Status Assessment 2022 - Atlantic salmon
| Assessment of status | Distribution | Abundance | Condition | Previous OSPAR status assessment | Status | |
|---|---|---|---|---|---|---|
Region | I | ↔3,5 | ↔2,3,5 | ↔3,5 | ● | Poor |
| II | ↔3,5 | ↓2,3,5 | ↔3,5 | ● | Poor | |
| III | ↔3,5 | ↓2,3,5 | ↓3,5 | ● | Poor | |
| IV | ↔3,5 | ↔2,3,5 | ↔3,5 | ● | Poor | |
| V | N/A | |||||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: If in QSR 2010 then enter Regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ) based on Chapter 10 Table 10.1 and Table 10.2 . If a more recent status assessment is available, then enter ‘poor’/’good’
Status*(overall assessment)
red – poor
green – good
? – status unknown
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressure
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threats or impacts (overall assessment)
red – significant threat or impact;
green–no evidence of a significant threat or impact
Blue cells – insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
| Assessment of threats | Barrier to species movement (Building of dams and navigation weirs) | Input of other substances (synthetic/non-synthetic) (Water pollution) | Extraction of, or mortality/injury to, wild species (Hydrological manipulation) | Removal of target species (Fisheries exploitation) | Climate change | Ocean acidification | Genetic modification and translocation of indigenous species, Introduction of microbial pathogens, Input of nutrients, Input of organic matter, Input of genetically modified species / translocation of species, Loss of/change to natural biological communities (Farmed salmonid aquaculture) | Input or spread of non-indigenous species [Non-native salmonid invasion] | Threat | |
|---|---|---|---|---|---|---|---|---|---|---|
Region | I | ↔5 | ↔5 | ↔5 | ↓2,5 | ↔5 | ↔2,5 | ↑5 | ?5 | |
| II | ↔5 | ↔5 | ↔5 | ↓2,5 | ↑3,5 | ↔5 | ↑5 | ?5 | ||
| III | ↔5 | ↔5 | ↔5 | ↔2,5 | ↑3,5 | ↓5 | ↔5 | ?5 | ||
| IV | ↔5 | ↔5 | ↔5 | ↔5 | ↑5 | ↔5 | ↔5 | ↔5 | ||
| V | NA | |||||||||
Confidence
Confidence is high for the overall status assessment as most Contracting Parties complete annual stock assessments based on substantial data collection and analyses, and all experts agreed on the current situation (distribution, abundance and condition) for the Atlantic salmon population.
Confidence is medium to low on threats as many are associated with knowledge gaps (climate change, invasions, small-scale hydropower installations). Also, trends are sometimes difficult to assess within the same OSPAR Regions, where threats might not affect all populations to the same extent.
Background Information
Year added to OSPAR List: 2003 (OSPAR, 2008). The original evaluation of Atlantic salmon against the Texel-Faial criteria referred to global/regional importance, sensitivity and decline criteria, with information also provided on threats.
Summary extracted from the Case Report (OSPAR, 2008):
- Global/regional importance: Atlantic salmon is present in all OSPAR Regions (I to IV).
- Sensitivity: The species juveniles are particularly sensitive to freshwater quality and accessibility (Baglinière et al., 2005).
- Decline: The abundance has dramatically declined since the mid-19th century especially in Germany, Belgium and the Netherlands where it totally disappeared. The situation in the southern part of Europe is also critical.
- Threats: Threats to Atlantic salmon occur in both freshwater and marine environments. Anthropogenic pressures (e.g. agriculture, industry, dams) strongly affect juvenile mortality in fresh water. In the marine environment, the main threats are fisheries (direct and by-catch), interactions with individuals from marine Atlantic salmon farms and climate change.
Last status assessment: 2008. OSPAR (2010) concluded that Atlantic salmon populations are affected by numerous threats both in marine and freshwater ecosystems. Future challenges would be to better understand mortality causes, especially during the marine part of the life cycle.
Geographical Range and Distribution
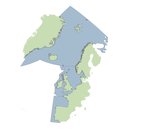
The overall marine distribution of Atlantic salmon populations, originating from European rivers, overlaps to various degrees and extent, in OSPAR regions, from Arctic waters (OSPAR Region I, Norwegian Sea) to Atlantic waters of the Iberian Peninsula (OSPAR Region IV, Figure 1). Overall, the marine distribution of Atlantic salmon has not changed significantly over the past decade for OSPAR Regions I to IV, but several recent studies have suggested that some Atlantic salmon populations, in Norway and Denmark, could have extended their migration further north into Arctic waters (Jensen et al., 2014; Rikardsen et al., 2021).
Regarding freshwater distribution, there is still a lack of data to map the precise distribution within river basins where reproduction of Atlantic salmon occurs. Nonetheless, Norway still holds approximately 25% of extant Atlantic salmon populations in more than 400 watercourses (Forseth et al., 2017). During the past decades, improvements in habitat quality and quantity in European rivers have facilitated the return or the spread of Atlantic salmon populations. This is the case in France in the river Seine (Perrier et al., 2010) and the Adour-Gaves basin (Prouzet & Michelet, 2019). In Scotland, the removal of barriers has helped to increase available juvenile habitat and production (92/43/EEC Scotland report 2019; (Buddendorf et al., 2019; Malcolm et al., 2019)). In Wales, there has also been considerable investments into removing barriers to migration (Salmon for Tomorrow Programme). In Germany, several restocking programs have been active the past decades with various successes and failures notably in the Rhine basin (Monnerjahn, 2011). In 2018 and 2019, restocking actions were implemented in Minho River (Portugal) with fish from Galicia; there are currently no results for their success or failure (Project Migramiño; http://migraminho.org/). Overall, the freshwater distribution of Atlantic salmon has not changed significantly since the previous OSPAR assessment in 2010. In Denmark extensive dam removal and river restoration have made hundreds of kilometres of spawning and rearing habitat available for salmon, increasing spawning population from a few hundred to over 10 000) in the last decades (Koed et al., 2020).
