Status Assessment 2023 - Black-legged Kittiwake
| Assessment of status1 | Non-Breeding Distribution | Non-Breeding Population size | Breeding Distribution | Breeding Population size | Condition i.e. breeding productivity | Breeding Status (overall assessment)3 | Previous OSPAR status assessment 2010 | |
|---|---|---|---|---|---|---|---|---|
Region | I | ? | ? | 5 ←→ | 1↓ | 1↓ | 1↓ | ● |
| II | ? | 1↑2 | 5 ←→ | 1↓ | 1↓ | 1↓ | ● | |
| III | ? | ? | 1 ↓ | 1↓ | 1↓ | 1↓ | ○ | |
| IV | ? | ? | 1 ↓ | 1↓ | ? | 1↓ | ○ | |
| V | ? | ? | NA | NA | NA | ? | ○ | |
| Assessment of Pressures | Climate change impacts (adirect: shift in breeding distribution; bindirect: reduced food supply) | Over-exploitation of natural resources - fishing depletion of small forage fish stocks (e.g. sandeel and herring) | Threat or impact (conclusion based on the above rows) | ||
|---|---|---|---|---|---|
Region | I | 3 ↓b | 3 ←→ | 3 ↓ | |
| II | 3 ←→b | 3 ↓ | 3 ↓ | ||
| III | 3↑a | 5←→ | 3,5↑ | ||
| IV | 3a, 4b | ←→ | ? | 3,4←→ | |
| V | ? | ? | ? | ||
NOTES:
- Kittiwake are migratory – they breed onshore in the spring and summer and then range over large areas of the north Atlantic, often far away from land. They are therefore subject to different threats when breeding and when away from the colonies. The table provides separate assessments of population size and distribution of breeding and non-breeding populations.
- Assessment of wintering numbers offshore in the southern North Sea only, using data form NL, BE and DE. Source: Pilot assessment of B1 Marine bird abundance – non-breeding birds offshore (OSPAR, 2023b).
- Overall status determined by integrating indicator assessments – see QSR 2023 Thematic Assessment on Marine Birds and CEMP Guidelines for the integration rules for marine bird assessments.
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document i.e. 2009 using data up to 2006 (OSPAR, 2009))
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: If in QSR 2010 then enter regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ). If a more recent status assessment is available, then enter ‘poor’/’good’
Status (overall assessment)
red – poor
green – good
Blue cells –? status unknown, insufficient information available,
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressure
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threat or impact [overall assessment]
red – significant threat or impact;
green –no evidence of a significant threat or impact
Blue cells –? insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
Confidence
High
Background Information
Year added to OSPAR List of Threatened and/or Declining Species and Habitats (T&D List): 2008 (OSPAR 2009)
- Global/regional importance: 85% of the breeding population of the subspecies R. t. tridactyla occurred within the OSPAR Area at the time of listing (2008).
- Decline: Using breeding population data up to 2006, the species was evaluated as significantly declined at the time of listing, in particular, in Greenland, Norway and the UK.
- Sensitivity: The species was considered sensitive at the time of listing due to its low resilience to adverse effects from human activity, with recovery likely to be slow due to its life history characteristics (long-lived and relatively slow to reproduce).
- Anthropogenic pressures and biological factors: The species was considered threatened due to changes in the availability of key prey species, linked to possible climate change factors and over-fishing in parts of its range, e.g. sandeel fisheries in the UK. Predation in some areas, e.g. by Great Skuas Stercorarius skua in Shetland, and White-tailed eagles Haliaeetus albicilla in Norway, were also cited as threats.
Last status assessment: 2009 (based on data up to 2006) - OSPAR (2009).
Geographical Range and Distribution
Breeding distribution
The breeding distribution of Black-legged kittiwakes is confined to more northerly latitudes on both sides of the Atlantic (Figure 1) and is restricted by the availability of sea cliffs where they nest. Modelling predicts that by 2100, the climate in the eastern North Sea and in the Bay of Biscay and Iberian Coast will no longer be suitable for breeding kittiwakes (Russell et al. 2015). Indeed, only a few pairs now remain breeding in the entire Bay of Biscay OSPAR Region (Simian et al. 2018; Legroux 2019), although climate is probably affecting them indirectly by reducing the abundance of their prey (Martínez-Abraín et al. 2019). No constriction in range is yet evident in the North Sea. However, in the Celtic Seas, the southeast corner of their range is constricting northwards: all colonies have disappeared from Brittany in NW France during the last 10 years (Legroux 2019) and many colonies have also gone extinct along the SW coast of the UK (McMurdo-Hamilton 2016).
