Status Assessment 2022 - Sea lamprey
| Assessment of status | Distribution | Population /abundance | Condition (such as breeding productivity) | Previous OSPAR status assessment | Status (overall assessment) | |
|---|---|---|---|---|---|---|
Region | I | ←→ 1,5 | ←→ 1,5 | ←→ 1,5 | ● | Poor |
| II | ←→ 1,5 | ←→ 1,5 | ←→ 1,5 | ● | Poor | |
| III | ←→ 1,5 | ←→ 1,5 | ←→ 1,5 | ● | Poor | |
| IV | ←→ 1,5 | ←→ 1,5 | ←→ 1,5 | ● | Poor | |
| V | NA | |||||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: If in QSR 2010 then enter Regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ) based on Chapter 10 Table 10.1 and Table 10.2 . If a more recent status assessment is available, then enter ‘poor’/’good’
Status*(overall assessment)
red – poor
green – good
? – status unknown
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressure
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threats or impacts (overall assessment)
red – significant threat or impact;
green–no evidence of a significant threat or impact
Blue cells – insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
| Assessment of threats | Barrier to species movement (Obstacles blocking access to upstream migration) | Freshwater habitat loss (Other habitat deterioration) | Removal of target species/non-target species (Fishing at estuary/river, bycatch at sea) | Input of other substances, synthetic and non-synthetic | Food limitation | Climate change | Evidence of threats/impacts | |
|---|---|---|---|---|---|---|---|---|
Region | I | ←→1,5 | ←→5 | NA | ←→1 | ? | ? | |
| II | ←→1,5 | ←→1,5 | ? | ←→1 | ? | ? | ||
| III | ←→1,5 | ←→1,5 | ? | ←→1 | ? | ? | ||
| IV | ←→1,5 | ←→1,5 | ←→1,5 | ←→1 | ? | ←→1,5 | ||
| V | NA | |||||||
Confidence
Few quantitative data exist regarding the abundance of sea lamprey. Thus, the assessment of trends relies to some extent on expert judgement. The confidence in the threat assessment may to some extent be low, because the understanding of singular as well as interactive effects of environmental pollution, harvesting, habitat modification etc. is poor. Nevertheless, the Red List status of sea lamprey in different countries, at least the most recent ones, should be reliable.
Background Information
Year added to OSPAR list: OSPAR 2009
The original evaluation of sea lamprey against the Texel-Faial criteria referred to decline, rarity and sensitivity, with information also provided on threats, was carried out in 2009 (Curd 2009).
- Global/regional importance: Sea lamprey is an anadromous species spawning in rivers. In the OSPAR Maritime Area of concern, the nursery areas for sea lamprey are in the Atlantic Ocean, North Sea, Skagerrak, Kattegat and Mediterranean Sea. There is no estimate of the numbers of adult sea lamprey entering freshwater to spawn within the four OSPAR regions. However, some data exists including those based on expert judgement.
- Decline: The abundance of sea lamprey has declined significantly over the past decades throughout OSPAR Regions I to IV as shown by the national Red List assessments.
- Sensitivity: Sea lamprey was nominated for inclusion on the OSPAR List on the basis of an evaluation of its status according to the Criteria for the Identification of Species and Habitats in need of Protection and their Method of Application (the Texel-Faial Criteria) (OSPAR 2003). Particular reference was given to its decline in recent years/decades, rarity, and sensitivity to different threats from human activities in its freshwater stage. At the southern limits of the Sea lamprey distribution area, the increasing frequency of dry years with prolonged droughts followed by waterway siltation and fires, are expected to severely impact lamprey production (Moser et al., 2020). Dewatering, dredging, hypoxia, chemical pollution, and predation on larvae by native or non-native fish species during their downstream migration. The larval lamprey is considered to be fairly resilient during the period when they are burrowed into the silt of rivers and streams, for several years (cf. Clemens 2019). However, during this larval stage, dredging activities can be a considerable threat.
- Anthropogenic pressures and biological factors: The principal threats to sea lamprey are obstacles blocking access to spawning areas, destruction and removal of suitable spawning and larval habitat, reduction of water flow, overexploitation, illegal fishing in brackish and fresh waters and poor water quality. fishing at estuaries and rivers, bycatch at sea and pollution are much more serious threats in southern regions of OSPAR.
Last status assessment: OSPAR (2009) concluded that sea lamprey qualified under its criteria due to a strong and continuously population decline in recent years, uncontrolled threats and inadequate conservation measures.
