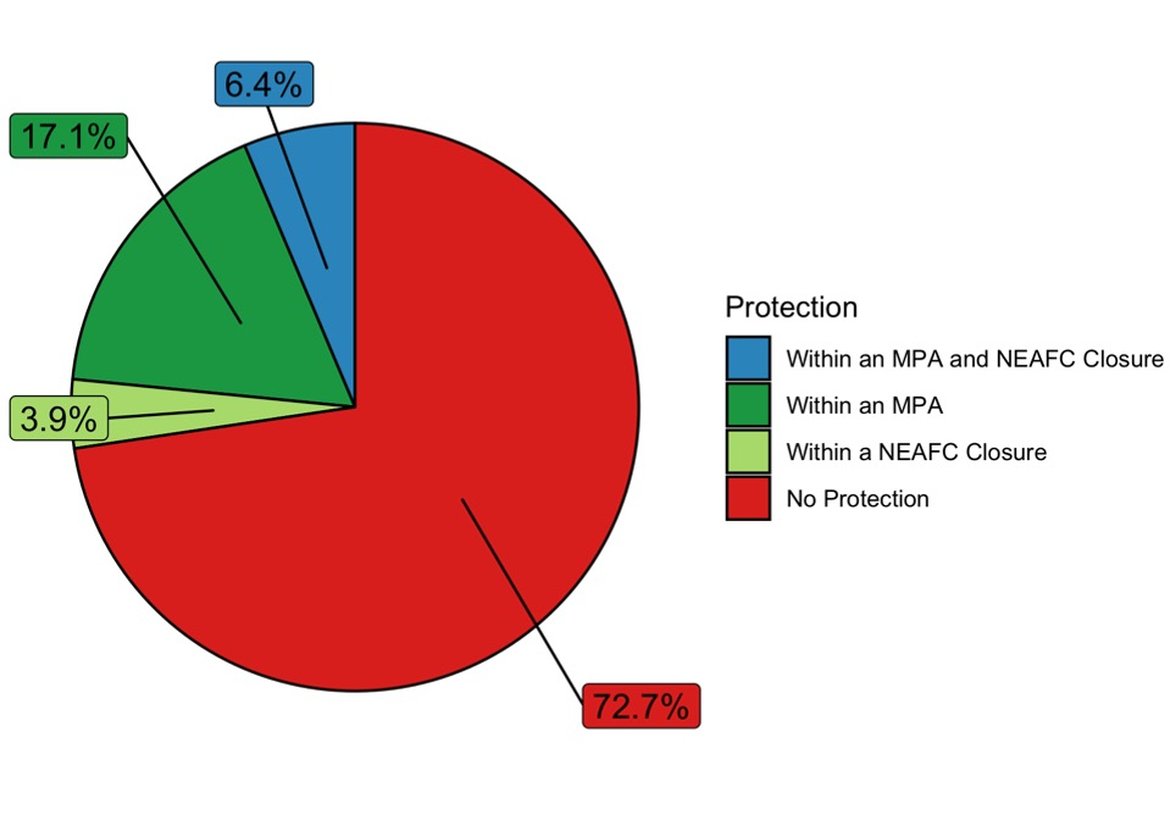
Figure 6: Distribution of Lophelia reef records with respect to MPAs and Fisheries Closures within the OSPAR Area.