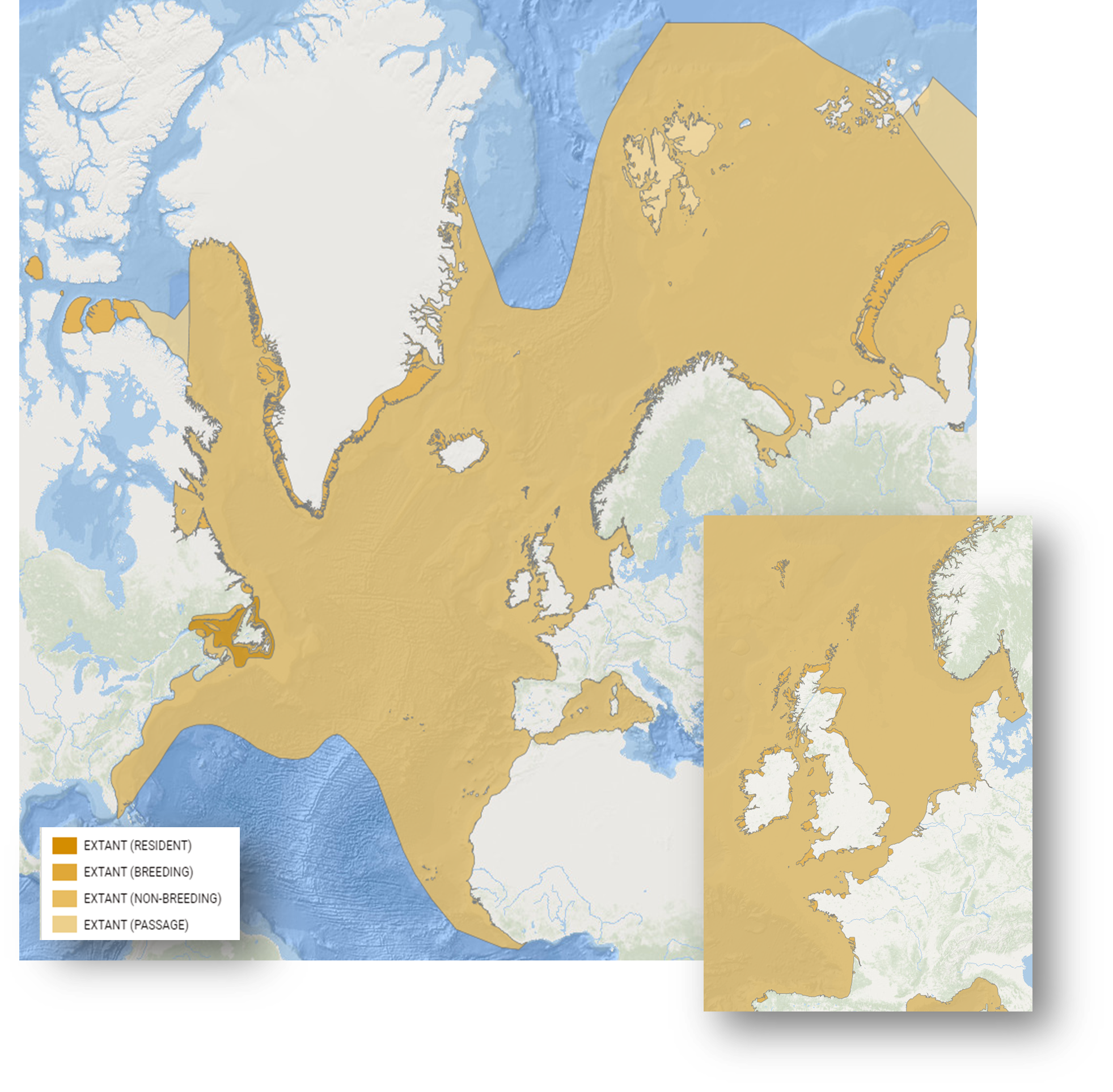
Figure 1: Breeding and non-breeding distribution of Black-legged kittiwake - sub-species R. t. tridactyla (Source: IUCN Red List at https://www.iucnredlist.org/species/22694497/155617539).