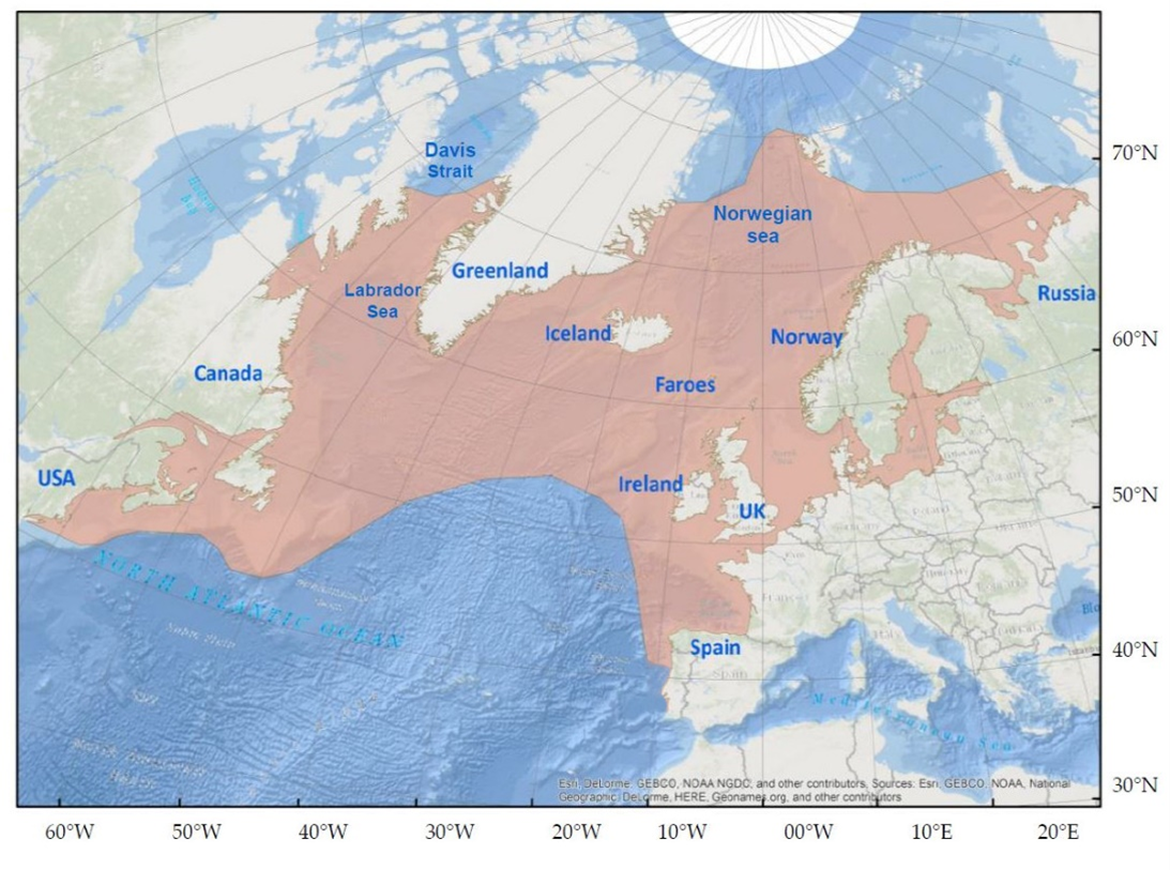
Figure 1: The North Atlantic with the geographic distribution of Atlantic salmon indicated in light red (Maoiléidigh, 2018)
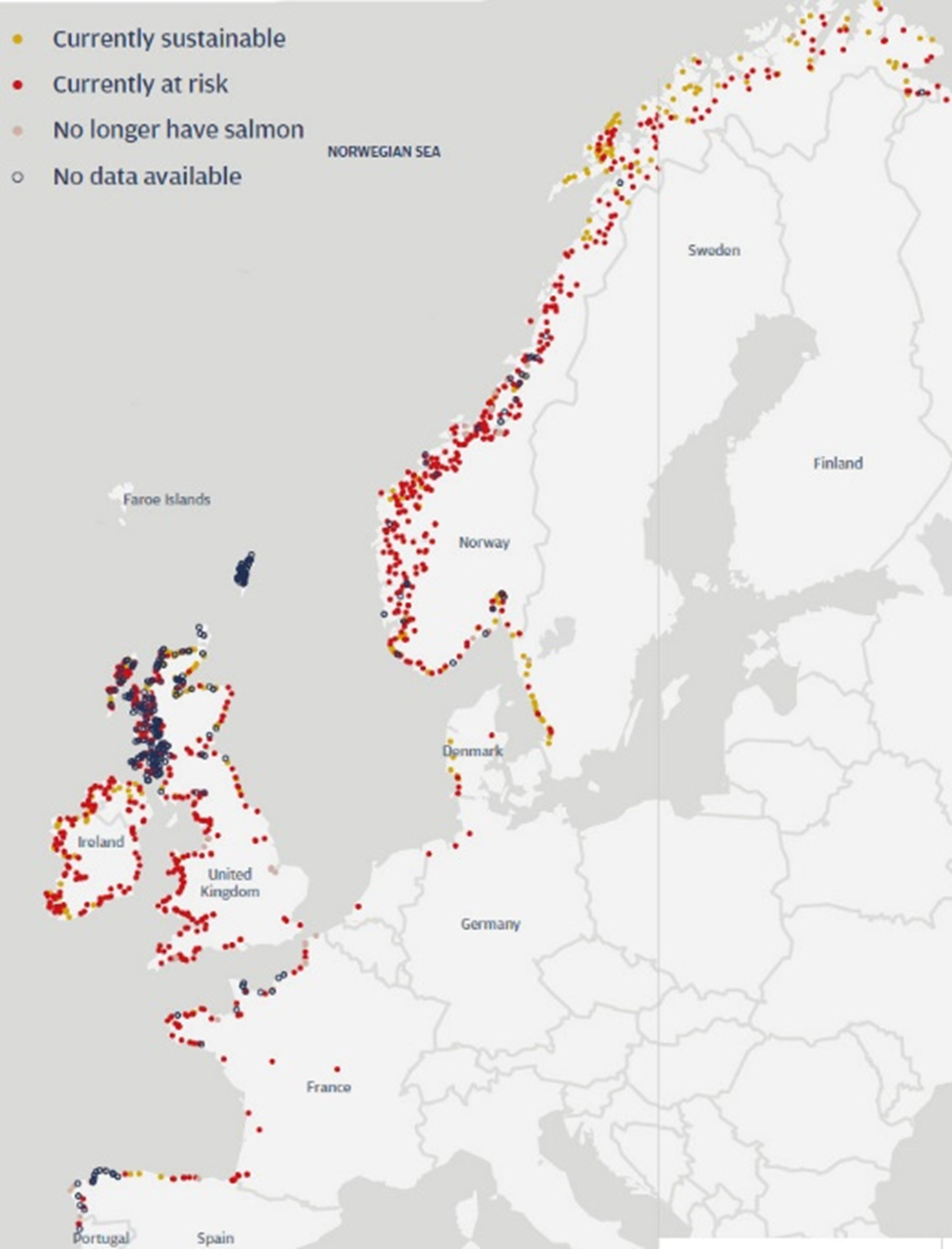
Figure 2: NASCO risk categorization of Atlantic salmon stocks across European rivers, adapted from (NASCO, 2020b). The categories (i.e. colours) used here are those selected by NASCO and do not always translate to stock state categories used by the nations/jurisdictions.
Population/Abundance
For the past decade, the population abundance of Atlantic salmon has declined to an historically low level, notably in OSPAR Region I (Figure 3 and 4; for Norway, see (Forseth et al., 2020)). The situation in other Regions is mixed among stocks and sea ages. The situation in France is estimated to be quite constant. In the UK (OSPAR Regions II and III), the Pre Fishery Abundance of maturing 1SW (i.e., individuals that return as mature fish to spawn only after one-Sea-Winter at sea, also called grilse; Figure 3) and MSW (i.e., individuals which have matured after more than one winter at sea; Figure 4) salmon showed variable trends. In the Iberian peninsula (OSPAR Region IV), populations had declined dramatically over the last decade (Mota et al., 2016; Nicola et al., 2018). Recent recovery programs in Denmark (OSPAR Region II) produced encouraging growth of a restocking population in the last ten years with a majority of returning adult fish being of wild origin (ICES, 2021; Koed et al., 2020; NASCO, 2020b); their effects on wild populations should continue to be documented. Despite these general trends at the national scale, it is important to consider that trends in individual Atlantic salmon stocks may be quite variable within a Region. For example, a study aggregating data from 29 monitored rivers from France between 1980 and 2010, (Legrand et al., 2020) found a slightly significant increase of Atlantic salmon abundance at the national scale but with contrasting trends between several basins (across OSPAR Regions II, III and IV).
