Status Assessment 2022 - Zostera beds
| Assessment of status | Distribution | Extent | Condition | Previous OSPAR status assessment | Status (overall assessment) | |
|---|---|---|---|---|---|---|
Region | I | ↓ | ↓ | ↓ | ● | Poor1, 5 |
| II |  |  |  | ● | Poor1, 5 | |
| III | ←→ | ←→ | ←→ | ● | Poor1, 4, 5 | |
| IV | ←→ | ↓ | ←→ | ● | Poor1, 4, 5 | |
| V | NA | |||||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed

chaos symbol demonstrating that the available information is pointing in all directions within the specific region
Previous status assessment: If in QSR 2010 then enter Regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ) based on Chapter 10 Table 10.1 and Table 10.2 . If a more recent status assessment is available, then enter ‘poor’/’good’
Status*(overall assessment)
red – poor
green – good
? – status unknown
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressure
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain

chaos symbol demonstrating that the available information is pointing in all directions within the specific region
NA - Not applicable
Threats or impacts (overall assessment)
red – significant threat or impact;
green–no evidence of a significant threat or impact
Blue cells – insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
| Assessment of threats | Nutrient enrichment | Fishing pressure | Tourism and recreational activities | Changes in suspended solids | Extractions and installations | Temperature increases | Introduction or spread of non-indigenous species | Threat or impact | |
|---|---|---|---|---|---|---|---|---|---|
Region | I | ↑ | ↓ | NA | NA | ↑ | ↑ | NA | Significant threat1, 5 |
| II |  | ↑ | ↑ | ↑ | ←→ | ↑ | ↑ | Significant threat1, 4, 5 | |
| III | ↑ | ↑ | ↑ | ? | ? | ↑ | ↑ | 1, 4, 5 | |
| IV | ? | ←→ | ↓ | ? | ? | ↑ | ↑ | 1, 4, 5 | |
| V | NA | ||||||||
Confidence
High – distribution and extent
Medium – condition
For threats, there is a high confidence on some topics and medium on others
Background Information
Year added to OSPAR List: Zostera beds were added to the OSPAR List of threatened and/or declining species and habitats in 2008 (OSPAR 2008, OSPAR 2009)
- The habitat has exhibited long-term and large-scale decline and is threatened in (at least parts of) all four OSPAR Regions (I to IV) where it occurs, with most evidence of declines from the Greater North Sea area (Region II). The basic criteria for the original nomination were decline, ecological significance and sensitivity, with information also provided on threats.
- In the previous assessment, habitat damage, nutrient and organic enrichment, hazardous substances, microbial pathogens, siltation rate changes, climate change, introduction and competition from alien species and removal of target and non-target species were all listed as factors that affect Zostera beds and can threaten the extent and quality of this habitat (Anon 2000, OSPAR 2009). The previous assessment highlighted that the evidence of decline in Zostera beds was extensive in specific locations within the OSPAR Maritime Area, with the most detailed studies revealing a decline in the Greater North Sea area (Den Hartog & Polderman, 1975, Rasmussen, 1977, Davison & Hughes, 1998, Jones et al., 2000, Baden et al., 2003, Boström et al., 2003). Factors that threaten Zostera beds were said to vary in intensity across space, but most of them occur throughout the OSPAR Maritime Area (OSPAR, 2009).
Geographical Range and Distribution
Of the two major Zostera species, Zostera marina and Z. noltii, Z. marina is more widespread. It has a distribution within the OSPAR Maritime Area extending from the Arctic (the OSPAR parts of Greenland are unknown with regard to Zostera occurrence although Z. marina occurs in south-western Greenland (see Olesen et al., 2015 and Marbá et al., 2018) down to Gibraltar (see the map). Zostera marina is generally found in the lower intertidal and the subtidal zone, although it can also be found in the intertidal zone in many places such as in France (Boyé et al., 2021). Zostera noltii is less common, lives higher up on the shore compared to Z. marina, and does not occur further north than northern Scotland and the Bergen area in south-western Norway.
Presence/absence and areal distribution of seagrasses are commonly used indicators of status and change in distribution at the landscape scale. Colonisation depth is another useful distribution indicator (Borum et al., 2004, OSPAR 2009, Marbá et al., 2013), which is tightly coupled with water clarity (Duarte, 1991). Over a longer 100-year perspective, there have been huge decreases in Zostera distribution in the northern region including in Denmark, Sweden, Germany, the Netherlands and France (Airoldi & Beck, 2007, Boström et al., 2014, Godet et al., 2008, de los Santos et al., 2019, Krause-Jensen et al., 2021). Over a shorter 10-year perspective, there are signs of further decreases in some of the northern parts of the OSPAR Maritime Area such as in Norway and Sweden (Moksnes et al., 2018, 2021, Rinde et al., 2021) as well as in certain areas around the British Isles (Jones & Unsworth 2016, NPWS 2019, Green et al., 2021), whereas in Denmark, Netherlands and France the distribution trend is uncertain, stable or may even display a slight increase (Wilkes et al., 2017, Auby et al., 2018, 2020ab, Marbá et al., 2018, Zwarts et al., 2018, de los Santos et al., 2019, Van Deelen et al., 2019, Krause-Jensen et al., 2020, 2021, Aubert et al., 2021ab, Aubin et al., 2021, Grall et al., 2021, Guerrero-Meseguer et al., 2021, Hansen & Høgslund 2021, Moffat et al., 2021, Naustvoll et al., 2021, Ollivier et al. 2021, Schutter et al. 2021, see also the table on distribution above).