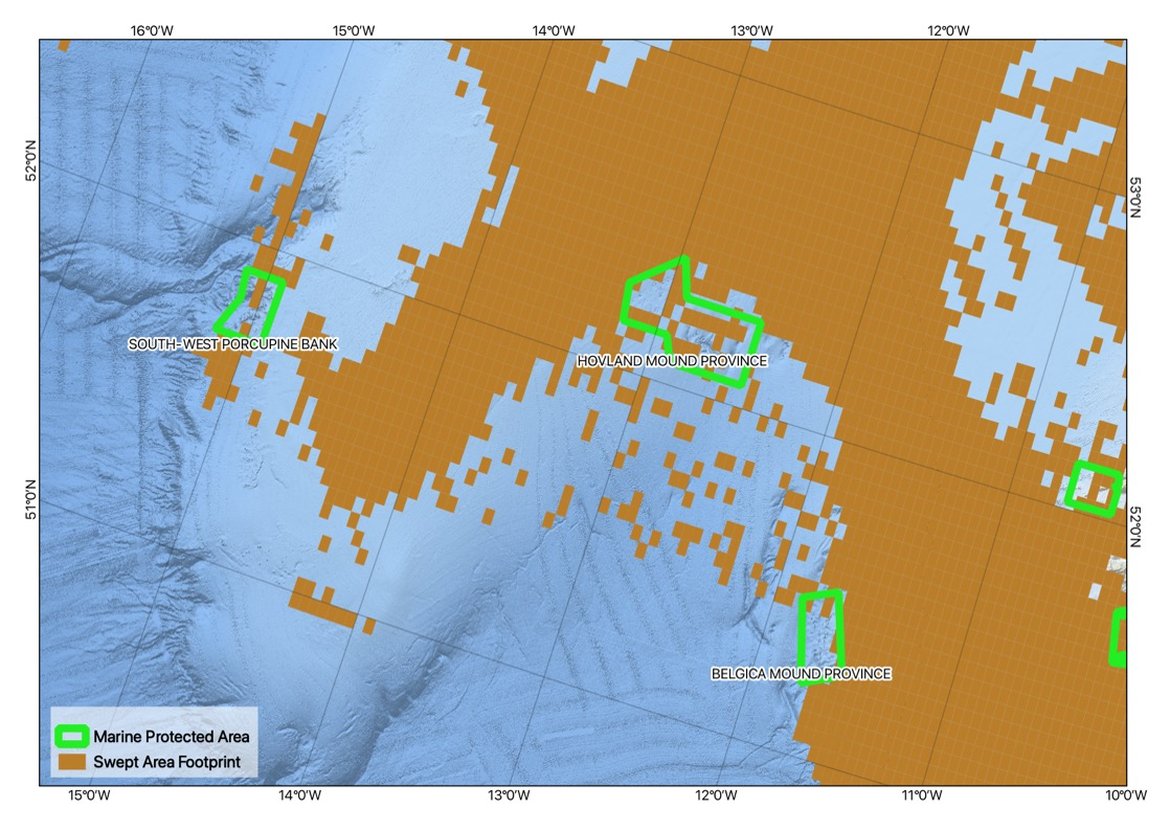
Figure 7: Example of bottom contacting fishing activity within the Hovland Mound Province SAC, Ireland in 2016. Map based on spatial data layers of swept area ratio produced by ICES in response to a 2017 request by OSPAR.
Measures that address key pressures from human activities or conserve the species/habitat
OSPAR measures:
3.1 a Iceland and Norway have closed all areas where Lophelia reefs are known to exist to bottom fishing; The United Kingdom, France, Ireland and Sweden have introduced legislation that allows protection of Lophelia reefs. Sweden has also implemented a total ban of fishing in areas with both dead and live coral reefs from October 2021.
3.1 b Iceland, Ireland, Norway, Spain, Sweden and the United Kingdom consider management measures to protect Lophelia reefs are effective. Iceland and Ireland may consider further measures to address key threats or pressures. Norway has reviewed existing closures implemented in 1999 and added additional protected areas in 2016.
3.1 c Iceland, Ireland, Norway, Spain, Sweden and the United Kingdom have implemented programmes mapping the distribution of Lophelia reefs.
3.1 d In Iceland, fisheries observers record coral by-catch, and companies attempt to record coral by-catch information from their vessels; Ireland is broadening its knowledge through research; Sweden is involving fisherman in a LIFE project; the United Kingdom is integrating data from fisheries surveys. In the EU, fisheries vessels are obliged to report VME bycatch between 400 and 800 m depth.
3.1 e Iceland, Ireland, Norway, Sweden and the United Kingdom make all VMS data available to regulators.
3.1 f France, Iceland, Ireland, Norway, Spain, Sweden and the United Kingdom all regularly report new data on the distribution, quality and extent of Lophelia reef habitat to the OSPAR habitat mapping database.
3.1 g France, Ireland, Norway, Spain, Sweden and the United Kingdom have all designated MPAs for Lophelia reefs. Designating of sites in Iceland is in progress.