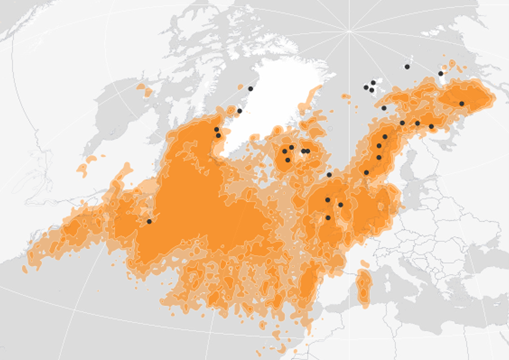
Figure 2: Distribution of Black-legged kittiwakes ouside the breeding season (i.e. excludes May-July) during 2007 to 2021. Distribution of 1 285 individual kittiwakes fitted with geolocators at 28 colonies (black dots). Source: //seapop.no/en/seatrack/ (accessed on 12/09/22).
Non-breeding distribution
Individual kittiwakes cover thousands of kilometres outside the breeding season (Figue 2). Birds breeding in the North Sea and Celtic Seas leave colonies and head north to waters around Greenland and will then spend the winter in the Western Atlantic off Newfoundland and mainland Canada (Frederiksen et al. 2012). Some will also head south to the Azores and off mainland Portugal or as far south as 30deg N. Birds breeding at more northerly colonies tend to remain furter north in the winter and some stay north of the Arctic circle. As the SEATRACK dataset on individual bird movements expands into the future, it may be possible to detect changes indistribution of kittiwakes at sea outside the breeding season across their entire range in the North Atlantic.
Method of assessment: 1 Data driven - Bay of Biscay, Celtic Seas; 5 Expert judgement - other regions
Population/Abundance
Trends in OSPAR’s common indicator of marine bird abundance (Figure 3) show that there have been substantial declines in breeding numbers of kittiwakes since the baseline in 1991 in all four regions where they breed. In 1991, there were around 1,8 million pairs of kittiwakes in the monitored areas, but by 2015-2020 there were only 600 000 pairs (Table 1). The decline in Arctic waters was the most substantial in terms of numbers of birds and overall decline - 70%. The declining status of kittiwakes breeding in the Celtic Seas and Bay of Biscay was not identified when kittiwakes were added to the T&D List in 2008, or in the subsequent background document (OSPAR, 2009).
Table 1: Predicted number of breeding pairs and relative breeding abundance (a proportion of the baseline in 1991) of kittiwakes in each OSPAR Region compared to the period 2001-06, used in the last assessment in 2009 (Source: OSPAR 2023a; ~A. Chabrolle pers. Comm.).