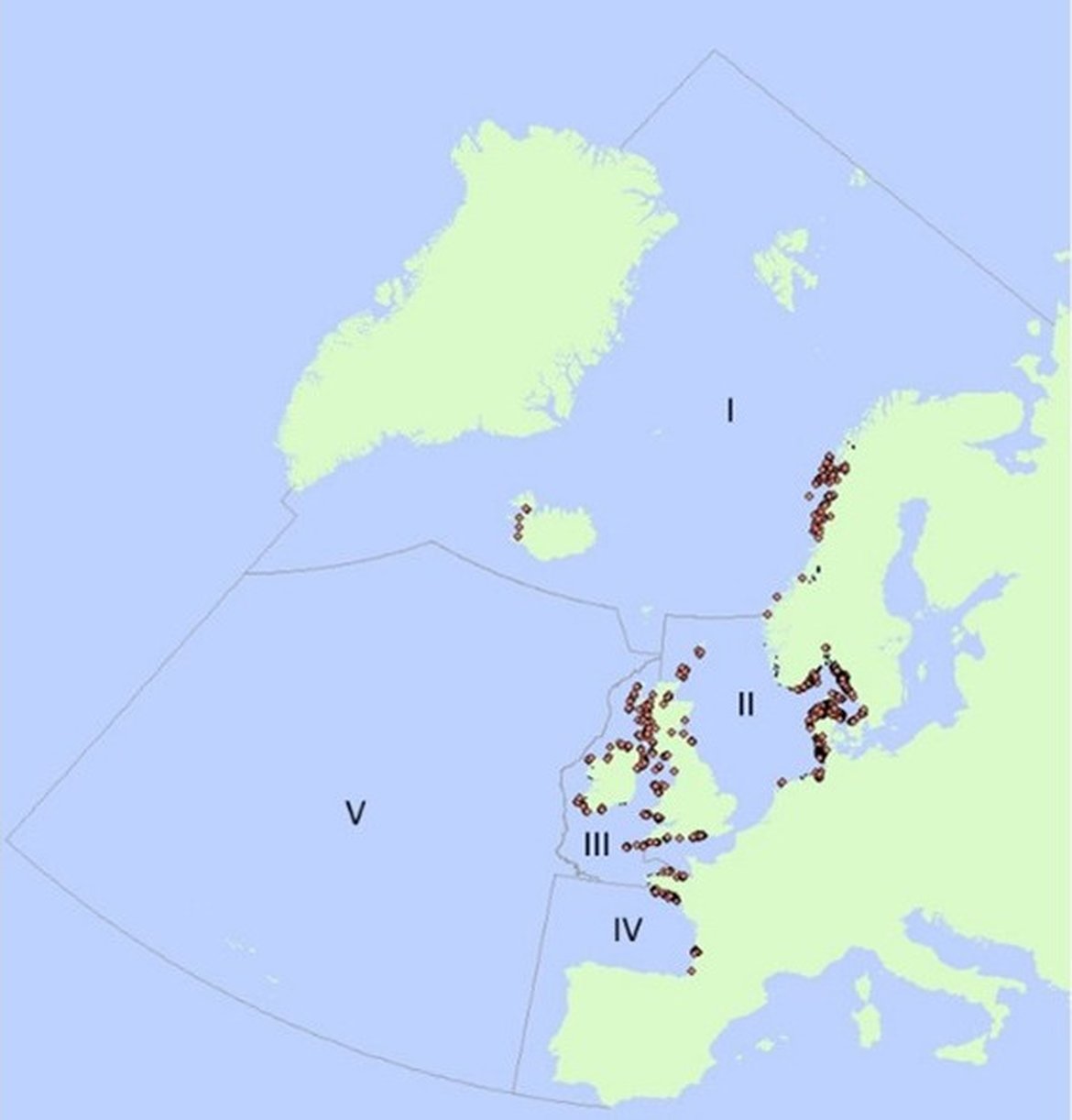
Figure 1: Documented Zostera bed distribution within the OSPAR Maritime Area. Data from OSPAR threatened and/or declining species and habitats database.
For Regions I and II: Recent information from the Nordic region and the Arctic (including the White Sea) is available in Boström et al. (2014), Krause-Jensen et al. (2020), Frigstad et al. (2021). For Spain and Portugal in Region IV, some information is available in García-Redondo et al. (2019).
Extent
Seagrass abundance/extent is often measured as coverage, biomass or shoot density, while local extent of meadows can be measured as depth limit or meadow size/area (Borum et al., 2004, OSPAR, 2009, Marbá et al., 2013). At a spatial scale of the OSPAR Maritime Area, there have been clear decreases in extent until the late 20th century, however more recently there have been signs of some recovery/increase e.g. in the Norwegian part of Skagerrak, Wales, Ireland and France (de los Santos et al., 2019).
Over a 10-year perspective there is still evidence of clear continued decreases in extent in some countries/areas, e.g. in parts of southern Norway, Sweden and some areas around the British Isles (e.g. Jones & Unsworth 2016, Ribaudo et al., 2016, Moksnes et al., 2018, 2021, NPWS 2019, Green et al., 2021, Rinde et al., 2021), whereas in other areas, the trend differs between sites and can be characterized as uncertain, stable or in some places even showing an increase (Unsworth et al., 2017, Wilkes et al., 2017, Auby et al., 2018, 2020ab, Burton et al., 2018, Zwarts et al., 2018, de los Santos et al., 2019, Van Deelen et al., 2019, Krause-Jensen et al., 2020, 2021, Aubert et al., 2021ab, Aubin et al., 2021, Boyé et al., 2021, Grall et al., 2021, Guerrero-Meseguer et al., 2021, Hansen & Høgslund 2021, Moffat et al., 2021, Naustvoll et al. 2021, Ollivier et al., 2021, Schutter et al., 2021).
The main causes for decline are wasting-disease, nutrient enrichment, marine pollution, reduced water quality and destruction by anthropogenic activities such as construction work, mooring, anchoring and fisheries (D’Avack et al., 2015, Jones & Unsworth 2016, Eriander et al., 2017, Wilkes et al., 2017, Branco et al., 2018, Cognat et al., 2018, Frigstad et al., 2018, Jones et al., 2018, Moksnes et al., 2018, 2021, Gardiner, 2020, Ouisse et al., 2020, Román et al., 2020, Bertelli et al., 2021, Garmendia et al., 2021).
Patchy and sparse Zostera beds with a cover of <60% may suffer greater losses e.g. during storms than do dense, more stable uniform beds with self-protective properties (e.g., Maxwell et al., 2016). While reductions in the extent of Zostera beds may be rapid, recolonization may require much longer time periods (Maxwell et al., 2016, Moksnes et al., 2018) however this depends on the species and the local context (Kilminster et al. 2015, O´Brien et al., 2017, Boyé et al., 2021). Further details on extent are given in the table above.
Condition
There is a general lack of detail with regard to Zostera bed condition in the OSPAR Maritime Area. The condition of Zostera beds is also characterized as notoriously hard to assess (Kilminster et al., 2015, Boyé et al., 2021). Along the coast of mid-Norway, in the Oslofjord and in certain areas around the British Isles, there are signs of decreases in Zostera bed condition (e.g., Jones & Unsworth, 2016, NPWS, 2019, Green et al., 2021, Rinde et al., 2021). The trends in condition for Sweden, Wales, Denmark are uncertain (e.g., Jones & Unsworth, 2016, Unsworth et al., 2017, Bertelli et al., 2021). The condition seems to be stable in Scotland, although there are examples of declines in Ireland (Region III), in the Netherlands (Region II) and in parts of France (e.g., Wilkes et al., 2017, Bertelli et al., 2021, Boyé et al., 2021). The main causes for decline in condition are wasting-disease, nutrient enrichment, marine pollution, reduced water quality and light climate, destruction by anthropogenic activities such as construction work, mooring, anchoring, fisheries and temperature increases (Bockelmann et al., 2013, Jones & Unsworth, 2016, Cognat et al., 2018, Moksnes et al., 2018, Ouisse et al., 2020, Green et al., 2021, Krause-Jensen et al., 2021, Rinde et al.,uuuu 2021). To improve the assessment of Zostera bed condition, it could be relevant to carry out more scientific studies assessing ecosystem functions such as habitat provision, stimulation of biodiversity, carbon sequestration, etc. Further details on condition are given in the table above.