Geographical Range and Distribution
The sea lamprey is an anadromous species distributed on both sides of the North Atlantic (Wilson & Veneranta 2019 (Eds.)) (Figure 1 and Figure 2). In North America, it occurs on the east coast from Labrador (Canada) in the north (53°N) to Florida (USA) in the south (30°N). In Europe, it can be found from the Barents Sea (70°N) in the north, to the Iberian Peninsula (38°N) in the southwest, and to the Adriatic Sea (40°N) in the southeast. Occasionally, it is also found in the Aegean Sea and the Levantine Sea, i.e. in eastern Mediterranean. It also occurs off Iceland, Greenland, and in the North Sea. However, it is rare in the Baltic Sea. Sea lamprey has occasionally also been found at lower latitudes in northern Africa. Several landlocked populations of sea lamprey inhabit the North American Great Lakes, but to our knowledge none in Europe (Kottelat & Freyhof 2007). In the Iberian Peninsula, it occurs in most major rivers flowing into the Cantabrian Sea and the Atlantic Ocean (Mateus et al., 2012), as well as some into the Mediterranean.
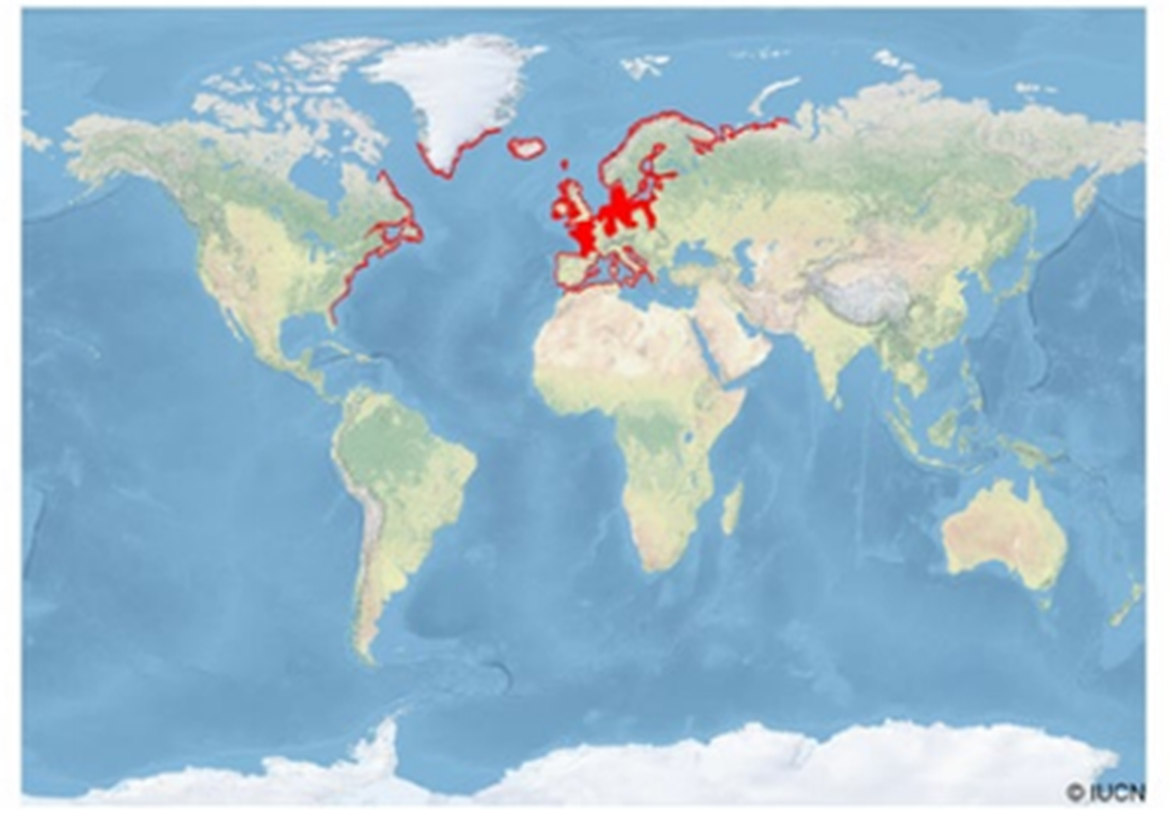
Figure 1: The distribution of sea lamprey worldwide
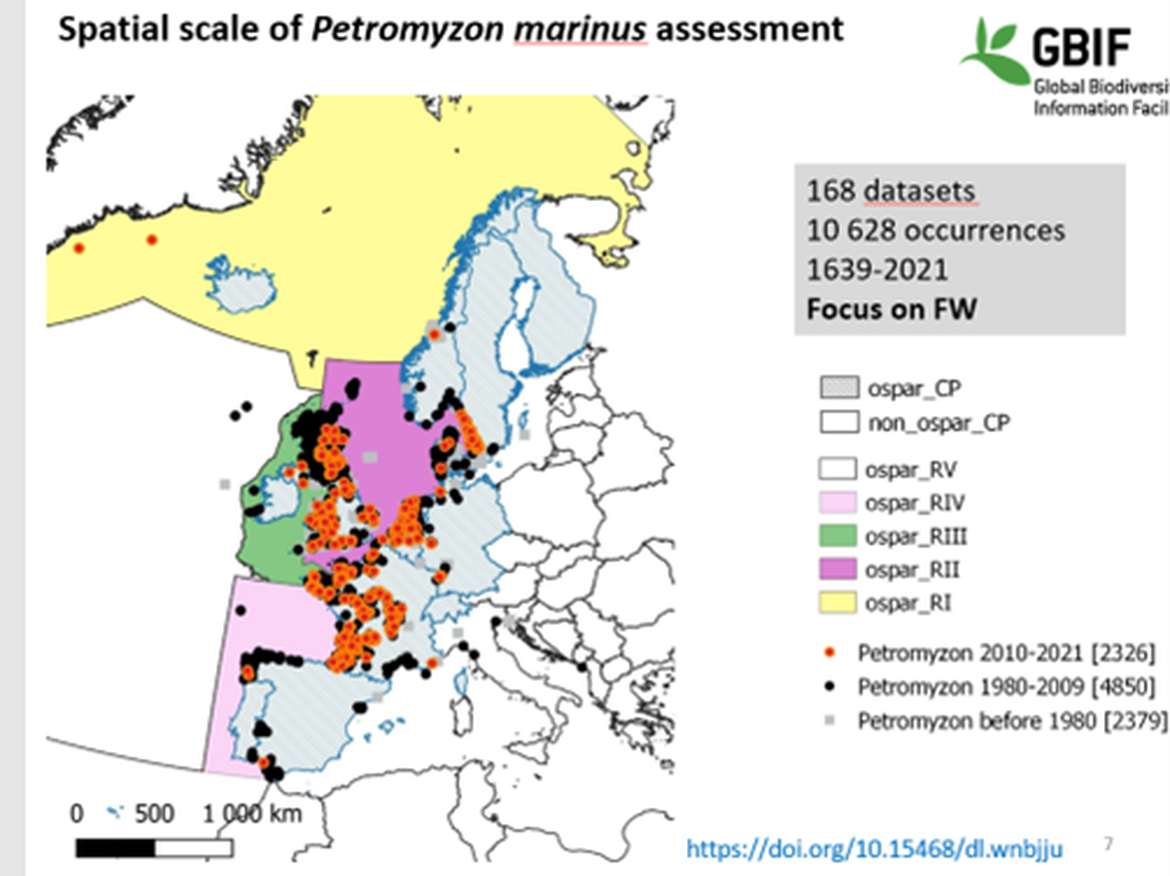
Figure 2 The distribution of sea lamprey in more detail in Europe based on different sources between 1980 and 2021.