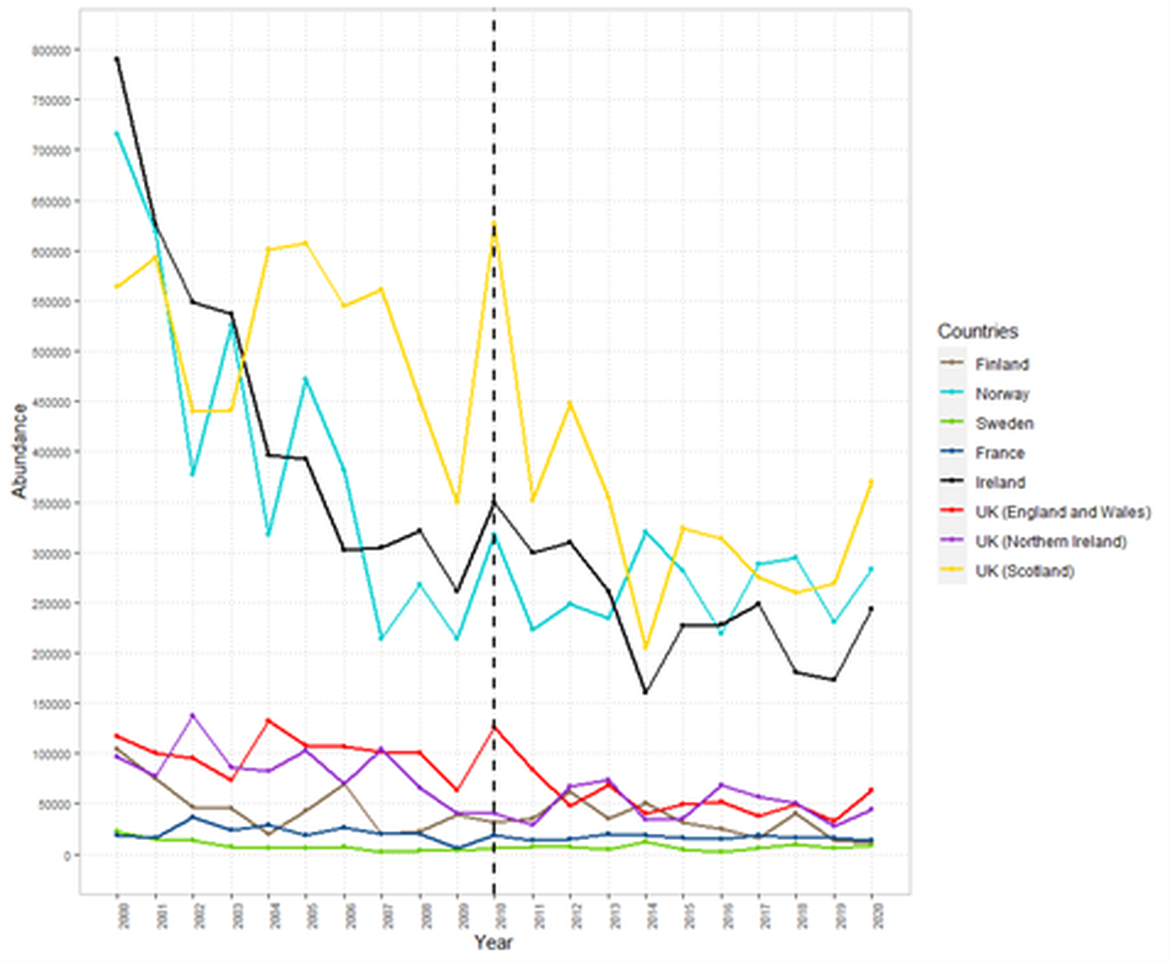
Figure 3: Pre-fishery abundance (PFA) of one-Sea-Winter (1 SW) Atlantic salmon between 2000 and 2020 (ICES, 2021). Black dotted line indicates date of last OSPAR assessment for Atlantic salmon.
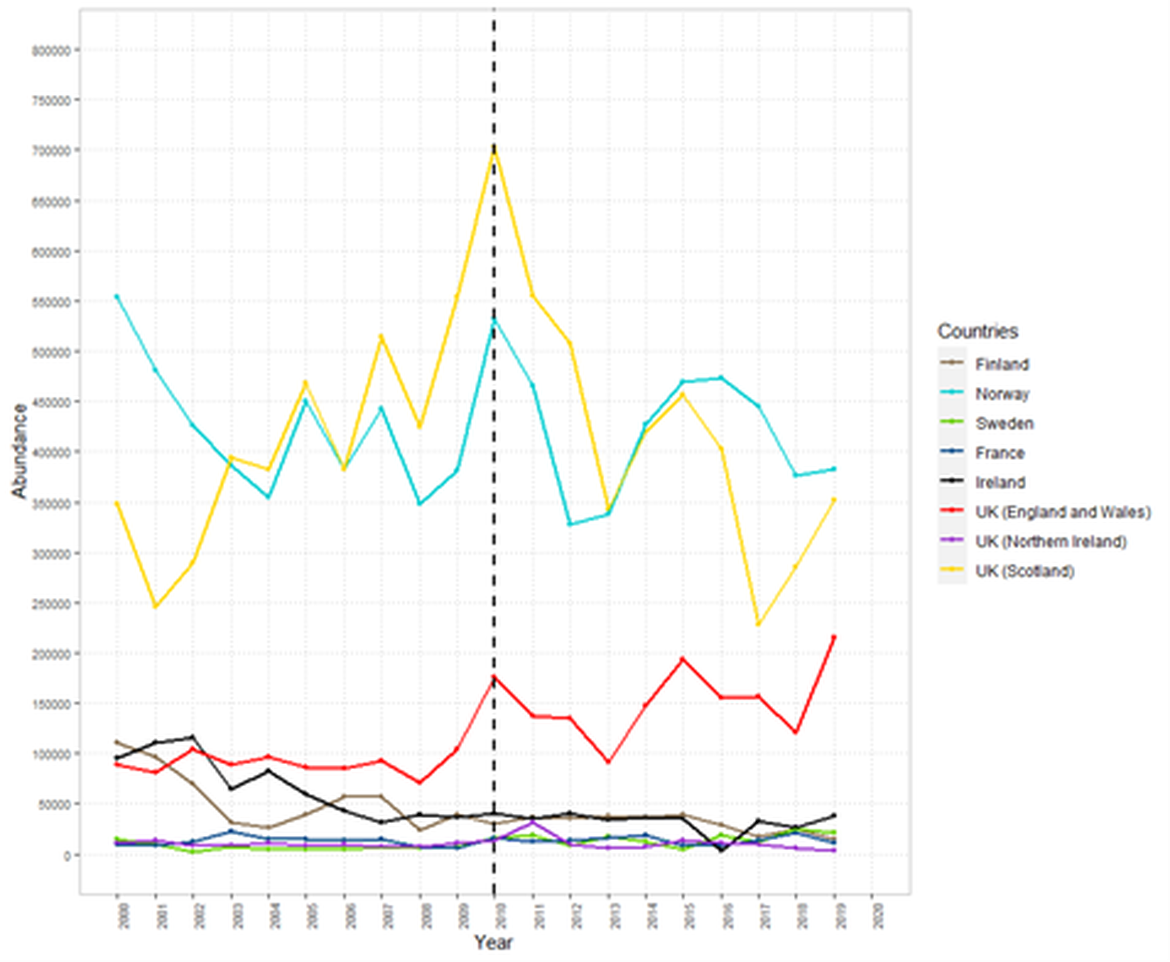
Figure 4: Pre-fishery abundance of Multi-Sea-Winter (MSW) Atlantic salmon between 2000 and 2019 (ICES, 2021). Black dotted line indicates date of last OSPAR assessment for Atlantic salmon.
Condition
Several studies have reported long-term reductions in body size and condition as well as changes in growth metrics (Forseth et al., 2020; Quinn et al., 2006; Saura et al., 2010; Todd et al., 2021), often more strongly affecting 1SW than MSW fish (Bacon et al., 2009). Several studies further demonstrated that low fecundity and reduced reproductive potential of populations are correlated with loss of weight and lower growth rates (de Eyto et al., 2015; Jacobson et al., 2021). While over the long-term the general trend in salmon condition has been downward, particularly for 1SW stocks in Celtic Sea (see for instance the study of Todd et al., 2021), there is some evidence to suggesting the trend is stabilizing in other OSPAR Regions albeit at an historically low level.