Threats and Impacts
Most threats to Zostera beds are directly linked to human activities (Elliott et al., 2017). Examples of these include physical disturbance/habitat damage, marine pollution/hazardous substances, removal of target/non-target species within fisheries and increased turbidity/siltation rate changes. Further threats linked to human activities, which are even harder to manage, are large-scale nutrient/organic enrichment, non-indigenous species, microbial pathogens and climate change (D’Avack et al., 2015, Jones & Unsworth, 2016, Eriander et al., 2017, Wilkes et al., 2017, Branco et al., 2018, Frigstad et al., 2018, Jones et al., 2018, Gardiner, 2020, Román et al., 2020, Bertelli et al., 2021, Garmendia et al., 2021, Krause-Jensen et al., 2021).
Most of the reported threats have been increasing in most Regions/countries and are expected to continue to increase (see also the table on threats and impacts above). The effect of several stressors acting simultaneously on Zostera beds is quite poorly studied, but information is building (see e.g., Moreno-Marin et al., 2018, Vieira et al., 2020, Boyé et al., 2021, Krause-Jensen et al., 2021). Threats such as increased turbidity of the water/reduced light penetration and sediment resuspension can be exacerbated by climate change in the future, and climate change may also increase the need to further reduce other stressors to facilitate expansion of Zostera beds to deeper, cooler waters (Krause-Jensen et al., 2021). Another likely effect of climate change may be an expansion of the northern distribution limit (leading edge of distribution – e.g., Greenland, see e.g., Krause-Jensen et al., 2020) and contraction of the trailing edge of distribution (in Portugal/Spain).
When dealing with threats, protection and avoidance of damage to remaining Zostera beds is cheaper and possibly more efficient than attempting habitat restoration. Habitat restoration can be challenging, although it can be needed in some areas. For further details, see the table on threat and impact above.
Measures that address key pressures from human activities or conserve the species/habitat
Below numbered 3.1 a–k are some actions from OSPAR Recommendation 2012/04 on the protection of Zostera beds, and information from OSPAR implementation reporting in 2019, that to some extent have been undertaken in many Contracting Parties to OSPAR:
3.1 a. Consider the introduction of national legislation to protect Zostera beds
National legislation to protect Zostera beds has been implemented in Germany, the Netherlands, Spain, Sweden and in the United Kingdom, while Norway reports partial implementation (stronger for Zostera noltii). The Zostera beds are mainly protected by implementation of EU directives in Denmark, France and Ireland.3.1 b. Assess whether existing management measures for the protection of Zostera beds are effective and determine what further measures are needed to address the key threats;
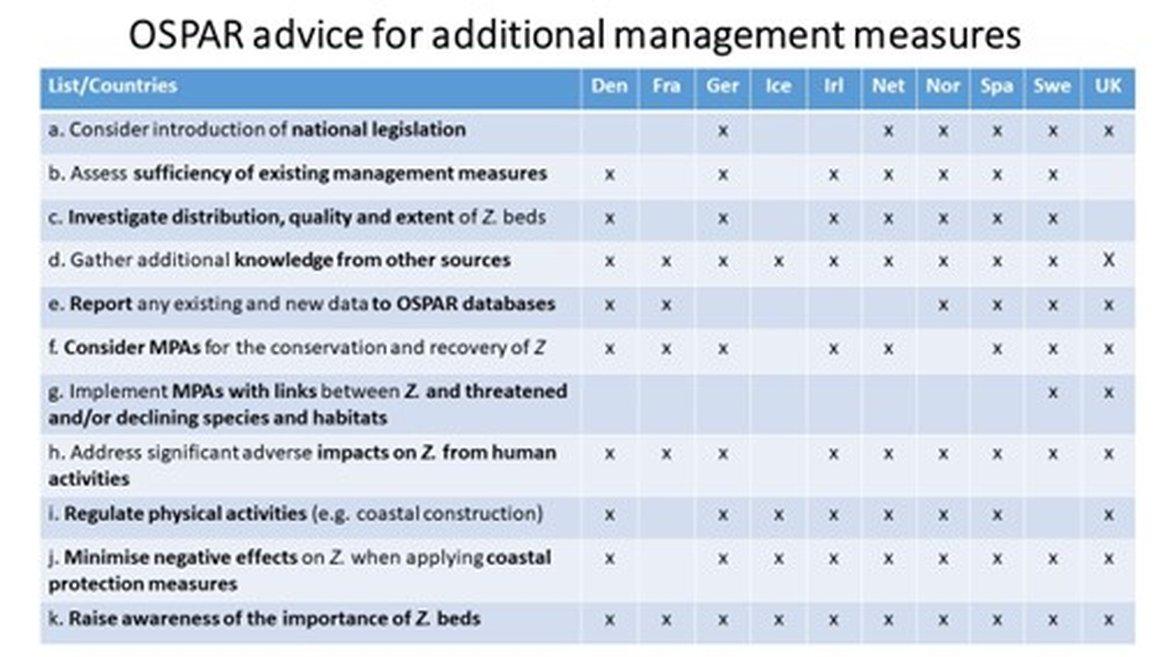
Table 1: Summarised OSPAR implementation reporting in 2019 on the Recommendation 2012/04 for Zostera beds
Indirect assessments of management efficiency have been made. Denmark and Sweden have investigated how previous measures have impacted conservation by evaluating (i) the extent that MPAs had been designated for Zostera beds and (ii) through an analysis of legal protection. Further measures include local restoration projects in Denmark, Norway and Sweden (compilation in Krause-Jensen et al. submitted); special protection, monitoring and reporting in Germany; enhanced conservation status in Ireland; initiation of an agenda for management, measures, research and monitoring of seagrass in the Netherlands; regulations and instructions under the planning and building act in Norway and regional conservation plans in Spain.