Reference: GBIF, Global Biodiversity Information Facility. This map is missing the presence in some rivers in Portugal: Minho, Lima, Cávado, Douro, Vouga, Mondego, Tagus and Guadiana (Pedro R. Almeida, pers. comm.) and rivers in northern Norway (Hesthagen et al. 2020).
Population/Abundance
The status of sea lamprey within the OSPAR Maritime Area has been continuously deteriorating since the last assessment in 2009. However, recent trends throughout OSPAR Regions I to IV may differ. In Sweden, the Red List status has changed in recent years from Nearly Threatened in 2015 to Endangered in 2020. In the Netherlands, the number of sea lamprey have declined steadily since 1960, with minimum numbers reached in the 1970s and 1980s. However, they never completely disappeared from the Meuse and the Rhine. Since 1990, there has been a slight, but continuous increase in numbers in some rivers. Historical datasets for four European rivers up to late 2000 (Adour, Garonne, Vilaine and Rhine) show an increased number of sea lamprey since the 1990s (Beaulaton et al., 2008).
Regarding the most important populations of sea lamprey, which are concentrated in the southwestern regions of the continent (north–central Portugal, north–northwest of Spain, and west–southwest of France; Beaulaton et al. (2008), Mateus et al. (2012), Silva et al. (2016)), the Galician rivers present the highest densities and biomasses of larvae recorded in Europe. Between 2007 and 2011, Silva et al. (2016) reported signs of population increase. However, this trend requires further confirmation.