3.1 h Only Sweden has a specific (adaptive) management plan for MPAs designated for Lophelia reefs. Management of activities likely to impact the reefs is routinely carried out amongst the other contracting parties.
3.1 i Iceland, Ireland, Spain, Sweden and the United Kingdom report various steps to address significant adverse impacts on Lophelia reefs arising from human activities in their waters, for example through fisheries legislation, including the Common Fisheries Policy, through MSFD, and through the Habitats Directive and internationally through support and advice for NEAFC (via ICES WGDEC). LIFE programme, LIFE Lophelia, is ongoing 2019 to 2025 with method development for restoration of Lophelia reefs (www.lifelophelia.se).
Actions taken beyond the scope of the OSPAR measures:
Within the North-East Atlantic Fisheries Commission (NEAFC) Regulatory Area, measures to protect VMEs include the implementation of bottom fishing closure areas and the use of “move-on” rules for fishing vessels, related to encounters of specific quantities of VME indicators within bycatch from bottom-contacting gears (NEAFC Recommendation 19 2014).
Within EU waters, Regulation (EU) 2016/2336 restricts bottom fishing below 400 m to the 2009-2011 fishing footprint, prohibits bottom fishing below 400 m where VMEs are known or likely to occur (through designation of VME closures) and places a complete ban on bottom trawling deeper than 800 m. Within UK waters, regulation (EU) 2016/2336 is transposed into the Common Fisheries Policy and Aquaculture (Amendment etc.) (EU Exit) Regulations 2019, with the same fishing restrictions and prohibitions in place. In Norwegian waters, Regulation 2011/755 restricts fishing with bottom gear on vulnerable benthic habitats in areas deeper than 1000 m in the Norwegian Economic Zone, including the Fisheries Protection Zone near Svalbard, and the Fisheries Zone around Jan Mayen.
Regulation (EU) 2019/1241 Annex II provides closed areas for the protection of sensitive habitats. It prohibits deployment of bottom trawls, bottom set gillnets, entangling nets and trammel nets within specified areas around El Cachucho, Madeira and the Canary Islands, and the Azores. It further prohibits deployment of bottom set longlines within El Cachucho. It specifies five areas closed to bottom trawls, bottom set gillnets, entangling nets, trammel nets and bottom set longlines in the Porcupine Seabight and Porcupine Bank, and prohibits bottom trawling around the Darwin Mounds.
Conclusion (including management considerations)
While the full distribution of Lophelia pertusa reef habitat in the OSPAR Maritime Area is not yet known, offshore exploration programmes, both national and EU funded, have contributed to improved knowledge of their locations. Evidence of local damage from trawling, fishing activity in MPAs and assessment of potential impact due to the overlap of fishing pressure with reef habitat suggest that reef extent is still under threat particularly reefs that are currently unprotected. Over the longer term, Lophelia reefs are projected to come under increasing stress from environmental changes linked to climate impacts.
Evaluation of L. pertusa reefs against the Texel-Faial criteria suggests that consideration should be given to including ‘sensitive’ to expected climate change induced environmental changes along with the criteria listed in the 2010 OSPAR assessment. Also it is recommended that the title of the habitat on the OSPAR List be amended to ‘Stony Coral’ reef to reflect the inclusion of other corals, such as Madrepora oculata (cf. Arnaud-Haond et al., 2017) and Solenosmilia variabilis, which are within the scope of this habitat type and would take account of the recent reclassification of Lophelia pertusa as Desmophyllum pertusum (Addamo et al., 2016).
The confidence for this assessment is low to medium given the caveats associated with calculating fishing pressure using Global Fishing Watch data. Modelling of the predicted response of reefs to changing environmental conditions associated with one IPCC scenario provides only one possible trajectory for the likely response of reefs. It does highlight, however, the importance of establishing an OSPAR wide monitoring network to provide an early indication of biological changes taking place at reef locations.
During the next reporting cycle, re-assessment of the impact of fishing should take place in six years to determine whether evolving fisheries regulations and expansion of MPA networks to meet UN CBD 2030 targets have reduced the fishing threat with due consideration of Annex 5 Article 4. A longer term ten year assessment should be planned to address potential changes in reef function and structure linked to climate change.