3.1 c. Investigate systematically the distribution, quality and extent of Zostera beds, by means of seabed habitat surveys and monitoring, in order to complete the knowledge base and provide indicators for the state and recovery of the habitat;
As reported, systematic monitoring is implemented in Denmark, France, Germany, Iceland, Ireland, the Netherlands, Norway, Spain, Sweden and in the UK, although the indicator development seems to be lagging behind. There are, however, several indicators in place. In Denmark, eelgrass depth limits are used as a key indicator, while in Germany, there are also indicators reflecting the status of eelgrass. Further mapping of Zostera beds is needed, e.g., based on analysis of satellite images that could be repeated at regular intervals.
3.1 d. Whenever applicable seek ways and means to broaden the knowledge base on the occurrence of Zostera beds by gathering additional knowledge from sources such as commercial and recreational fishers, fisheries research and the general public;
As reported, efforts within this area have been taken in Denmark, Germany, Ireland, the Netherlands, Norway (only in the Oslofjord), Spain, Sweden and in the United Kingdom.
3.1 e. Report any existing and new data on the distribution, quality and extent of Zostera beds habitat to the OSPAR habitat mapping database;
This reporting to OSPAR is regularly implemented by Denmark, France, Norway, Spain, Sweden and the United Kingdom.
3.1 f. Consider whether any site within its jurisdiction justifies selection as a marine protected area for the conservation and recovery of Zostera beds;
This has been taken care of, at least to various extents, for some Zostera bed areas in Denmark, France, Germany, Ireland, the Netherlands, Spain, Sweden and in the United Kingdom.
3.1 g. Implement paragraph f with regards to the particular link established between Zostera beds and any species noted in the OSPAR List of Threatened and/or Declining Species and Habitats;
Sweden: Atlantic cod and European eel are species associated with Zostera beds and this is often highlighted in management plans. United Kingdom: Long and short-snouted seahorse have been designated for protection within three sites with seagrass.
3.1 h. Address any significant adverse impacts on Zostera beds arising from human activities in waters under its jurisdiction;
This is addressed through EU directives such as the EU Water Framework Directive and the EU Habitats Directive as well as the Marine Strategy Framework Directive and national acts and legislation at various levels in Denmark, France, Germany, Ireland, the Netherlands, Norway, Spain, Sweden and in the United Kingdom.
3.1 i. Regulate land reclamation, coastal constructions, including marinas and ports, and downscaling of water exchange between open sea and inshore shallow waters, e.g. lagoons;
As being reported from the different countries, this is regulated through the EU Environmental Impact Assessment Directive as well as the EU Water Framework Directive and the EU Habitats Directive, through strict licensing requirements or through implementation of regulations of National Park Laws or national laws in Denmark, Iceland, Ireland, Germany, the Netherlands, Norway, Spain and in the United Kingdom.
3.1 j. Adapt coastal protection measures in such a way that undesired negative effects on Zostera beds are minimised;
Coastal protection measures are applied in such a way that negative effects on Zostera beds are minimized in Denmark, Germany, Iceland, Ireland, the Netherlands, Norway, Spain, Sweden and in the United Kingdom.
3.1 k. Raise awareness of the importance of Zostera beds among relevant management authorities, the fishery sector and the general public. A multitude of different campaigns for various forms of awareness rising of the importance of Zostera beds have been done in Denmark, France, Germany, Iceland, Ireland, the Netherlands, Norway, Spain, Sweden and in the United Kingdom.
Seagrass monitoring programs could benefit from the inclusion of observations of habitat quality in addition to seagrass indicators of distribution and extent. Useful biotic indicators are presence as amounts of filamentous algae, abundance of epiphytes and key fauna. Useful environmental indicators are water quality and clarity, climatic variables (e.g., water temperature and storm frequency and intensity) and sedimentation (Borum et al., 2004, OSPAR, 2009, Marbá et al., 2013, Duffy et al., 2019, Staehr et al., 2019).
Management considerations for seagrass beds include minimising damage, avoidance of causing damage, establishment of protected areas, controlling inputs of pollutants and possible reintroduction or restoration. Promoting awareness of the importance of seagrass beds could assist in minimising certain pressures such as trampling and anchor damage. Protected areas could be designated under the OSPAR MPA-program, under the EU Habitats Directive by inclusion in the Natura 2000 network, or under national schemes such as the Marine Conservation Zones in England, Wales and Northern Ireland and Nature Conservation Marine Protected Areas in Scotland. Additionally, many other management considerations can be taken nationally and locally or settled as agreements within international co-operations such as between Denmark, Germany and the Netherlands for the Wadden Sea, which is a UNESCO World heritage site.
The assessment also brings up possible additional needs for new management measures. These include studies of changes in Zostera beds due to climate change and the possible increase in turbidity of the water/reduced light penetration and resuspension (e.g., due to increased storm frequencies and intensities). Also, changes in coastal development due to increased needs for coastal protection leading to increased risks for habitat loss and increased risks of coastal squeeze, i.e., the coastal zone in “built up” areas becoming narrower due to a rising sea level, may also need to be taken into consideration ensuring that the management becomes “climate ready”. The effect of eelgrass beds on coastal protection, as well as other functional aspects (biodiversity) of eelgrass beds also deserve more attention
Conclusion (including management considerations)
The status of Zostera beds with regard to distribution, extent and condition is poor throughout the OSPAR Maritime Area. In Norway and Sweden, and in some parts around the British Isles (Region II), there are evidence of clear decreases. In other areas, the information of trends is either uncertain or the situation is stable in recent years after years of decline, with a few areas demonstrating improved status. Examples of threats are habitat damage, hazardous substances, non-indigenous species, removal of target/non-target species within fisheries, increased turbidity/siltation rate changes, large-scale nutrient/organic enrichment, microbial pathogens and climate change. Some of these threats/pressures have increased in many Regions/countries during the past ten years and some are also expected to continue to increase over the coming years, although there are also decreasing trends for some pressures/threats possibly related to an improved management focus during the past 20 years (de los Santos et al., 2019). The confidence level for the assessments of the distribution, extent and condition of the Zostera beds and the threats is generally at a medium to a high level.
Management considerations for seagrass beds include, amongst others: minimizing damage or avoidance of causing damage, establishment of protected areas, controlling inputs of pollutants, increasing awareness of the importance of Zostera beds through information and possible reintroduction or restoration. Protecting and avoiding damage to remaining Zostera beds is, however, cheaper and possibly more efficient than attempting expensive restoration measures with high risks of failures (van Katwijk et al. 2015, Moksnes et al., 2018). Re-assessment of the status of and threats to Zostera beds should preferably be done during six year cycles.