In France, data from fish pass monitoring in the upstream reaches of the Garonne and Dordogne rivers show a dramatic decrease from tens of thousands of adult lampreys during the 2000s, to nearly zero at present (Gracia et Caut 2015, Carry et al., 2017).
In Portugal, sea lampreys were historically reported from Minho, Lima, Neiva, Cávado, Ave, Douro, Vouga, Mondego, Tagus, and Guadiana basins (Baldaque da Silva 1891). Presently, they have disappeared from River Ave and River Neiva and considering the official fishing records, lamprey catches are mostly (about 85%) from the northern and central rivers (Minho, Lima, Vouga and Mondego rivers), that have been previously identified as distinct management units (Lança et al. 2014). In Mondego River where several measures implemented to improve river connectivity took place, sea lamprey larvae abundance upstream from the first obstacle to migration located 45 km from the river mouth increased by about 40% from 2011 to 2016 (Moser et al., 2020), reaching a peak of 100 fold in 2019 (Pedro R. Almeida, unpublished data). However, since 2015, monitoring data from the fish pass placed at this obstacle showed a substantial decrease in total numbers of adult spawners in several years.
Current management and restoration measures appear so far to be highly insufficient to allow for a full recovery of sea lamprey in the OSPAR Regions.
Condition
The impact of human activities has led to a drastic restriction and fragmentation of the distribution area of sea lamprey in freshwaters and has contributed to its placement on the Red list of threatened species in Europe (see Beaulaton et al., 2008, Curd 2009, Wilson & Veneranta 2019 (Eds.). Some Red list assessments in OSPAR Regions I to IV indicate that the species is still under threat (Table 1). In Northern Europe, sea lamprey is Near Threatened in Norway and Endangered in Sweden. In Denmark, sea lamprey is rare and historical descriptions indicates a population decline in at least some rivers (Olesen et al., 2008, 2009). In 2010 it was also considered rare and regarded as Vulnerable. However, in 2019 their status was stated as Data Deficient (DD) (Carl & Møller 2019). In the Helcom area, sea lamprey was classified as Endangered in 2007, but had improved to Vulnerable in 2013. (HELCOM 2013). In Germany, Wales, Republic of Ireland and France, sea lamprey is classified as Near Threatened. In Wales, sea lamprey is regarded as Near Threatened. In England and Scotland, the status is unknown, but the same status is presumed. In Belgium, sea lamprey is either Extinct (Flanders) or Regionally Extinct (Wallonia). In Spain and Portugal, sea lamprey is classified as Vulnerable, except for some populations inhabiting rivers in the southern basins of Spain where it is Endangered.
| Country | Second most recent assessment (year) | Status at the second most recent assessment | Recent assessment (year) | Status at the most recent assessment |
|---|---|---|---|---|
| Norway | 2015 | NT | 2021 | NT |
| Sweden | 2015 | NT | 2020 | EN |
| Denmark | 2010 | VU | 2019 | DD |
| Germany | 1998 | EN | 2009 | NT |
| Belgium, Wallonia | 2010 | RE | ||
| Belgium,Flanders | 2014 | EN | ||
| The Netherlands | 1997 | EN | ||
| Scotland | 2021* | NT* | ||
| England | 2021* | NT* | ||
| Wales | 2021 | NT | ||
| N. Ireland | 2021 | EN | ||
| Rep. Ireland | 2011 | NT | ||
| France | 2005 | VU | 2009 | NT |
| Spain | 2001 | VU | 2011 | EN |
| Portugal | 2005 | VU |
Threats and Impacts
Obstacles to migration, commercial fishing and illegal fishing in brackish waters as well as in rivers are the main threats to sea lamprey in southern areas such as in Portugal (Pereira et al., 2017, Clemens et al., 2020, Stratoudakis et al., 2020, Almeida et al. 2021).
In freshwater, along with river fragmentation by dams and weirs, habitat degradation associated with dewatering, dredging, hypoxia and degraded water quality (Wilson & Veneranta 2019 (Eds.) are among the main threats associated with the loss of suitable spawning and larval habitat. Moreover, increasing frequency of dry years with prolonged droughts followed by waterway siltation and fires, are expected to severely impact sea lamprey productivity (Moser et al., 2020).
In Norway and Sweden, barriers (dams and weirs etc.) blocking access to spawning sites and changes of the hydrodynamic regimes for hydroelectrical purposes are considered to be the most serious threat. This will reduce the habitat available for the larval stage and thus recruitment with effects not only on larval survival but also on spawning possibilities.
In Germany, obstacles to migration, canalized rivers with high water flow velocities, and loss of large shark, bony fish and marine mammals for attachment in the ocean are the main threats.