Knowledge Gaps
Accurate distribution maps of Lophelia reefs in the OSPAR Maritime Area are required to better assess threats from human activities especially bottom trawling. The OSPAR threatened and/or declining habitats database should therefore be the most up to date and comprehensive source of occurrence records for the region and it is imperative that Contracting Parties continue to update the database regularly and if possible in response to an annual data call.
More stringent monitoring of vessel compliance with fisheries regulations particularly in MPAs is required.
Consideration should be given to an assessment of OSPAR MPA network coherence using recently developed seascape genetics approaches combined with particle tracking (as a proxy for larval dispersion) (e.g. Ross et al., 2017) in high resolution ocean models used in conjunction with multi-objective network optimization techniques (Fox et al., 2019) or marine spatial planning decision support tools (e.g. Combes et al., 2020).
Method used
Main source of information:
- OSPAR data assessment only
- Assessment derived from third party assessment
- Assessment derived from a mix of OSPAR data assessment and assessments from third parties
Assessment is based upon:
a) complete survey or a statistically robust estimate (e.g. a dedicated mapping or survey or a robust predictive model with representative sample of occurrence data, calibration and satisfactory evaluation of its predictive performance using good data on environmental conditions across entire species range);
b) based mainly on extrapolation from a limited amount of data (e.g. other predictive models or extrapolation using less complete sample of occurrence and environmental data);
c) based mainly on expert opinion with very limited data;
d) insufficient or no data available.
Addamo et al. 2012. Desmophyllum dianthus (Esper, 1794) in the Scleractinian Phylogeny and Its Intraspecific Diversity. PLoS ONE 7(11): e50215. doi: https://doi.org/10.1371/journal.pone.0050215
Addamo et al. 2016. Merging scleractinian genera: the overwhelming genetic similarity between solitary Desmophyllum and colonial Lophelia. BMC Evol. Biol. 16, 108.
Arnaud-Haond, S., Van den Beld, I.M.J., Becheler, R., Orejas, C., Menot, L., Frank, N., Grehan, A. & Bourillet, J.F. 2017. Two “pillars” of cold-water coral reefs along Atlantic European margins: Prevalent association of Madrepora oculata with Lophelia pertusa , from reef to colony scale. Deep Sea Research Part II: Topical Studies in Oceanography, 145, 110-119.
Beazley L., Kenchington, E, Korabik M, Fenton D, King M. 2021. Other effective area-based conservation measures promote recovery in a cold-water coral reef. Global Ecology and Conservation 26 e01485, https://doi.org/10.1016/j.gecco.2021.e01485
Buhl-Mortensen, L, Olafsdottir SH, Buhl-Mortensen, P, Burgos, JM, Ragnarsson, SA. 2015. Distribution of nine cold-water coral species (Scleractinia and Gorgonacea) in the cold temperate North Atlantic in light of bathymetry and hydrography. Hydrobiologia, 759:39-61. DOI: https://doi.org/10.1007/s10750-014-2116-x
Buhl-Mortensen, P. 2017. Coral reefs in the Southern Barents Sea: habitat description and the effects of bottom fishing, Marine Biology Research, DOI: https://doi.org/10.1080/17451000.2017.1331040
Buhl-Mortensen, L., Burgos, J., Steingrund, P., Buhl-Mortensen, P., Ólafsdóttir, S., and Ragnarsson, S. 2019. Vulnerable Marine Ecosystems (VME) Coral and Sponge VMEs in Arctic and Sub-Arctic Waters-Distribution and Threats. TemaNord 2019:519. Nordic Council of Ministers, 144.
Combes et al. 2021. Systematic conservation planning at an ocean basin scale: identifying a viable network of deep-sea protected areas in the North Atlantic and the Mediterranean. Front. Mar. Sci., https://doi.org/10.3389/fmars.2021.611358
Davies, J., Guillaumont, B., Tempera, F., Vertino, A., Beuck, L., Ólafsdóttir, S. H., Smith, C. J., Fosså, J. H., van den Beld, I. M. J., Savini, A., Rengstorf, A., Bayle, C., Bourillet, J. F., Arnaud-Haond, S., and Grehan, A. (2017). "A new classification scheme of European cold-water coral habitats: implications for ecosystem-based management of the deep sea. Deep Sea Research Part II: Topical Studies in Oceanography, 145, 1-7.