Knowledge Gaps
The status of Zostera beds over the entire OSPAR Maritime Area and within different Regions/countries is very mixed/complex and this makes it difficult to reach conclusions for broad Regions. There are significant data and knowledge gaps in some Regions/countries.
Although there are many studies on Zostera beds and Zostera distribution and extent are widely mapped (see references above), there is still a poor understanding of the wider picture at a regional scale particularly in relation to trends over time and the condition of the habitat. National monitoring data are lacking from several Regions and monitoring efforts appear to be patchy making it difficult to draw regional conclusions (Jones & Unsworth, 2016, de los Santos et al., 2019). There are significant data and knowledge gaps in Region I and clear gaps in the amount of reported information in Region IV. There is also a general lack of baseline data and eelgrass maps and a lack of knowledge of how different pressures may interact to disturb and damage the habitat. In addition to the human-induced threats mentioned, the extent of seagrass beds may also change due to natural factors such as severe storms, exposure to air, freshwater pulses, grazing by birds and increased seawater temperatures. Changes following human-induced threats in comparison to changes related to natural variability can also be hard to determine.
A lot of reported information is lacking for many Contracting Parties. There is some information available on records of the distribution and extent of Zostera beds, but the condition and the threat status are seldom known. Historical baseline data on distribution are also missing from most countries and may not exist. In order to improve the status assessment of Zostera beds in the OSPAR Maritime Area, each Contracting Party should provide: 1. A list of regions/locations where Zostera beds occur (ideally, Zostera maps should be made), the condition of these beds and an indication of where the beds are under threat and/or in decline. 2. A description of recent trends (i.e., recent decades and last ten years) and likely changes in extent over the next ten years. 3. A description of threats and impacts. 4. A description of any management measures to protect the Zostera beds and any monitoring programs. However, a great deal of this information has already been assembled and delivered by many OSPAR Contracting Parties.
Method used
Main source of information: 3 b c
- OSPAR data assessment only
- Assessment derived from third party assessment
- Assessment derived from a mix of OSPAR data assessment and assessments from third parties
Assessment is based upon:
a) complete survey or a statistically robust estimate (e.g. a dedicated mapping or survey or a robust predictive model with representative sample of occurrence data, calibration and satisfactory evaluation of its predictive performance using good data on environmental conditions across entire species range);
b) extrapolation from a limited amount of data (e.g., other predictive models or extrapolation using less complete sample of occurrence and environmental data);
c) expert opinion with very limited data;
d) insufficient or no data available.
The assessment is based on a compilation of national responses to a data call (lead: Sweden, Patrik Kraufvelin) and includes regional assessment results from a consensus reaching discussion (overall assessment in the summary table) at an OSPAR workshop on Zostera held on October 13th 2021 as well as contributions before, during and after the POSH-meeting December 6th 2021. The following contracting parties provided national information and data used in the assessment: Denmark, France, Germany, Ireland, Norway, Sweden and UK. Many experts have given valuable input to the assessment: Clara Alvarez Alonso, Isabelle Auby, Lena Avellan, Aurelien Boyé, Florian Carius, Siri Elmegaard, Jacques Grall, Christina Halling, Marc Herlyn, Eduardo Infantes Oanes, Mikael Hjorth Jensen, Kari Holden, Martin Søndergaard Jørgensen, Jörn Kohlus, Kerstin Kolbe, Dorte Krause-Jensen*, Anna Karlsson, Flora Kent, Marie-Louise Krawack, Marie La Riviere, Natasha Lough, Susan McCambridge, Michael Mcleod, Clare McMorrow, Barbara Middleton, Chris Moulton, Miriam Mueller, Oliver Ó Cadhla, Anders G Olsson, Antoine Pebayle, Cathrine Bøgh Pedersen, Eirik Drabløs Pettersen, Elisabeth Rosendal, Gregor Scheiffart, Steen Schwærter, Sebastian Storey, Sebastian Sundberg, Thomas Bruun Valdemarsen, Sofie Voerman, Peter Webster, Thorsten Werner, Benedikte Wiggering, Kirsty Woodcock and Claire Young.
*The Danish reporting is to a large extent based on additional revisions and information provided by Dorte Krause-Jensen after the POSH-meeting.
Airoldi, L., Beck, M.W., 2007. Loss, status and trends for coastal marine habitats of Europe. Oceanography and Marine Biology: An Annual Review, 45: 357–405.
Anon, 2000. UK Biodiversity Group Tranche 2 Action Plans. Volume V – maritime species and habitats. English Nature, Northminster House, UK, 242 pp.
Aubert, F., Sauriau, P.-G., Guenneteau, S., 2021a. Contrôle de surveillance DCE 2020 de la masse d’eau côtière «FRFC02 - Pertuis charentais» et de la masse d’eau de transition «FRFT09 - Estuaire de la Gironde» pour les herbiers de Zostera (Zosterella) noltei Hornemann: Rapport final. Rapport CNRS Cohabys- Ifremer - Agence de l'Eau Adour Garonne , La Rochelle, 66 pp. (In French).
Aubert, F., Sauriau, P.-G., Pineau, P., Gervot, N., 2021b. Contrôle de surveillance DCE 2020 des masses d’eau côtière «FRGC53 - Pertuis Breton» et «FRGC48 - Baie de Bourgneuf» et de la masse d’eau de transition «FRGT30 - Estuaire du Lay» pour les herbiers de Zostera noltei Hornemann : rapport final. Rapport CNRS Cohabys- Ifremer - Agence de l'Eau Adour Garonne , La Rochelle, 70 pp. (In French).
Aubin S., Monnier B., Husset M.C., 2021. Suivi des herbiers à Zostera marina du secteur Ouest-Cotentin dans le cadre du contrôle de surveillance de la Directive Cadre Européenne sur l’Eau (2000/60/CE). Rapport d'activité des campagnes de prélèvements 2020. Rapport MNHN, 24 pp. (In French).
Auby, I., Oger-Jeanneret, H., Trut, G., Ganthy, F., Rigoiun, L., de Casamajor, M.-N., Sanchez, F., Lissardy, M., Fortune, M., Manach, S., Bizzozero, L., Rollet, C., Lejolivet, A., Desroy, N., Foveau, A., Le Mao, P., Sauriau, P.G., Aubert, A., Cajeri, P., Curti, C.,Duvard, A., Latry, L., Lachaussee, N., Pineau, P., Plumejeaud-Perreau, C., Aubin, S., Droual, G., Fournier, J., Garcia, A., Guillaudeau, J., Hubert, C., Humbert, S., Janson, A.L., Mase, C., Panizza, A.C., Grall, J., Maguer, M., Hily, C., Hacquebart, P., Joncourt, Y., Baffreau, A., Timsit, P., 2018. Classement des masses d’eau du littoral Manche-Atlantique sur la base de l’indicateur DCE “Angiospermes” (2012-2016). ODE/UL/LER/AR18.002. (In French).