Baseline 19912 | 2001-2006 | 2014-2019 (2015-20# 2019-20~) | |||
|---|---|---|---|---|---|
| OSPAR Region | No. Pairs | No. pairs | Relative abundance | No. pairs | Relative abundance |
| Arctic (Norwegian part only) | 1 233 810 | 419 495 | 0,34 | 370 143# | 0,30# |
| Greater North Sea | 505 694 | 277 051 | 0,55 | 183,304 | 0,36 |
| Celtic Seas (UK & France) | 87 079 | 59 458 | 0,68 | 36 524 | 0,42 |
| Bay of Biscay & Iberian Coast | 263 | 108 | 0,41 | 22~ | 0,08~ |
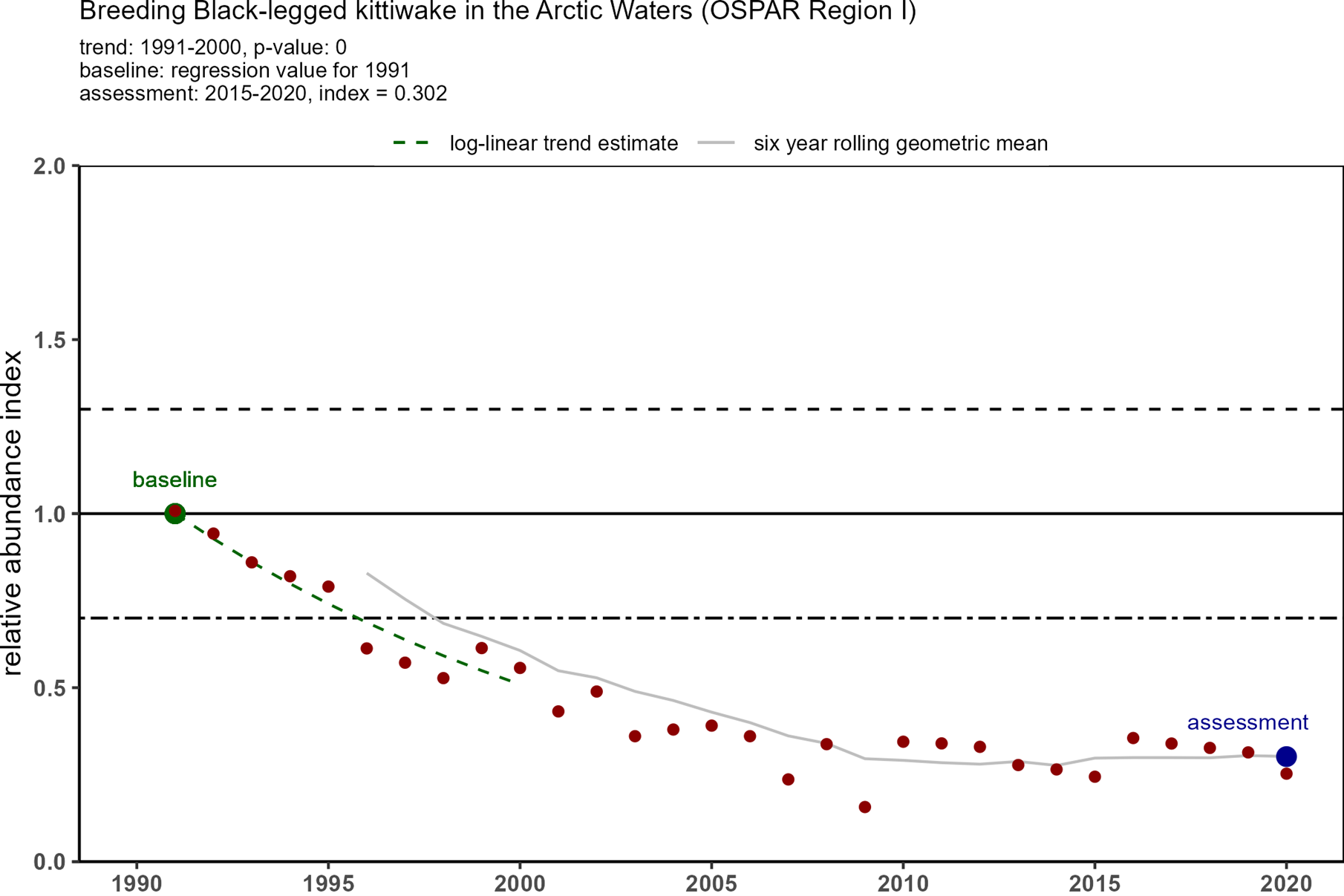