Measures that address key pressures from human activities or conserve the species/habitat
Analysis of yield data from the commercial fisheries in either the ocean or rivers may detect possible over-exploitation and implementation of a proper adaptative fishery management for sea lamprey. Fishways monitoring can provide crucial information regarding fishways efficiency and contribute to mitigate impacts associated with barriers to migration. The distribution of spawners and redds along rivers may point to problems of accessibility and inability to colonize rivers to their capacity (Kelly & King 2001, Igoe et al., 2004, King et al., 2011). Thus, redds and adults may be concentrated downstream of major weirs, being restricted to the lower reaches of rivers. Few data exists on the breeding success. However, low densities of larvae are a measure of either habitat disruption of spawning grounds or high mortality due to poor water quality. In the River Ave in northern Portugal, the sea lamprey population has now vanished, probably due to pollution (Mateus et al., 2012). Industrial pollution is probably also responsible for the extremely low density of sea lamprey larvae in the lower reaches of the River Cávado (Almeida et al., 2002).
Conclusion (including management considerations)
No improvement in the status of sea lamprey in OSPAR Regions I to IV since the first assessment in 2009 can be documented. Thus, the present assessment confirms that sea lamprey is still a threatened and declining species. This is evident both in terms of Red List assessments and distribution. Barriers/obstacles, habitat degradation, over-harvesting and pollution remain the most serious threats for sea lamprey populations. Climate change might also be a threat in some areas.
There has been an increased focus on sea lamprey during recent years, and several research programmes have been initiated. However, in some regions/countries there is a lack of sufficiently long time series to be able to detect consistent trends in the number of spawners. Furthermore, national legislation and different mitigation measures have been introduced in some European countries, to sustain sea lamprey numbers and to protect their habitat (Stratoudakis et al., 2016, Clemens et al., 2020). Several countries have also initiated monitoring programs, e.g. Sweden, Wales and Portugal (Stratoudakis et al., 2016, 2020).
Habitat restoration only takes place in a few countries. Barrier removal should benefit sea lamprey by increasing available habitat. Fishways vary in effectiveness and are often inadequate. Thus, long-term monitoring programs should ensure the correct assessment of its efficiency and identification of proper adjustments. This implies research opportunities encompassing ecohydraulics, biotelemetry and engineering (Lucas et al., 2020).
Water quality should be monitored and improved if necessary.
Maintenance of a sustainable fishery and stocks must also be encouraged, and legislation should be carefully revised in all countries holding sea lamprey fishery. An adaptive fishery management could benefit both conservation and exploitation of sea lamprey. At present, specific conservation measures targeted at protecting sea lamprey are now initiated and different threats should be reduced. Thus, sea lamprey should still be included in the OSPAR List of Declining Species.
Knowledge Gaps
Data on abundance and stock structure of sea lamprey in different regions of OSPAR should be improved in order to assess trends. Sea lamprey status in the Red List is presently to a large extent based on expert judgement. Furthermore, several countries have not carried out any assessment over the past 10 to 15 years. Estimates of stage-specific mortality, distribution at sea and preferred host are required. Overfishing may affect sea lamprey in some areas where it is still commercially important (Wilson & Veneranta 2019 (Eds.), Almeida et al., 2021). However, there is insufficient reliable data on the exploitation rates and the impact of illegal fishing. Furthermore, optimizing sea lamprey passage through different fishways should be a main concern (Pereira et al., 2017, 2019). Any impact of climate change-induced flow alteration and increased temperatures in rivers should be studied. Its impacts on species distribution and availability of hosts must also be better understood. The parasitic oceanic life stage is still a “black box” for anadromous lampreys in general. For a global overview of the pressures that affect this species abundance more information is needed concerning the dynamic lamprey-host interactions, combined with studies on oceanographic limiting factors affecting the number of spawners (Quintella et al., 2021).
Method used
Main source of information:
Assessment is based on a mixture of information given by expert appointed by OSPAR delegates in each country and review of relevant literature. In addition, a questionnaire was sent to all 13 partners (See additional information concerning methods used). OSPAR did not appoint any experts for Belgium and Spain. Thus, our assessment needed to some extent to rely on written sources available, which may result in insufficient evaluation. Information given by these scientists were highly valuable; Germany by M. Müller and R. Fricke , Northern Ireland by Dennis Ensing, Portugal by Pedro R. Almeida, Catarina S. Mateus, Esmeralda Pereira, Sílvia Pedro and Bernardo R. Quintella, Sweden by Mikael Svensson and Wales by Tristan Hatton-Ellis, Alex Scorey & Ida Nielsen. Pedro R. Almeida and Mikael Svensson gave valuable comments on a draft of the report.