Auby, I., Lissardy, M., d’Amico, F., Aubert, F., 2020a. Suivi stationnel (2007-2019) de l'herbier de zostères naines (Zostera noltei) et calcul de l’indicateur «Angiospermes» (2019) dans la masse d'eau côtière FRFC09 – Lac d'Hossegor. RST/LER/AR/19.012. (In French).
Auby, I., Rigouin, L., Ganthy, F., Trut, G., Oger-Jeanneret, H., Gouriou, L., Bujan, S., Devaux, L., Aubert, F., Dalloyau, S., Péré, C., 2020b. Suivi stationnel (2006-2019) des herbiers de zostères (Zostera noltei et Zostera marina) et calcul de l’indicateur «Angiospermes» (2019) dans la masse d'eau côtière FRFC06 – Arcachon amont - Bassin Hydrographique Adour-Garonne. ODE/LER/AR/20.014. (In French).
d'Avack, E.A.S., Tyler-Walters, H., Wilding, C., 2015. Zostera marina/angustifolia beds on lower shore or infralittoral clean or muddy sand. In: Tyler-Walters, H., Hiscock, K. (Eds.), Marine Life Information Network: Biology and Sensitivity Key Information Reviews, [on-line]. Plymouth: Marine Biological Association of the United Kingdom. https://mhc.jncc.gov.uk/biotopes/jnccmncr00000234.
Baden, S., Gullström, M., Lundén, B., Pihl, L., Rosenberg, R., 2003. Vanishing seagrass (Zostera marina, L.) in Swedish coastal waters. Ambio 32: 374–377.
Bertelli, C.M., Bull, J.C., Cullen-Unsworth, L.C., Unsworth R.K.F., 2021. Unravelling the spatial and temporal plasticity of eelgrass meadows. Frontiers in Plant Science 12: 939.
Bockelmann, A.C., Tams, V., Ploog, J., Schubert, P.R., Reusch, T.B., 2013. Quantitative PCR reveals strong spatial and temporal variation of the wasting disease pathogen, Labyrinthula zosterae in northern European eelgrass (Zostera marina) beds. PLoS One 8: e62169.
Borum, J., Duarte, C.M., Krause-Jensen, D., Greve, T.M. (Eds.), 2004. European Seagrasses: An introduction to monitoring and management. The M & MS project. ISBN: 87-89143-21-3. Internet version: Available at http://www.seagrasses.org
Boström, C., Baden, S.P., Krause-Jensen, D., 2003. The seagrasses of Scandinavia and the Baltic Sea. In: Green, E.P., Short, F.T., Spalding, M.D. (Eds.) UNEP. World Atlas of Seagrasses: present status and future conservation. Chapter 1. pp. 35–45.
Boström, C., Baden, S., Bockelmann, A.-C., Dromph, K., Fredriksen, S., Gustafsson, C., Krause-Jensen, D., Möller, T., Laurentius, S., Nielsen, B., Olesen, J., Pihl, L., Rinde, E., 2014. Distribution, structure and function of Nordic eelgrass (Zostera marina) ecosystems: implications for coastal management and conservation. Aquatic Conservation: Marine and Freshwater Ecosystems 24: 410–434.
Boyé, A., Gauthier, O., Becheler, R., Le Garrec, V., Hily, C., Maguer, M., Grall, J., 2021. Drivers and limits of phenotypic responses in vulnerable seagrass populations: Zostera marina in the intertidal. Journal of Ecology 110: 144–161.
Branco, J., Pedro, S., Alves, A.S., Ribeiro, C., Meteratski, P., Pires, R., Caçador, I., Adão, H., 2018. Natural recovery of Zostera noltii seagrass beds and benthic nematode assemblage responses to physical disturbance caused by traditional harvesting activities. Journal of Experimental Marine Biology and Ecology 502: 191–202.
Burton, M., Lock, K., Newman, P., Jones, J., 2018. Skomer Marine Conservation Zone Project Status Report 2018. NRW Evidence Report 324, 138 pp.
Cognat, M., Ganthy, F., Auby, I., Barraquand, F., Rigouin, L., Sottolichio, A., 2018. Environmental factors controlling biomass development of seagrass meadows of Zostera noltei after a drastic decline (Arcachon Bay, France). Journal of Sea Research 140: 87–104.
Davison, D.M., Hughes, D.J., 1998. Zostera species: An overview of dynamic and sensitivity characteristics for conservation management of marine SACs. Scottish Association of Marine Sciences (UK Marine SACs Project), Oban.
Den Hartog, C., Polderman, P.J.G., 1975. Changes in the seagrass populations of the Dutch Waddenzee. Aquatic Botany 1: 15–26.
de los Santos, C.B., Krause-Jensen, D., Alcovarro, T, Marbá, N., Duarte, C.M., van Katwijk, M.M., Pérez, M., Romera, J., Sánchez-Lizaso, J.L., Roca, G., Jankowska, E., Pérez-Lloréns, J.L., Fournier, J., Montefalcone, M., Pergent, G., Ruiz, J.M., Cabaço, S., Cook, K., Wilkes, R.J., Moy, F.E., Trayter, G.M.-R., Araño, X.S., de Jong, D., Fernández-Torquemada, Y., Auby, I., Vergara, J.J., Santos, R., 2019. Recent trend reversal for declining European seagrass meadows. Nature Communications 10: 3356.
Duarte, C.M., 1991. Seagrass depth limits. Aquatic Botany 40: 363–377.
Duffy, J.E., Benedetti-Cecchi, L., Trinanes, J.A., Muller-Karger, F.E., Ambo-Rappe, R., Boström, C., Buschmann, A.H., Byrnes, J., Coles, R.G., Creed, J., Cullen-Unsworth, L., Diaz-Pulido, G., Duarte, C.M., Edgar, G.J., Fortes, M.D., Goni, G.J., Hu, C., Huang, X., Hurd, C.L., Konar, B., Krause-Jensen, D., Krumhansl, K., Macreadie, P.I., Marsh, H., McKenzie, L.J., Mieszkowska, N., Miloslavich, P., Montes, E., Nakaoka, M., Norderhaug, K.M., Nordlund, L.M., Orth, R.J., Prathep, A., Putman, N.F., Samper-Villarreal, J., Serrao, E.A., Short, F., Pinto, I.S., Steinberg, P.D., Stuart-Smith, R., Unsworth, R.K.F., van Keulen, M., van Tussenbroek, B.I., Wang, M., Waycott, M., Weatherdon, L.V., Wernberg, T., Yaakub, S.M., 2019. Toward a coordinated global observing system for seagrasses and marine macroalgae. Frontiers in Marine Science 6: 317.