Threats and Impacts
Atlantic salmon are exposed to many threats during both freshwater and marine phases of their life cycle.
- Dams and navigation weirs
European rivers are densely fragmented by many barriers and dams, with 60% considered not free from barriers (Grill et al., 2019), and with the highest barrier densities found in Central Europe and the lowest in the most remote sparsely populated upland areas (e.g. Scandinavia and Scotland) (Belletti et al., 2020). This fragmentation greatly affects the lateral connectivity of rivers and both upstream and downstream migration of Atlantic salmon (Havn et al., 2020). While this situation remained relatively constant in most OSPAR regions over the last ten years, recent dam removal programs have reduced the threat in France (e.g. at Selune river since 2012) and UK (e.g. for Scotland, 92/43/EEC Scotland report 2019; and for England and Wales, NASCO (2017, 2020a), Buddendorf et al., 2019; Malcolm et al., 2019). .
- Water pollution (sewage, pesticides/herbicides, heavy metal contamination, hormones, eutrophication)
Poor water quality is a threat for the species. In 2018, a total of 38% of surface water bodies were considered in good chemical status in the EU (source: https://water.europa.eu/freshwater/data-maps-and-tools/water-framework-directive-surface-water-data-products/surface-water-chemical-status ). Surface water bodies in north-eastern Europe (e.g. France, Belgium, Germany, The Netherlands, Sweden) have very low chemical status with more than 60% of their surface waters reported to be less than good. UK, western and southern parts of France, Spain and Portugal have a high proportion of water bodies with good chemical status. However, for the last decade, there was no significant trend in this threat status for all OSPAR Regions.
- Fisheries exploitation
Fisheries exploitation of Atlantic salmon was a great concern in the past century, but average annual exploitation by commercial (cf. Figure 6) and recreational fisheries have greatly decreased over the last 60 years and are presently at very low levels (ICES, 2021). Fishing remains, nonetheless, a threat for many small or endangered populations and exploitation rates are still under careful review and management. In Norway (Forseth et al., 2020) and Sweden, exploitation rates had decreased for the last ten years due to new legislation and the closure of fisheries (2015 in Sweden). Exploitation rates had also greatly decreased in Scotland and Northern Ireland (with changes to legislation, the prohibition of some fisheries and the voluntary uptake in catch and release fishing) while it remained constant in France, Spain and Ireland. Many commercial net fisheries are closed in England with mandatory catch and release in others. A total ban on killing salmon was introduced in Wales in 2020 covering both commercial and recreational fishing, although both are still allowed on a catch and release basis. Additionally, the netting season has been reduced.
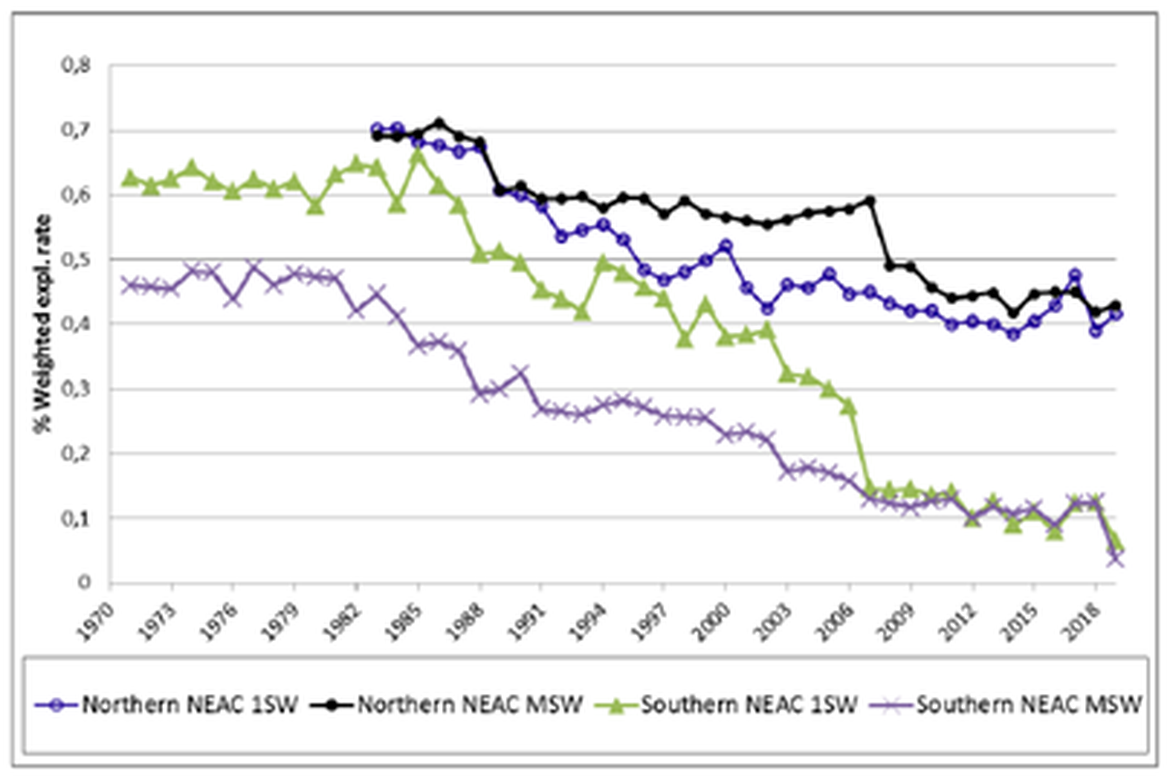
Figure 6 : Mean annual exploitation rate of wild One-Sea-Winter (1SW) and Multiple-Sea-Winter (MSW) Atlantic salmon by commercial and recreational fisheries in Northern and Southern NEAC (North Eastern Atlantic Commission) countries.; adapted from (ICES, 2021).
- Acidification
Once a major concern, especially in the northern most part of the OSPAR Region (I and II) acidification is associated with the premature mortality of wild Atlantic salmon and extirpation of some populations, acidification and acid rain seem to have decreased over the past decade. Indeed, emissions of all key air pollutants have reduced significantly (Figure 7) and for the most important acidifying compound, sulphur dioxide, emissions in Europe have decreased by 80% or more since the peaks around 1980–1990 (Grennfelt et al., 2020). Further, liming has made an important contribution to the restoration of salmon in formerly acidified rivers in Norway and Sweden (Hesthagen et al., 2011). Overall, the threat is either stated as stable (OSPAR Regions I and II), decreasing (OSPAR Region III) or not an issue (OSPAR Region IV).