Almada, V. C., Pereira, A. M., Robalo, J. I., Fonseca, J. P., Levy, A., Maia, C. & Valente, A. 2008. Mitochondrial DNA fails to reveal genetic structure in sea-lampreys along European shores. Molecular Phylogenetics and Evolution 46: 391-396.
Almeida, P.R, Quintella, B,R., Dias, N.M. & Andrade, N. 2002. The anadromous sea lamprey in Portugal: biology and conservation perspectives. In: Moser M, Bayer J, MacKinlay D (Eds.). The biology of lampreys, symposium proceedings, International Congress on the Biology of Fish, American Fisheries Society, 21–25 July, Vancouver, British Columbia, pp. 49–58.
Almeida, P.R., Arakawa, H., Aronsuu, K., Baker, C., Blair, S-R, Beaulaton, L., Belo, A.F., Kitson, J., Kucheryavyy, A., Kynard, B., Lucas, M.C., Moser, M., Potaka, B., Romakkaniemi, A., Staponkus, R., Tamarapa, S., Yanai, S., Yang, G., Zhang, T. & Zhuang, P. 2021. Lamprey fisheries: history, trends and management. J. Great Lakes Res. in press.
Araújo, M. J., Silva, S., Stratoudakis, Y., Gonçalves, M., Lopez, R., Carneiro,M., et al. 2016. Sea lamprey fisheries in the Iberian Peninsula. Jawless Fishes World 2: 115-148.
Baldaque da Silva, A. A. 1891. Estado actual das pescas de Portugal. Imprensa. acional, Lisbon (this reference is given by Pedro R. Almeida).
Beaulaton, L., Taverny, C. & Castelnaud, G. 2008. Fishing, abundance and life history traits of the anadromous sea lamprey (Petromyzon marinus) in Europe. Fish. Res. 92: 90-101.
Bouletreau, S, Carry, L., Meyer, E., Filloux, D., Menchi, O, Mataix, V. & Santoul, F. 2020. High predation of native sea lamprey during spawning migration. Scientific Report 10 (6122): 1-9.
Bracken, F.S., Rooney, S.M., Kelly-Quinn, M. & King, J.J. 2018. Identifying spawning sites and other critical habitat in lotic systems using eDNA “snapshots”: A case study using the sea lamprey Petromyzon marinus L. Ecol. Evol. 9: 553-567.
Carry L, Filloux, D, Caut, I 2017. Suivi de la lamproi marine sur la Dordogne et la Garonne. MIGADO –Migrateurs Garonne Dordogne.
Carl, H. & Møller, P.R. 2019. Fisk. In: Moeslund, J.E. et al. (eds.). Den danske Rødliste 2019. Aarhus Universitet. DCE- Nationalt Center for Miljø og Energi.redlis.au.dk.
Clemens, B.J. 2019. A call for standard terminology for lamprey life stages. Fisheries 44: 243–245.
Clemens, B.J., Arakawa, H., Baker, C., Coghlan, S., Kucheryavyy, A., Lampman, R., Lança, M.J., Mateus, C.S., Miller, A., Nazari, H., Pequeño, G., Sutton, T.M. & Yanai, S. 2020. Management of anadromous lampreys: Common threats, different approaches. J. Great Lakes Res. in press.
Curd, A. 2009. Background document sea lamprey Petromyzon marinus. OSPAR Commission Biodiversity Series Publication Number: 431/2009. New Court, London. United Kingdom.
Davies, C.E, Shelley, J, Harding, P.T., Mclean, I.F.G., Gardiner, R. & Peirson, G (eds.). 2004. Freshwater fishes in Britain. The species and their distribution. Harley Books, Colchester.
Davies, R. 2016. Sea lamprey monitoring on the River Tywi 2011-2014. Natural Resources Wales, St Mellons, Cardiff. Ref no: NFAT/16/02. 18 pp.
Goodwin, CE., Dick, J.T.A. & Elwood, R.W. 2009. A preliminary assessment of the distribution of the sea lamprey (Petromyzon marinus L), river lamprey (Lampetra fluviatilis (L.)) and brook lamprey (Lampetra planeri (Bloch)) in Northern Ireland. Biology and Environment: Proceedings of the Royal Irish Academy 109B, 47-52; DOI: 10.3318/BIOE.2009. 109.1.47
Havs- och vattenmyndigheten 2020. Åtgärdsprogram för havsnejonöga (Petromyzon marinus). (Sea lamprey - Single species action plan.) In Swedish with English summary.