Elliott, M., Burdon, D., Atkins, J.P., Borja, A., Cormier, R., De Jonge, V.N., Turner, R.K., 2017. “And DPSIR begat DAPSI(W)R(M)!” – A unifying framework for marine environmental management. Marine Pollution Bulletin 118: 27–40.
Eriander, L., Laas, K., Bergström, P., Gipperth, L., Moksnes, P.O.. 2017. The effects of small-scale coastal development on the eelgrass (Zostera marina L.) distribution along the Swedish west coast–Ecological impact and legal challenges. Ocean and Coastal Management 148: 182–194.
Frigstad, H., Andersen, G.S., Trannum, H.C., Naustvoll, L.J., Kaste, Ø., Hjermann, D.Ø. 2018. Synthesis of climate relevant results from selected monitoring programs in the coastal zone. Part 2: Quantitative analyses. Miljødirektoratet rapport M-1220|2018. 54 pp.
Frigstad, H., Gundersen, H., Andersen, G.S., Borgersen, G., Kvile, K.Ø., Krause-Jensen, D., Boström, C., Bekkby, T., d’Auriac, M., Ruus, A., Thormar, J., Asdal, K., Hancke, K., 2021. Blue Carbon–climate adaptation, CO2 uptake and sequestration of carbon in Nordic blue forests: Results from the Nordic Blue Carbon Project. Nordic Council of Ministers. Tema Nord 2020:541.
García-Redondo, V., Bárbara, I., Díaz-Tapia, P., 2019. Zostera marina meadows in the northwestern Spain: distribution, characteristics and anthropogenic pressures. Biodiversity and Conservation 28: 1743–1757.
Gardiner, T., 2020. The conservation status of dwarf eelgrass (Zostera noltei). In book: Reference Module in Earth Systems and Environmental Sciences. DOI: 10.1016/B978-0-12-821139-7.00060-X
Garmendia, J.M., Valle, M., Borja, A., Chust, G., Rodrígues, J.G., Franco, J., 2021. Estimated footprint of shellfishing activities in Zostera noltei meadows in a northern Spain estuary: Lessons for management. Estuarine, Coastal and Shelf Science 254: 107320.
Godet, L., Fournier, J., van Katwijk, M. M., Olivier, F., Le Mao, P., Retière, C., 2008. Before and after wasting disease in common eelgrass Zostera marina along the French Atlantic coasts: a general overview and first accurate mapping. Diseases of Aquatic Organisms 79: 249–255.
Grall J., Maguer M., Tauran A., Lescop M., Le Garrec V., 2021. Contrat UBO – Ifremer 2020. Rapport final - Année 2020, 36 pp. (In French).
Green, A.E., Unsworth, R.K., Chadwick, M.A., Jones, P.J., 2021. Historical analysis exposes catastrophic seagrass loss for the United Kingdom. Frontiers in Plant Science 12: 261.
Guerrero-Meseguer, L., Veiga, P., Sampaio, L., Rubal, M., 2021. Resurgence of Zostera marina in the Ria de Aveiro lagoon, Portugal. Aquatic Botany 169: 103338.
Hansen J.W., Høgslund S. (Eds.) 2021. Marine områder 2019. NOVANA. Aarhus Universitet, DCE – Nationalt Center for Miljø og Energi, 174 pp. - Videnskabelig rapport fra DCE nr. 418. (In Danish).
Jones, B.L., Unsworth, R.K.F., 2016. The perilous state of seagrass in the British Isles. Royal Society Open Science 3: 150596.
Jones, B.L., Cullen-Unsworth L.C., Unsworth R.K.F., 2018. Tracking nitrogen source using δ15N reveals human and agricultural drivers of seagrass degradation across the British Isles. Frontiers in Plant Science 9: 133.
Jones, L.A., Hiscock, K., Connor, D.W., 2000. Marine habitat reviews. A summary of ecological requirements and sensitivity characteristics for the conservation and management of marine SACs. Joint Nature Conservation Committee, Peterborough. (UK Marine SACs Project report).
van Katwijk, M.M., Thorhaug, A., Marbá, N., Orth, R.J., Duarte, C.M., Kendrick, G.A., Althuizen, I.H.J., Balestri, E., Bernard, G., Cambridge M.L., Cunha, A., Durance, C., Giesen, W., Han, Q., Hosokawa, S., Kiswara, W, Komatsu, T., Lardicci, C., Lee, K.-S., Meinesz, A., Nakaoka, M., O´Brien, K.R., Paling, E.I., Pickerell, C., Ransijn, A.M.A., Verduin, J.J., 2015. Global analysis of seagrass restoration: the importance of large-scale planting. Journal of Applied Ecology 53: 567–578.
Kilminster, K., McMahon, K., Waycott, M., Kendrick, G.A., Scanes, P., McKenzie, L., O’Brien, K.R., Lyons, M., Ferguson, A., Maxwell, P., Glasby, T., Udy, J., 2015. Unravelling complexity in seagrass systems for management: Australia as a microcosm. Science of the Total Environment 534: 97–109.
Krause-Jensen, D., Archambault, P., Assis, J., Bartsch, I., Bischof, K., Filbee-Dexter, K., Dunton, K.H., Maximova, O., Ragnarsdóttir, S.B., Sejs, M.K., Simakova, U., Spiridonov, V., Wegeberg, S., Winding, M.H.S., Duarte, C.M., 2020. Imprint of climate change on pan-Arctic marine vegetation. Frontiers in Marine Science 7: 1129.
Krause-Jensen, D., Duarte, C.M., Sand-Jensen, K., Carstensen, J., 2021. Century-long records reveal shifting challenges to seagrass recovery. Global Change Biology 27: 563–575.
Marbà, N., Krause-Jensen, D., Alcoverro, T., Birk, S., Pedersen, A., Neto, J.M., Orfanidis, S., Garmendia, J.M., Muxica, I., Borja, A., Dencheva, K., Duarte, C.M., 2013. Diversity of European seagrass indicators: patterns within and across regions. Hydrobiologia 704: 265–278.
Marbà, N., Krause-Jensen, D., Masqué, P., Duarte, C.M., 2018. Expanding Greenland seagrass meadows contribute new sediment carbon sinks. Scientific Reports 8: 1–8.