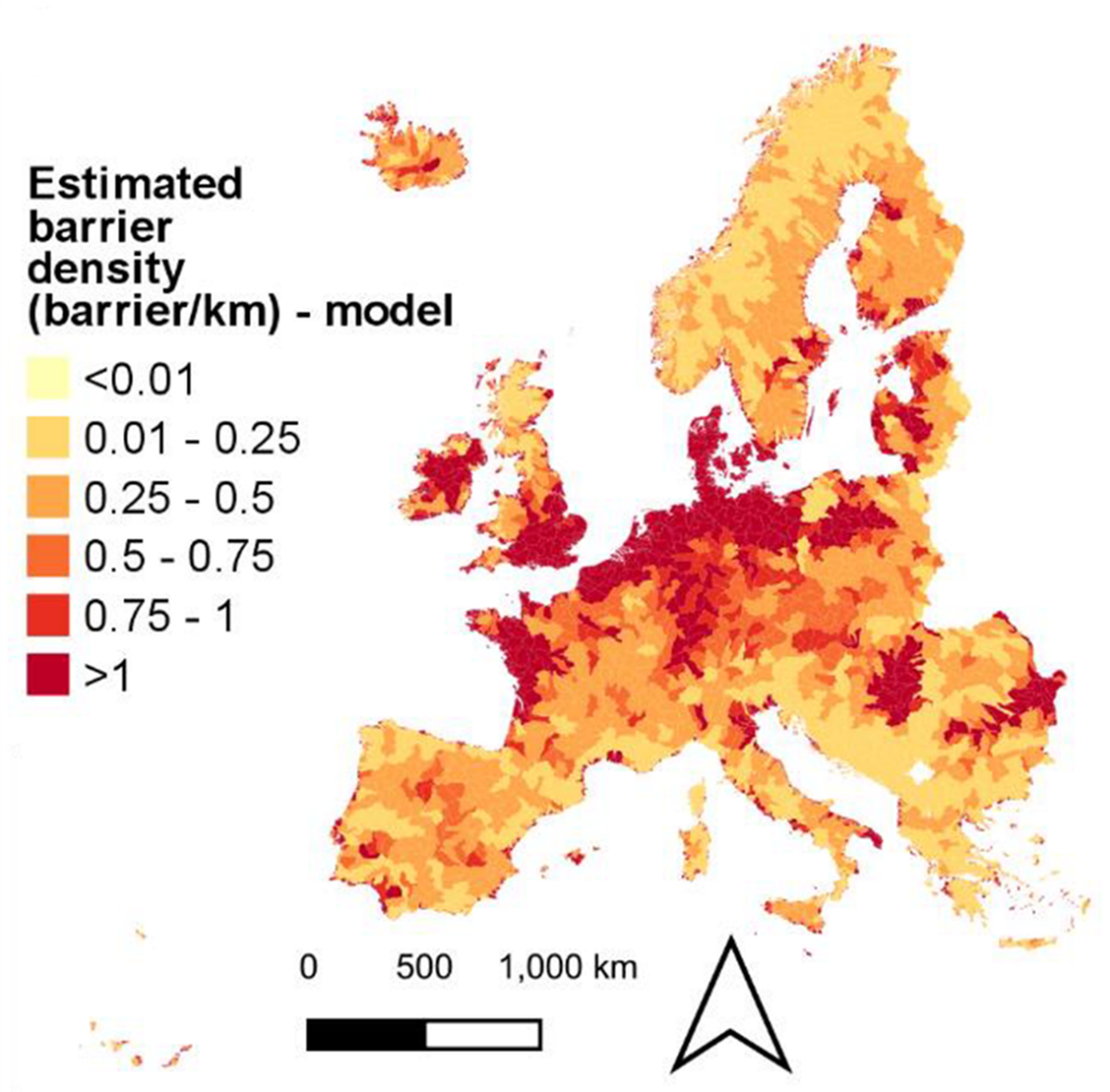
Figure 5: Extent of river fragmentation in Europe. The map shows the barrier density (barrier/km) in sub-catchments across Europe based on barrier modelling (Belletti et al., 2020)
- Hydrological manipulation, riverbed engineering schemes
In many European countries, hydropower production is affecting the quality and connectivity of river ecosystems. Many older concessions are being revised or closed leading to a potential decrease in threats. However, small-scale hydropower stations (S-SHS) programs are expanding in several countries and raise new concerns about the negative impacts on Atlantic salmon. Data are lacking, however, to enable an assessment of all potential effects (but see Bakken et al. (2012) for a review of effects in Norway). Overall, the present situation is not changing in OSPAR regions but the potential threat of S-SHS needs further study in the near future and probably better legislation to protect key habitats in some countries.
- Climate change
With most future climatic scenarios predicting higher temperatures and a rise in extreme events (e.g. flooding and drought), there is growing concern over the many effects on salmon feeding, growth, survival and migratory routes (reviewed by (Jonsson & Jonsson, 2009)). Nonetheless, recent climatic events had already been found to have effects both during the freshwater and marine phases of the salmon life cycle. Gregory et al. (2020) reported a salmon recruitment crash in 2016 in response to warm spawning temperatures and high river flows during the earliest life stages in Wales. Rikardsen et al. (2021) found that with the rise of sea temperature, migration routes of salmon population seemed to extend farther north. This could affect salmon populations if they cannot adapt to those longer migration routes, especially for southern populations (Portugal, Spain) which may need to travel longer distances to encounter appropriate foraging areas (Nicola et al., 2018). However, the effect of rising water temperatures is not uniform, with the potential for increases in marine survival for some stocks and decreasing marine survival for others (ICES, 2017).
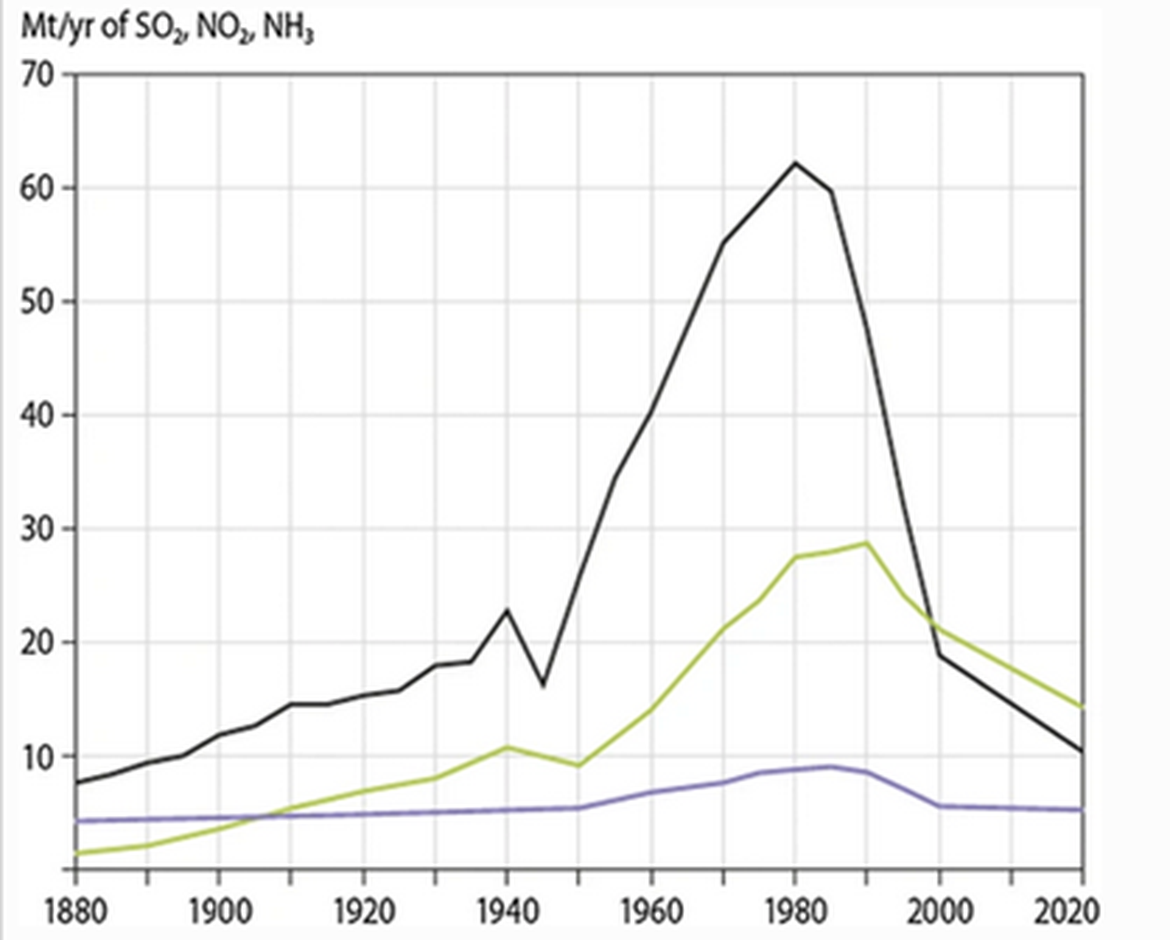
Figure 7: European emissions of Sulphur dioxide (black), nitrogen oxides (green) and ammonia (blue), 1880-2020, adapted from Grennfelt et al. (2020)
- Farmed salmonid aquaculture
Farmed salmonid aquaculture pose a recognized threat on wild salmon populations where it occurs (Norway, OSPAR Regions I & II; Scotland & Ireland, OSPAR Region III). Primary impacts include the effects of sea lice on post-smolts and genetic introgression by fish farm escapes which may negatively affect salmon health and survival (ICES, 2016). There is currently no known threat of salmon farming in OSPAR Region IV.