Fox et al. 2019. An Efficient Multi-Objective Optimization Method for Use in the Design of Marine Protected Area Networks. Front. Mar. Sci., 05 https://doi.org/10.3389/fmars.2019.00017
Georgian et al. 2016. Biogeographic variability in the physiological response of the cold‐water coral Lophelia pertusa to ocean acidification. Marine Ecology 37: 1345-1359.
Hennige et al. 2020. Crumbling Reefs and Cold-Water Coral Habitat Loss in a Future Ocean: Evidence of “Coralporosis” as an Indicator of Habitat Integrity. Front. Mar. Sci., 17 https://doi.org/10.3389/fmars.2020.00668
Kazanidis, G., et al. 2020 Assessing the environmental status of selected North Atlantic deep-sea ecosystems. Ecological Indicators 119: 106624
Kurman et al. 2017. Intra-specific variation reveals potential for adaptation to ocean acidification in a cold-water coral from the Gulf of Mexico. Front. Mar. Sci., 04 https://doi.org/10.3389/fmars.2017.00111
Menot et al. 2021. The ChEReef project: What are the threats on cold-water coral reefs in the Bay of Biscay, how to monitor and protect them. Poster presentation at the 16th Deep Sea Biology Symposium, Brest, September 2021.
Morato, T. et al. 2020. Climate-induced changes in the suitable habitat of cold-water corals and commercially important deep-sea fishes in the North Atlantic. Global Change Biology 26: 2181-2202
Perez et al. 2018. Meridional overturning circulation conveys fast acidification to the deep Atlantic Ocean. Nature 554: 515–518.
Picton, B.E., Morrow, C.C., Scally, L., Pfeiffer, N., McGrath, F. 2021. Sensitive Ecosystem Assessment and ROV Exploration of Reef (SeaRover), Synthesis Report. Report prepared by MERC Consultants Ltd. on behalf of the Marine Institute, Galway 161 pp
Ragnarsson, S.A., J.M. Burgos, T. Kutti, I.van den Beld, H. Egilsdóttir, S. Arnaud-Haond, A. Grehan. (2017). The Impact of Anthropogenic Activity on Cold-Water Corals. Marine Animal Forests: The Ecology of Benthic Biodiversity Hotspots. S. Rossi, L. Bramanti, A. Gori and C. Orejas, Springer. DoI: https://doi.org/10.1007/978-3-319-17001-5_27-1
Ross R.E., Nimmo-Smith W.A.M., Howell K.L. (2017) Towards 'ecological coherence': assessing larval dispersal within a network of existing Marine Protected Areas. Deep Sea Research Part I. 126, 128-138. doi:https://doi.org/10.1016/j.dsr.2017.06.004
Sánchez, F., González-Pola, C., Druet, M. , García-Alegre, A., Acosta, J. , Cristobo, J., Parra, S., Ríos, P., Altuna, A., Gómez-Ballesteros, M. , Muñoz-Recio, A, Rivera, J., Díaz del Río, G. 2014. Habitat characterization of deep-water coral reefs in La Gaviera Canyon (Avilés Canyon System, Cantabrian Sea). Deep-Sea Res. II, http://dx.doi.org/10.1016/j.dsr2.2013.12.014i
Somoza, L., Ercilla, G., Urgorri, V., León, R., Medialdea, T., Paredes, M., Gonzalez, F.J., Nombela, M.A. 2014. Detection and mapping of cold-water coral mounds and living Lophelia reefs in the Galicia Bank, Atlantic NW Iberia margin. Marine Geology 349: 73-90.
Sheet reference:
BDC2022/Lophelia pertusa* reefs
* Based on molecular evidence, Addamo et al. (2012 and 2016) proposed that Lophelia be synonymized with Desmophyllum and the species become known as Desmophyllum pertusum. The basic difference between the two taxa is that Desmophyllum is solitary and Lophelia is colonial. Stephen Cairns (Chair of the Department of Invertebrate Zoology at the Smithsonian's National Museum of Natural History) urges caution before accepting this change until more studies are published on the matter. The debate is ongoing and is mentioned here for noting.