Maxwell, P.S., Eklöf, J.S., van Katwijk, M.M., O'Brien, K.R., de la Torre‐Castro, M., Boström, C., Bouma, T.J., Krause-Jensen, D., Unsworth, R.K.F., van Tussenbroek, B.I., van der Heide, T., 2017. The fundamental role of ecological feedback mechanisms for the adaptive management of seagrass ecosystems – a review. Biological Reviews 92: 1521–1538.
Moffat, C., Baxter, J., Berx, B., Bosley, K., Boulcott, P., Cox, M., Cruickshank, L., Gillham, K., Haynes, V., Roberts, A., Vaughan, D., Webster, L. (Eds.), 2021. Scotland's Marine Assessment 2020: Headlines and next steps. Scottish Government.
Moksnes, P.O., Eriander, L., Infantes, E., Holmer, M., 2018. Local regime shifts prevent natural recovery and restoration of lost eelgrass beds along the Swedish west coast. Estuaries and Coasts 41: 1712–1731.
Moksnes, P.-O., Röhr, M.E., Holmer, M., Eklöf, J.S., Eriander, L., Infantes, E., Boström, C., 2021. Major impacts and societal costs of seagrass loss on sediment carbon and nitrogen stocks. Ecosphere 12: e03658.
Moreno‐Marín, F., Brun, F.G., Pedersen, M.F., 2018. Additive response to multiple environmental stressors in the seagrass Zostera marina L. Limnology and Oceanography 63: 1528–1544.
Naustvoll, L.J., Thormar, J., Lundsør, E., Falkenhaug, T., Kroglund, T., Moy, F., 2021. ØKOKYST – delprogram Klima. Årsrapport 2020. Miljødirektoratet rapport M-1973|2021, 61 s. (In Norwegian).
NPWS, 2019. The Status of EU Protected Habitats and Species in Ireland. Volume 2: Habitat Assessments. Unpublished NPWS report. Edited by: Deirdre Lynn and Fionnuala O’Neil. 1128 pp.
O’Brien, K.R., Waycott, M., Maxwell, P., Kendrick, G.A., Udy, J.W., Ferguson, A.J.P., Kilminster, K., Scanes, P., McKenzie, L.J., McMahon, K., Adams, M.P., Samper-Villarreal, J., Collier, C., Lyons, M., Mumby, P.J., Radke, L., Christianen, M.J.A., Dennison, W.C., 2018. Seagrass ecosystem trajectory depends on the relative timescales of resistance, recovery and disturbance. Marine Pollution Bulletin 134: 166–176.
Olesen, B., Krause-Jensen, D., Marbà, N., Christensen, P.B., 2015. Eelgrass Zostera marina in subarctic Greenland: dense meadows with slow biomass turnover in cold waters. Marine Ecology Progress Series 518: 107–121.
Ollivier E., Lemoine A., Timsit O., Baffreau A., Garcia A., 2021. Acquisition de données biologiques. Suivi de peuplements benthiques dans le cadre de la surveillance DCE-Benthos. Action 20120. Rapport GEMEL Normandie, 47 pp. (In French).
OSPAR, 2008. Case Reports for the OSPAR List of Threatened and/or Declining Species and Habitats. OSPAR Commission, 261 pp.
OSPAR, 2009. Background Document for Zostera beds, Seagrass beds. OSPAR Commission, 39 pp.
Ouisse, V., Marchand-Jouravleff, I., Fiandrino, A., Feunteun, E., Ysnel, F., 2020. Swinging boat moorings: Spatial heterogeneous damage to eelgrass beds in a tidal ecosystem. Estuarine, Coastal and Shelf Science 235:106581.
Rasmussen, E., 1977. The wasting disease of eelgrass (Zostera marina) and its effects on environmental factors and fauna. In: C.P. McRoy & C. Helfferich (Eds.), Seagrass Ecosystems, A Scientific Perspective (pp. 1–51). Marcel Dekker Inc.
Ribaudo, C., Plus, M., Ganthy, F., Auby, I., 2016. Carbon sequestration loss following Zostera noltei decline in the Arcachon Bay (France). Estuarine, Coastal and Shelf Science 179: 4–11.
Rinde, E., Bekkby, T., Kvile, K., Andersen, G.S., Brkljacic, M., Anglès d’Auriac, M., Christie, H., Fagerli, C.W., Fredriksen, S., Moy, S., Staalstrøm, A., Tveiten, L., 2021. Kartlegging av et utvalg marine naturtyper i Oslofjorden. Miljødirektoratet rapport M-2066|2021, 74 pp. (In Norwegian).
Román, M., Fernández, E., Zamborain-Mason, J., Martínez, L., Méndez, G, 2020. Decadal changes in the spatial coverage of Zostera noltei in two seagrass meadows (Ría de Vigo; NW Spain). Regional Studies in Marine Science 36: 101264.
Schutter, M., de Jong, J. W., Van Deelen, J., 2021. Zeegraskartering MWTL Waddenzee en Oosterschelde 2020. Waterlichamen: Waddenzee en Oosterschelde (No. 20–281). Bureau Waardenburg B.V. (In Dutch).
Staehr, P.A., Göke, C., Holbach, A.M., Krause-Jensen, D., Timmermann, K., Upadhyay, S., Ørberg, S.B., 2019. Habitat model of eelgrass in Danish coastal waters: development, validation and management perspectives. Frontiers in Marine Science 6: 175.
Unsworth, R.K.F., Bertelli, C., Robinson, M., Mendzil, A., 2017. Status review and surveillance recommendation for seagrass (Zostera species) in Milford Haven Waterway. Milford Haven Waterway Environmental Survellaince Group. 9 pp.
Van Deelen, J.J., Stolk, A.H., Verduin, E.C., 2019. Zeegraskartering Oosterschelde en Westerschelde. Meetjaar 2019. Rijkswaterstaat Centrale Informatie Voorziening. (In Dutch).
Vieira, R., Martin, A., Engelen, A.H., Thomsen, M.S., Arenas, F., 2020. Interactive effects of co-occurring anthropogenic stressors on the seagrass, Zostera noltei. Ecological Indicators 109: 105780.
Wilkes, R., Bennion, M., McQuaid, N., Beer, C., McCullough-Annett, G., Colhoun, K., Inger, R., Morrison, L., 2017. Intertidal seagrass in Ireland: pressures, WFD status and an assessment of trace element contamination in intertidal habitats using Zostera noltei. Ecological Indicators 82: 117–130.
Zwarts, M., Verduin, E., Heusinkveld, J., 2018. Zeegraskartering MWTL Waddenzee. Meetjaar 2017. Rijkswaterstaat Centrale Informatie Voorziening. (In Dutch).
Sheet reference:
BDC2022/Zostera_beds