- Non-native salmonid invasion
Despite being captured in European rivers sporadically since 1960, since 2017 there have been numerous reports of significant numbers of Pink salmon entering Atlantic salmon rivers in several countries within the OSPAR Regions : Norway (Sandlund et al., 2019), Scotland (Armstrong et al., 2018; Copp, G. H, 2017), Ireland (Millane et al., 2019) and France (Beaulaton et al., 2021). However, population-level impacts from competition with Atlantic salmon is still unclear and more data and studies are needed, with a growing concern under climate change (i.e. expansion of distribution of pink salmon).
Measures that address key pressures from human activities or conserve the species/habitat
The following list of measures was derived from the Contracting Parties implementation reporting, complemented by national expert additions.
- All OSPAR Contracting Parties with salmon stocks have measures in place to protect salmon stocks, as in OSPAR Recommendation 2016/3. Most of these actions are described in the Implementation Plans for NASCO for those Contracting Parties that are also NASCO members (see https://nasco.int/conservation/implementation-plans-and-reporting/ ). Measures include fish passage facilities or removing obstacles to migration, recovering or introducing spawning gravels, restoring other habitats.
- Contracting Parties report annual stock assessments based on substantial data collection and analyses to ICES.
- All OSPAR Contracting Parties have legislative controls that seek to manage targeted salmon fisheries exploitation at levels that are sustainable
- Most Contracting Parties have designated Marine Protected Areas with Atlantic salmon as features
- Parties undertake a range of research programs covering genetics, population structure, life history, migration route, key functional habitats, marine survival, population dynamic modelling, optimal management, etc.
- Parties have undertaken awareness raising projects at local (education), national or international levels (International Year of the Salmon, World Fish Migration Days, etc.).
Conclusion (including management considerations)
For the last decade, since the last OSPAR assessment, this new assessment of Atlantic salmon reveals a poor situation at the scale of OSPAR Regions (I to IV). Studies of Atlantic salmon abundance suggested that the number of salmon is at best stable, but more often decreasing since 2010. There was also a common trend in the reduction of individual and population condition (loss of weight, length and reproductive potential) highlighted in all Regions although there is still a lack of data for many populations.
However, more than 2 000 genetically distinct salmon populations are distributed in water courses flowing towards the North Atlantic Ocean (Verspoor et al., 2007), and not all are experiencing similar trends or threats to the same extent. While there were successful efforts made on habitat restoration, enhancement of water quality and reintroduction programs (with success and failures), there is continuing concern about the expanding impacts of climate change on populations (Lennox et al., 2021; Thorstad et al., 2021). For example, the loss of thermal suitability of riverine habitats is indeed a growing threat and managers may need to adapt their future mitigation measures in some areas, e.g. planting riparian trees to provide solar shading, but this area requires a detailed understanding of the spatio-temporal variability of the phenomena affecting salmonids and their environments (Jackson et al., 2018). Those climate-induced changes may have further aggravated effects on specific Atlantic salmon populations, for instance in the southern part of OSPAR regions. Moreover, where migratory fish cross international borders (e.g. the Rhine Basin and its reintroducing program), management processes should include concerted and coordinated international actions to regulate fisheries and maintain river health.
Knowledge Gaps
- There remains limited data and understanding on the extent of the effects of climate change on the overall status of Atlantic salmon individuals, stocks, populations and international management units.
- Further data collection and harmonization to better acknowledge the impacts of non-fisheries threats on Atlantic salmon status
- Studies to acknowledge the impact of native (e.g. river lamprey (Kennedy et al., 2020) or great cormorant (Jepsen et al., (2019) and/or non-indigenous (Wels catfish in OSPAR region IV; see for instance Boulêtreau et al. (2018)) predators on Atlantic salmon survival. Together with other threats, this may hinder the recovery of some depleted salmon stocks.
- Overall, while many studies focus on the effect of a specific threat or class of threats, very few data exist on the combined effect from multiple pressures during different life stages, even with small effect, that could be detrimental for Atlantic salmon survival. It is moreover still very challenging to assess the causes of marine mortality of Atlantic salmon (see Crozier et al. (2018) for model improvements) given the very large area they occupy. This remains a key study area of institutions and international partnerships (e.g. NASCO).
Method used
Main source of information:
- Assessment lead was coordinated by France, namely J. Raitif (UMS OFB-CNRS-MNHN PatriNat) and A. Acou (UMS OFB-CNRS-MNHN PatriNat / Management of Diadromous Fish in their Environment OFB-INRAE-Institut Agro-UPPA, France).
- Two online meetings (23th of June and 23th of September 2021) and emailing were needed with national experts in order to complete data gathering, validate the methodology for assessment and review the final draft assessment.
- Experts involved in the assessment were:
Norway: P. Fiske
Sweden: I. A. Bergendahl
Denmark: N. Jepsen
United Kingdom: D. Ensing (ICES WGNAS Chairman), A. Walker, I. Davidson, J. Gillson, L. Talks, S. Walton, N. Hanson, C. Bean,
France: L. Beaulaton
Portugal: P.R. Almeida, C. Mateus, S. Silva, C. Alexandre
NASCO secretariat: E. Hatfield
Assessment is based upon:
a) Third party assessment (ICES WGNAS principally)
b) Literature studies
c) Direct or indirect data
d) Expert opinion of national experts
Armstrong, J. D., Bean, C. W., & Wells, A. (2018). The Scottish invasion of pink salmon in 2017. Journal of Fish Biology, 93(1), 8‑11. https://doi.org/10.1111/jfb.13680
Bacon, P. J., Palmer, S. C. F., MacLean, J. C., Smith, G. W., Whyte, B. D. M., Gurney, W. S. C., & Youngson, A. F. (2009). Empirical analyses of the length, weight, and condition of adult Atlantic salmon on return to the Scottish coast between 1963 and 2006. ICES Journal of Marine Science, 66(5), 844‑859. https://doi.org/10.1093/icesjms/fsp096
Baglinière, J.-L., Marchand, F., & Vauclin, V. (2005). Interannual changes in recruitment of the Atlantic salmon (Salmo salar) population in the River Oir (Lower Normandy, France) : Relationships with spawners and in-stream habitat. ICES Journal of Marine Science, 62(4), 695‑707. https://doi.org/10.1016/j.icesjms.2005.02.008
Bakken, T. H., Sundt, H., Ruud, A., & Harby, A. (2012). Development of Small Versus Large Hydropower in Norway– Comparison of Environmental Impacts. Energy Procedia, 20, 185‑199. https://doi.org/10.1016/j.egypro.2012.03.019
Beaulaton, L., Josset, Q., & Baglinière, J.-L. (2021). Le Saumon Rose (Oncorhynchus gorbuscha, Walbaum, 1792). Conduite à tenir et éléments d’écologie. (p. 25) [Report, OFB ; INRAE ; INSTITUT AGRO ; UPPA]. https://hal.archives-ouvertes.fr/hal-03287600
Belletti, B., Garcia de Leaniz, C., Jones, J., Bizzi, S., Börger, L., Segura, G., Castelletti, A., van de Bund, W., Aarestrup, K., Barry, J., Belka, K., Berkhuysen, A., Birnie-Gauvin, K., Bussettini, M., Carolli, M., Consuegra, S., Dopico, E., Feierfeil, T., Fernández, S., … Zalewski, M. (2020). More than one million barriers fragment Europe’s rivers. Nature, 588(7838), 436‑441. https://doi.org/10.1038/s41586-020-3005-2
Boulêtreau, S., Gaillagot, A., Carry, L., Tétard, S., De Oliveira, E., & Santoul, F. (2018). Adult Atlantic salmon have a new freshwater predator. PLOS ONE, 13(4), e0196046. https://doi.org/10.1371/journal.pone.0196046
Buddendorf, W. B., Jackson, F. L., Malcolm, I. A., Millidine, K. J., Geris, J., Wilkinson, M. E., & Soulsby, C. (2019). Integration of juvenile habitat quality and river connectivity models to understand and prioritise the management of barriers for Atlantic salmon populations across spatial scales. Science of The Total Environment, 655, 557‑566. https://doi.org/10.1016/j.scitotenv.2018.11.263
Copp, G. H. (2017). GB Non-native Species Rapid Risk Assessment (NRRA) : Rapid Risk Assessment of : Oncorhynchus gorbuscha (Walbaum) (pink or humpback salmon). https://www.cefas.co.uk/media/191673/cefas_rapidriskassessment_pinksalmon_05-09-17.pdf
Crozier, W., Whelan, K., Buoro, M., Chaput, G., Daniels, J., Grant, S., Hyatt, K., Irvine, J., Ó’Maoiléidigh, N., Prévost, E., Rivot, E., Russell, I., Schmidt, M., & Wells, B. (2018). Atlantic salmon mortality at sea : Developing an evidence-based « “Likely Suspects” » Framework. 38.