HELCOM. 2013. Species information sheet Petromyzon marinus. Helcom red list fish and lamprey species expert group. www.helcom.fi
Hesthagen, T, Vøllestad, A. & Økland, F. 2020. Distribution of sea lamprey Petromyzon marinus in Norwegian rivers. Fauna 73 (3-4): 66-81. In Norwegian with English summary.
Gracia S. & Caut, I. 2015. Suivi de la lamproie marine sur la Dordogne 2014. MIGADO - Migrateurs Garonne Dordogne.
Igoe, F., Quigley, D.T.G., Marnell, F., Meskell, E., O’Connor, W. & Byrne, C. 2004. The sea lamprey Petromyzon marinus (L.), river lamprey Lampetra fluviatilis (L.) and brook lamprey Lampetra planeri (Bloch) in Ireland: general biology, ecology, distribution and status with recommendations for conservation. Biology and Environment: Proceedings of the Royal Irish Academy 104B: 43–56.
Kelly, F.L. & King, J.J. 2001. A review of the ecology and distribution of three lamprey species, Lampetra fluviatilis (L), Lampetra planeri (L.), and Petromyzon marinus (L.): a context for conservation and biodiversity considerations in Ireland. Biology and Environment: Proceedings of the Royal Irish Academy. 101B (3):165-185.
King, J.L., Marnell, F., Kingston, N., Rosell, R., Boylan, P., Caffrey, J.M., FitzPatrick, Ú., Gargan, P.G., Kelly, F.L., O’Grady, M.F., Poole, R., Roche, W.K. & Cassidy, D. 2011. Ireland Red List No. 5: Amphibians, Reptiles & Freshwater Fish. National Parks and Wildlife Service, Department of Arts, Heritage and the Gaeltacht, Dublin, Ireland.
Kottelat, M. & Freyhof, J. 2007. Handbook of European Freshwater Fishes. Kottelat, Cornol, and Freyhof, Berlin. 646 pp. https://doi.org/10.1643/ot-08-098a.1
Lança MJ, Machado M, Mateus CS, Lourenço M, Ferreira AF, Quintella BR, et al. 2014. Investigating Population Structure of Sea Lamprey (Petromyzon marinus, L.) in Western Iberian Peninsula Using Morphological Characters and Heart Fatty Acid Signature Analyses. PLoS ONE 9(9): e108110. https://doi.org/10.1371/journal.pone.0108110
Lassalle, G., Béguer, M., Beaulaton, L. & Rochard, E. 2008. Diadromous fish conservation plans need to consider global warming issues: an approach using biogeographical models. Biol. Conserv. 141: 1105-1118.
Lucas, M. C., Bubb, D. H., Jang, M-H., Ha, K. & Masters, J. E. G. 2009. Availability of and access to critical habitats in regulated rivers: effects of low-head barriers on threatened lampreys. Freshwat. Biol. 54: 621-634. https://doi.org/10.1111/ j.1365-2427.2008.02136.
Lucas, M.C., Hume, J.B., Almeida, P.R. et al. 2020. Emerging conservation initiatives for lampreys: Research challenges and opportunities. J. Great Lakes Res. https://doi.org/10.1016/j.jglr.2020.06.004
Maitland, P. S., Renaud, C. B., Quintell, B. R. & Close, D. A. 2015. Conservation of native lampreys. In Lampreys: Biology, Conservation and Control, pp. 375–428. Ed. by M. F. Docker. Springer Fish and Fisheries Series, Dordrecht. 438 pp. https://doi.org/10.1007/978-94-017-9306- 3
Mateus, C. S., Rodríguez-Muñoz, R., Quintella, B.R., Alves, M, J. & Almeida, P. R. 2012. Lampreys of the Iberian Peninsula: distribution, population status and conservation. Endangered Species Res. 16: 183–198.
Mateus C. S., Docker M. F., Evanno G., Hess J. E., Hume J. B., Oliveira I. C., Souissi A. & Sutton T.M. 2021. Population structure in anadromous lampreys: Patterns and processes. J. Great Lakes Res. in press.
Moser, M.L, Almeida, P.R. King, J.J. & Pereira, E. 2020. Passage and freshwater habitat requirements of anadromous lampreys: Considerations for conservation and control. Journal of Great Lakes Research. https://doi.org/10.1016/ j.jglr.2020.07.011
Lassalle, G., Béguer, M., Beaulaton, L., Rochard, E., 2008. Diadromous fish conservation plans need to consider global warming issues: an approach using biogeographical models. Biol. Conserv. 141: 1105–1118.