de Eyto, E., White, J., Boylan, P., Clarke, B., Cotter, D., Doherty, D., Gargan, P., Kennedy, R., McGinnity, P., O’Maoiléidigh, N., & O’Higgins, K. (2015). The fecundity of wild Irish Atlantic salmon Salmo salar L. and its application for stock assessment purposes. Fisheries Research, 164, 159‑169. https://doi.org/10.1016/j.fishres.2014.11.017
Forseth, T., Barlaup, B. T., Finstad, B., Fiske, P., Gjøsæter, H., Falkegård, M., Hindar, A., Mo, T. A., Rikardsen, A. H., Thorstad, E. B., Vøllestad, L. A., & Wennevik, V. (2017). The major threats to Atlantic salmon in Norway. ICES Journal of Marine Science, 74(6), 1496‑1513. https://doi.org/10.1093/icesjms/fsx020
Forseth, T., Thorstad, E. B., & Fiske, P. (2020). Status of wild Atlantic salmon in Norway 2020. https://vitenskapsradet.no/Portals/vitenskapsradet/Pdf/Status%20of%20wild%20Atlantic%20salmon%20in%20Norway%202020T.pdf?ver=5edE21dH2DFOaPNai04Uig%3d%3d
Gregory, S. D., Bewes, V. E., Davey, A. J. H., Roberts, D. E., Gough, P., & Davidson, I. C. (2020). Environmental conditions modify density‐dependent salmonid recruitment : Insights into the 2016 recruitment crash in Wales. Freshwater Biology, 65(12), 2135‑2153. https://doi.org/10.1111/fwb.13609
Grennfelt, P., Engleryd, A., Forsius, M., Hov, Ø., Rodhe, H., & Cowling, E. (2020). Acid rain and air pollution : 50 years of progress in environmental science and policy. Ambio, 49(4), 849‑864. https://doi.org/10.1007/s13280-019-01244-4
Grill, G., Lehner, B., Thieme, M., Geenen, B., Tickner, D., Antonelli, F., Babu, S., Borrelli, P., Cheng, L., Crochetiere, H., Ehalt Macedo, H., Filgueiras, R., Goichot, M., Higgins, J., Hogan, Z., Lip, B., McClain, M. E., Meng, J., Mulligan, M., … Zarfl, C. (2019). Mapping the world’s free-flowing rivers. Nature, 569(7755), 215‑221. https://doi.org/10.1038/s41586-019-1111-9
Havn, T. B., Thorstad, E. B., Borcherding, J., Heermann, L., Teichert, M. A. K., Ingendahl, D., Tambets, M., Sæther, S. A., & Økland, F. (2020). Impacts of a weir and power station on downstream migrating Atlantic salmon smolts in a German river. River Research and Applications, 36(5), 784‑796. https://doi.org/10.1002/rra.3590
Hesthagen, T., Larsen, B. M., & Fiske, P. (2011). Liming restores Atlantic salmon (Salmo salar) populations in acidified Norwegian rivers. Canadian Journal of Fisheries and Aquatic Sciences, 68(2), 224‑231. https://doi.org/10.1139/F10-133
ICES. (2016). Report of the Workshop to address the NASCO request for advice on possible effects of salmonid aquaculture on wild Atlantic salmon populations in the North Atlantic (WKCULEF), 1-3 March 2016, Charlottenlund, Denmark. ICES CM 2016/ACOM:42. 44 pp.
ICES. (2017). Report of the Workshop on Potential Impacts of Climate Change on Atlantic Salmon Stock Dynamics (WKCCISAL) (ICES CM 2017/ACOM:39; p. 90). International Council for the Exploration of the Sea. https://www.ices.dk/sites/pub/Publication%20Reports/Expert%20Group%20Report/acom/2017/WKCCISAL/wkccisal_2017.pdf
ICES. (2021). Working Group on North Atlantic Salmon. https://doi.org/10.17895/ICES.PUB.7923
Jackson, F. L., Fryer, R. J., Hannah, D. M., Millar, C. P., & Malcolm, I. A. (2018). A spatio-temporal statistical model of maximum daily river temperatures to inform the management of Scotland’s Atlantic salmon rivers under climate change. Science of The Total Environment, 612, 1543‑1558. https://doi.org/10.1016/j.scitotenv.2017.09.010
Jacobson, P., Whitlock, R., Huss, M., Leonardsson, K., Östergren, J., & Gårdmark, A. (2021). Growth variation of Atlantic salmon Salmo salar at sea affects their population-specific reproductive potential. Marine Ecology Progress Series, 671, 165‑174. https://doi.org/10.3354/meps13734
Jensen, A. J., Karlsson, S., Fiske, P., Hansen, L. P., Østborg, G. M., & Hindar, K. (2014). Origin and life history of Atlantic salmon (Salmo salar) near their northernmost oceanic limit. Canadian Journal of Fisheries and Aquatic Sciences, 71(11), 1740‑1746. https://doi.org/10.1139/cjfas-2014-0169
Jepsen, N., Flávio, H., & Koed, A. (2019). The impact of Cormorant predation on Atlantic salmon and Sea trout smolt survival. Fisheries Management and Ecology, 26(2), 183‑186. https://doi.org/10.1111/fme.12329
Jonsson, B., & Jonsson, N. (2009). A review of the likely effects of climate change on anadromous Atlantic salmon Salmo salar and brown trout Salmo trutta, with particular reference to water temperature and flow. Journal of Fish Biology, 75(10), 2381‑2447. https://doi.org/10.1111/j.1095-8649.2009.02380.x
Kennedy, R. J., Campbell, W., Gallagher, K., & Evans, D. (2020). River lamprey present an unusual predation threat to Atlantic salmon smolts in Lough Neagh, Northern Ireland. Journal of Fish Biology, 97(4), 1265‑1267. https://doi.org/10.1111/jfb.14477
Koed, A., Birnie-Gauvin, K., Sivebæk, F., & Aarestrup, K. (2020). From endangered to sustainable : Multi-faceted management in rivers and coasts improves Atlantic salmon (Salmo salar) populations in Denmark. Fisheries Management and Ecology, 27(1), 64‑76. https://doi.org/10.1111/fme.12385
Legrand, M., Briand, C., Buisson, L., Artur, G., Azam, D., Baisez, A., Barracou, D., Bourré, N., Carry, L., Caudal, A.-L., Charrier, F., Corre, J., Croguennec, E., Mikaélian, S. D., Josset, Q., Gurun, L. L., Schaeffer, F., & Laffaille, P. (2020). Contrasting trends between species and catchments in diadromous fish counts over the last 30 years in France. Knowledge & Management of Aquatic Ecosystems, 421, 7. https://doi.org/10.1051/kmae/2019046
Lennox, R. J., Alexandre, C. M., Almeida, P. R., Bailey, K. M., Barlaup, B. T., Bøe, K., Breukelaar, A., Erkinaro, J., Forseth, T., Gabrielsen, S.-E., Halfyard, E., Hanssen, E. M., Karlsson, S., Koch, S., Koed, A., Langåker, R. M., Lo, H., Lucas, M. C., Mahlum, S., … Vollset, K. W. (2021). The quest for successful Atlantic salmon restoration : Perspectives, priorities, and maxims. ICES Journal of Marine Science, fsab201. https://doi.org/10.1093/icesjms/fsab201
Malcolm, I. A., Millidine, K. J., Glover, R. S., Jackson, F. L., Millar, C. P., & Fryer, R. J. (2019). Development of a large-scale juvenile density model to inform the assessment and management of Atlantic salmon (Salmo salar) populations in Scotland. Ecological Indicators, 96, 303‑316. https://doi.org/10.1016/j.ecolind.2018.09.005
Maoiléidigh, N. Ó. (2018). Fifty years of marine tag recoveries from Atlantic salmon. International Council for the Exploration of the Sea.