Niven, A.J. & McCauley, M. 2013. Lamprey Baseline Survey No3: River Foyle and Tributaries SAC. Loughs Agency, 22, Victoria Road, Derry~Londonderry
O’Connor, W. & Byrne, C. 2004. The sea lamprey Petromyzon marinus (L.), river lamprey Lampetra fluviatilis (L.) and brook lamprey Lampetra planeri (Bloch) in Ireland: general biology, ecology, distribution and status with recommendations for conservation. Biology and Environment: Proceedings of the Royal Irish Academy 104B: 43–56.
Olesen, T.M., Aarrestrup, K., Lassen, H.H., Jessen, B.H. & Carl, H. 2008. Eftersøgning af havlampret (Petromyzon marinus Linnaeus 1758 på gydevandring. Flora og Fauna 114 (1) 1-7. In Danish with English summary.
Olesen, T.M., Carl, H. & Aarrestrup, K. 2009. Havlampret (Petromyzon marinus Linnaeus 1758) in danske vandløp 1869-2009. Flora og Fauna 115 (2-3):45-60.In Danish with English summary
Pereira, E., Quintella, B.R., Mateus, C.S., Alexandrade, C.M., Belo, A.F., Telhado, A., Quadrado, M.F. & Almeida, P.R. 2017. Performance of a vertical-slot fish pass for the Sea Lamprey Petromyzon marinus L. and habitat recolonization. River Res. Appl. 33.
Pereira, E.; Cardoso, G.R.; Quintella, B.R., Mateus, C.S., Alexandre, C.M., Oliveira, R.L.; Belo, A.F.; et al. 2019. Proposals for optimizing sea lamprey passage through a vertical-slot fishway. Ecohydrology 12 4: e2087. http://dx.doi.org/10.1002/eco.2087.
Quintella, B.R., Póvoa, I & Almeida P.R 2009. Swimming behaviour of upriver migrating sea lamprey assessed by electromyogram telemetry. J. Appl. Ichthyol. 25: 46-54.
Quintella B.R., Clemens B.J., Sutton T., Lança M.J., Madenjian C.P., Happel, A. & Harvey, C.J. 2021. At-sea feeding ecology of parasitic lampreys. J. Great Lakes Res. in press.
Silva, S., Vieira-Lanero, R., Barca, S. & Cobo, F. 2016. Densities and biomass of larval sea lamprey populations (Petromyzon marinus Linnaeus, 1758) in north-western Spain and data comparisons with other European regions. Marine and Freshwat. Res. 68: 116–122 https://doi.org/10.1071/mf15065
Sorensen, L. P. & Hoye, T.R. 2007.A critical review of the discovery and application of a migratory pheromone in an invasive fish, the sea lamprey Petromyzon marinus. J. Fish Biol. (Supplement D) 71: 100-114.
Spice, E.K., Goodman, D.H., Reid, S.B. & Docker, M.F. 2012. Neither philopatric nor panmictic: microsatellite and mtDNA evidence suggests lack of natal homing but limits to dispersal in Pacific lamprey. Mol. Ecol. 21 (12): 2916-2930.
Stratoudakis, Y.; Mateus, C. S.; Quintella, B. R.; Antunes, C. & Almeida, P.R. 2016. Exploited anadromous fish in Portugal: Suggested direction for conservation and management. Marine Policy 73: 92-99.
Stratoudakis, Y., Correia, C., Belo, A.F. & Almeida, P.R. 2020. Improving participated management under poor fishers’ organization: Anadromous fishing in the estuary of Mondego River, Portugal. Marine Policy 119. https://doi.org/10.1016/j.marpol.2020.104049 104049
Wagner, C.M., Jones, M.L. Twohey,M.B. & Sorensen, P.W. 2006. A field test verifies that pheromones can be useful for sea lamprey (Petromyzon marinus) control in the Great Lakes. Can. J. Fish. Aquat. Sci. 63: 475–479
Waldman, J., Grunwald, C. & Wirgin, I. 2008. Sea lamprey Petromyzon marinus: an exception to the rule of homing in anadromous fishes. Evol. Biol. doi.org/10.1098rsbl.2008.0341
Wilson, K. & Veneranta, L. (Eds.). 2019. Data-limited diadromous species – review of European status. ICES Cooperative Research Report No. 348. 273 pp. https://doi.org/10.17895/ices.pub.5253
Zancolli, G., Foote, A., Seymour, M. & Creer, S. 2018. Assessing lamprey populations in Scottish rivers using eDNA: proof of concept. Scottish Natural Heritage Research Report No. 984.
Sheet reference:
BDC 2022/sea lamprey Petromyzon marinus