Millane, M., Walsh, L., Roche, W. K., & Gargan, P. G. (2019). Unprecedented widespread occurrence of Pink Salmon Oncorhynchus gorbuscha in Ireland in 2017. Journal of Fish Biology, 95(2), 651‑654. https://doi.org/10.1111/jfb.13994
Monnerjahn, U. (2011). Atlantic Salmon (Salmo salar L.) re-introduction in Germany : A status report on national programmes and activities. Journal of Applied Ichthyology, 27(s3), 33‑40. https://doi.org/10.1111/j.1439-0426.2011.01849.x
Mota, M., Rochard, E., & Antunes, C. (2016). Status of the Diadromous Fish of the Iberian Peninsula : Past, Present and Trends. 29, 1‑18.
NASCO. (2017). Annual Progress Report on Actions Taken Under the Implementation Plan for the Calendar Year 2016 EU-UK (England and Wales) (revised 28 April 2017 with updated estimate of unreported catch) (p. 28 p.). https://nasco.int/wp-content/uploads/2020/02/CNL_17_31rev_APR_EU_UK_EnglandandWales.pdf
NASCO. (2020a). Annual Progress Report on Actions Taken Under the Implementation Plan for the Calendar Year 2020—UK-England and Wales (p. 30 p.). https://nasco.int/wp-content/uploads/2021/04/CNL2129_Annual-Progress-Report_UK-England-and-Wales.pdf
NASCO. (2020b). State of North Atlantic Salmon.
Nicola, G. G., Elvira, B., Jonsson, B., Ayllón, D., & Almodóvar, A. (2018). Local and global climatic drivers of Atlantic salmon decline in southern Europe. Fisheries Research, 198, 78‑85. https://doi.org/10.1016/j.fishres.2017.10.012
OSPAR. (2008). Case reports of the OSPAR List of Threatened and/or Declining Species and Habitats (Biodiversity series, p. 150 pp.).
OSPAR. (2010). Background Document for Atlantic salmon Salmo salar (OSPAR Commision report, p. 25 pp. + annex).
Perrier, C., Evanno, G., Belliard, J., Guyomard, R., & Baglinière, J.-L. (2010). Natural recolonization of the Seine River by Atlantic salmon (Salmo salar) of multiple origins. Canadian Journal of Fisheries and Aquatic Sciences, 67(1), 1‑4. https://doi.org/10.1139/F09-190
Prouzet, P., & Michelet, N. (2019). Ecological Status of Atlantic Salmon (Salmo salar L.) in France : Need for an Ecosystemic Approach. In T. Komatsu, H.-J. Ceccaldi, J. Yoshida, P. Prouzet, & Y. Henocque (Éds.), Oceanography Challenges to Future Earth (p. 249‑276). Springer International Publishing. https://doi.org/10.1007/978-3-030-00138-4_20
Quinn, T. P., McGinnity, P., & Cross, T. F. (2006). Long-term declines in body size and shifts in run timing of Atlantic salmon in Ireland. Journal of Fish Biology, 68(6), 1713‑1730. https://doi.org/10.1111/j.0022-1112.2006.01017.x
Rikardsen, A. H., Righton, D., Strøm, J. F., Thorstad, E. B., Gargan, P., Sheehan, T., Økland, F., Chittenden, C. M., Hedger, R. D., Næsje, T. F., Renkawitz, M., Sturlaugsson, J., Caballero, P., Baktoft, H., Davidsen, J. G., Halttunen, E., Wright, S., Finstad, B., & Aarestrup, K. (2021). Redefining the oceanic distribution of Atlantic salmon. Scientific Reports, 11(1), 12266. https://doi.org/10.1038/s41598-021-91137-y
Sandlund, O. T., Berntsen, H. H., Fiske, P., Kuusela, J., Muladal, R., Niemelä, E., Uglem, I., Forseth, T., Mo, T. A., Thorstad, E. B., Veselov, A. E., Vollset, K. W., & Zubchenko, A. V. (2019). Pink salmon in Norway : The reluctant invader. Biological Invasions, 21(4), 1033‑1054. https://doi.org/10.1007/s10530-018-1904-z
Saura, M., Morán, P., Brotherstone, S., Caballero, A., Álvarez, J., & Villanueva, B. (2010). Predictions of response to selection caused by angling in a wild population of Atlantic salmon (Salmo salar). Freshwater Biology, 55(4), 923‑930. https://doi.org/10.1111/j.1365-2427.2009.02346.x
Thorstad, E. B., Bliss, D., Breau, C., Damon-Randall, K., Sundt-Hansen, L. E., Hatfield, E. M. C., Horsburgh, G., Hansen, H., Maoiléidigh, N. Ó., Sheehan, T., & Sutton, S. G. (2021). Atlantic salmon in a rapidly changing environment—Facing the challenges of reduced marine survival and climate change. Aquatic Conservation: Marine and Freshwater Ecosystems, 31(9), 2654‑2665. https://doi.org/10.1002/aqc.3624
Todd, C. D., Hanson, N. N., Boehme, L., Revie, C. W., & Marques, A. R. (2021). Variation in the post-smolt growth pattern of wild one sea-winter salmon (Salmo salar L.), and its linkage to surface warming in the eastern North Atlantic Ocean. Journal of Fish Biology, 98(1), 6‑16. https://doi.org/10.1111/jfb.14552
Verspoor, E., Stradmeyer, L., & Nielsen, J. L. (Éds.). (2007). The atlantic salmon : Genetics, conservation and management. Blackwell Publishing Ltd.; USGS Publications Warehouse. https://doi.org/10.1002/978047099584
Sheet reference:
BDC2022/Atlantic salmon