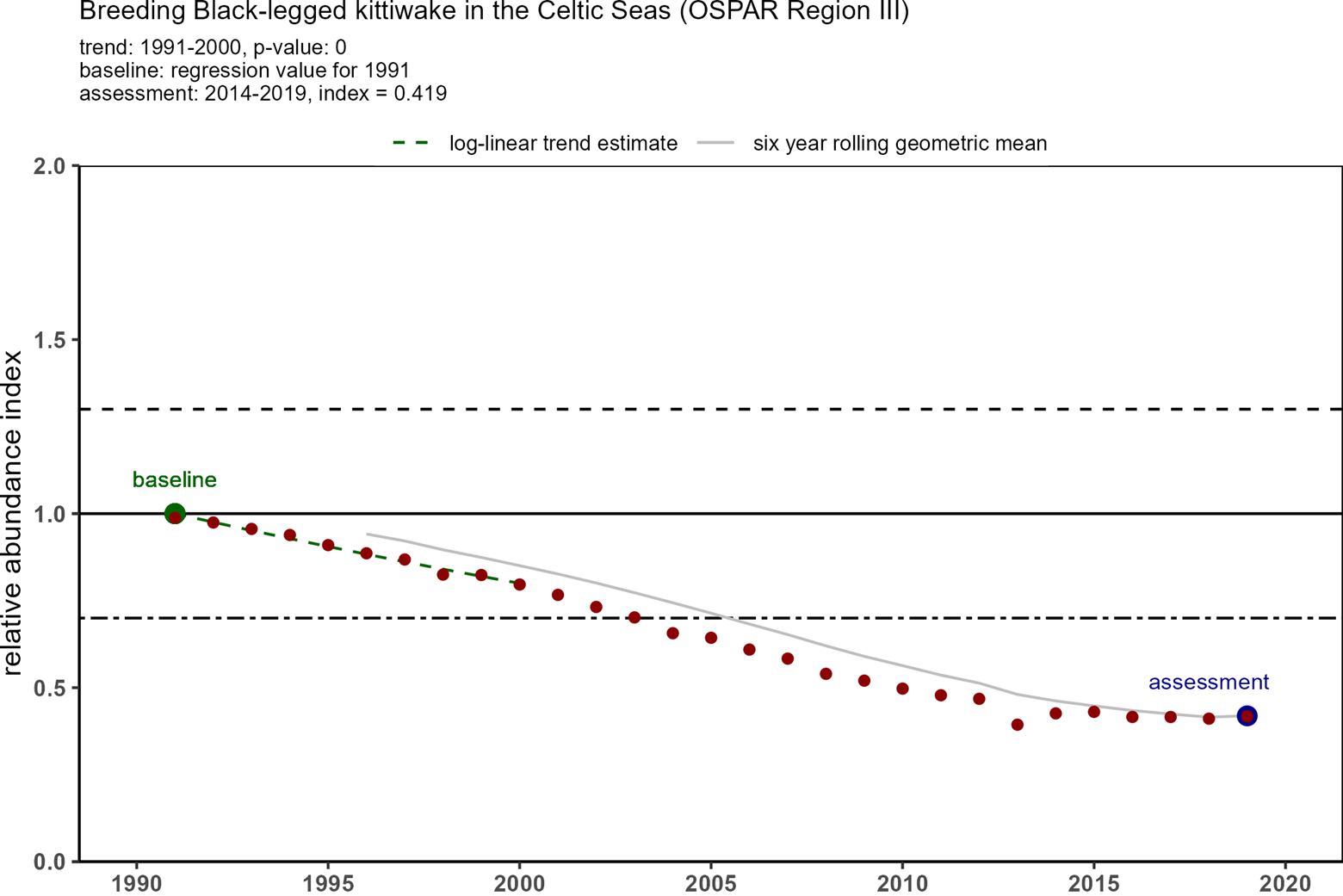
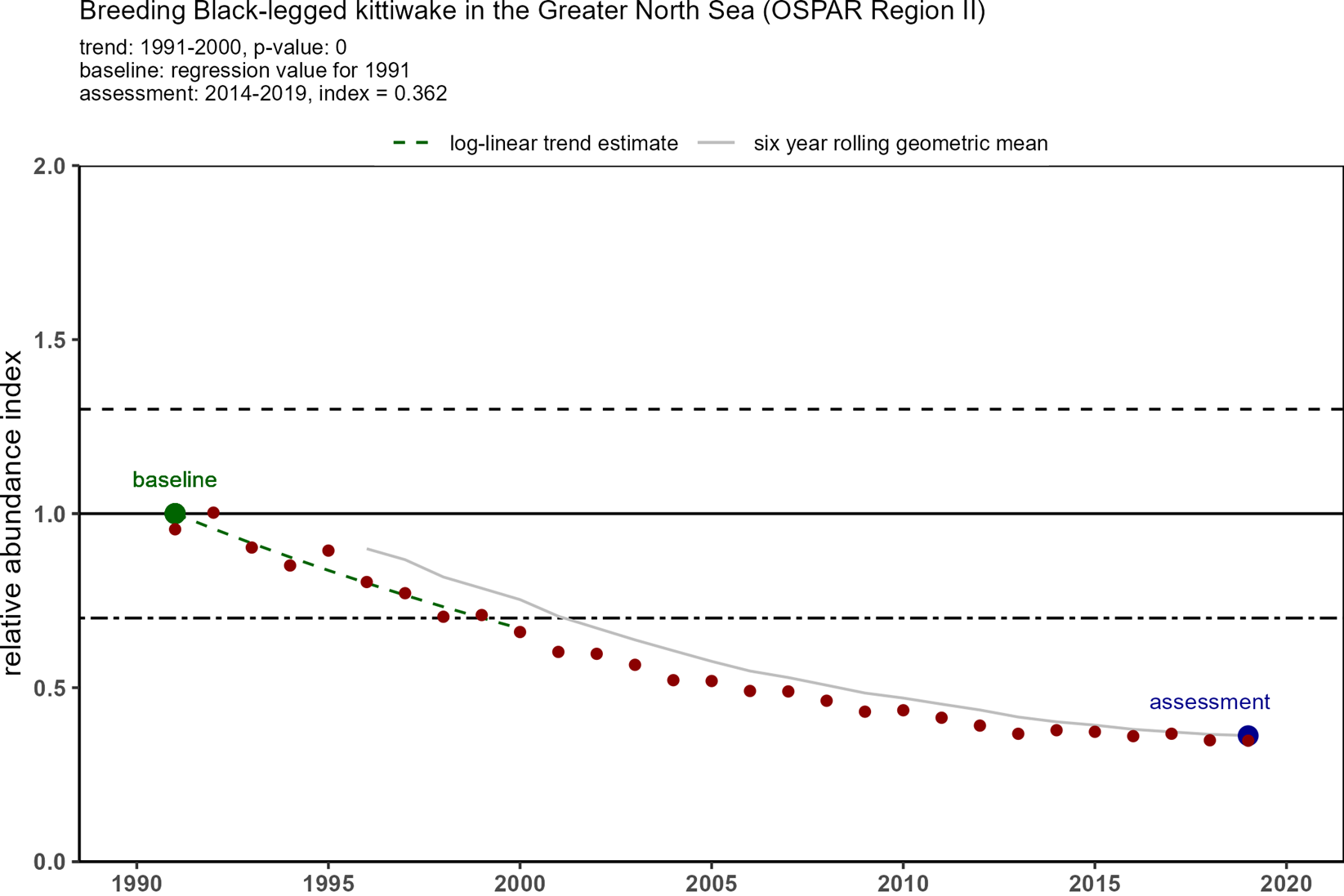
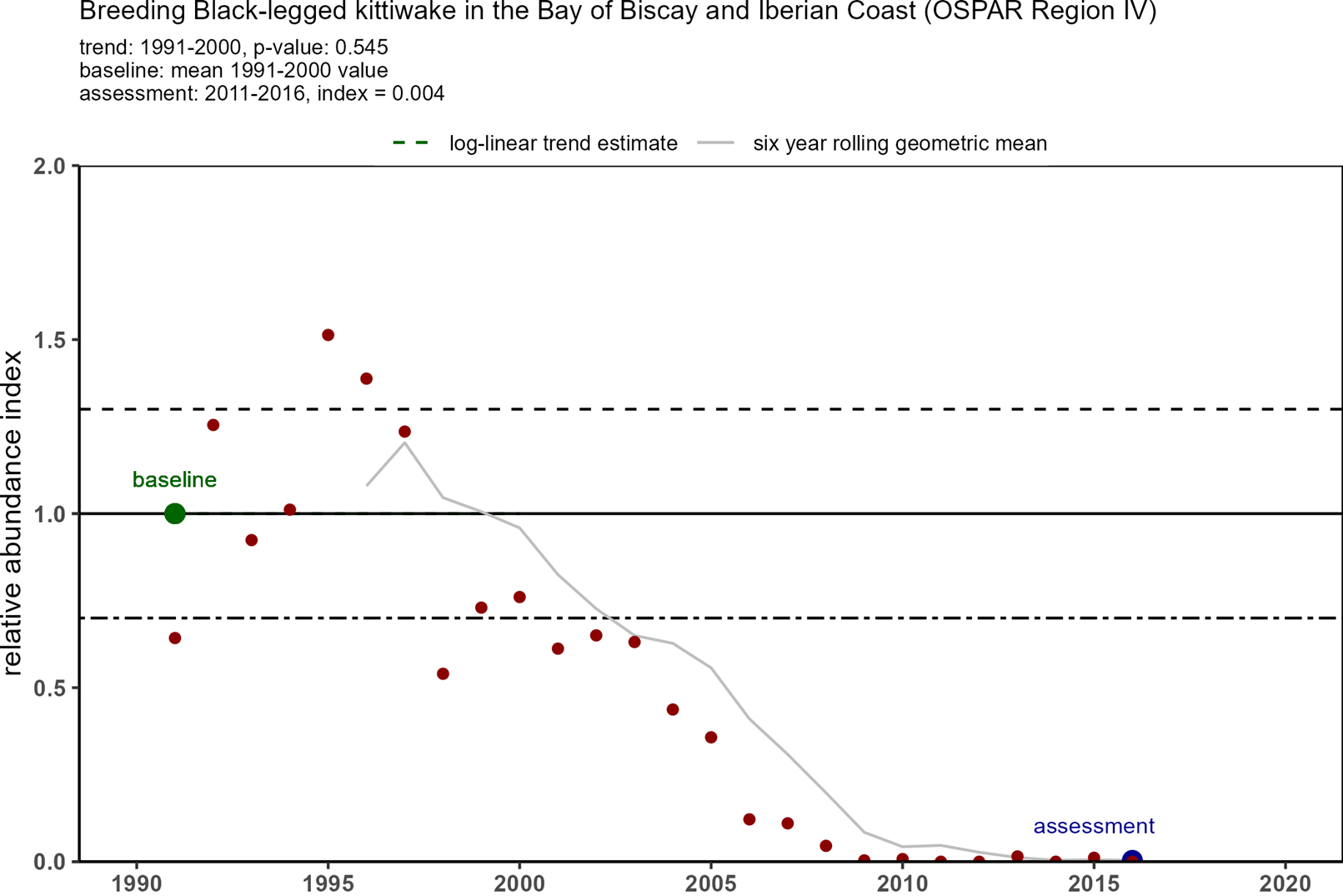
Figure 3: Trends in relative breeding abundance of black-legged kittiwake in each of OSPAR Regions I, II, III and IV, from 1991 onwards to 2020 (depending on Region). Datapoints represent yearly relative abundance values and the grey line represent the six-year rolling relative abundance geometric mean. The black line indicates the baseline which is calculated from the first ten years of data1. The black dotted line indicates the lower threshold value of 0,7 (for species that lay >1 egg). The assessment value (in blue) is the geometric mean of the last six years (e.g. 2015-2020). Kittiwake abundance was assessed as ’good’ or ’poor’ depending on whether the assessment value was higher or lower than the threshold value of 0,7. (Source: OSPAR 2023a).
Method of assessment: 1 Data driven; Source: OSPAR (2023a)
Condition
The condition of breeding populations of kittiwakes has been assessed using the common indicator of marine bird breeding productivity from the QSR2023 (OSPAR 2023b). The new approach predicts how observed levels of breeding productivity (number of chicks successfully fledging each year) may impact the long-term population growth rate of a species. It was not assessed in Region IV where very few, if any kittiwakes have bred since 2010. In the other Regions, kittiwake breeding productivity has fluctuated from year to year, but there was a clear decline during late 1990s and early 2000s to a low around 2010 (Figure 4). Subsequently, between 2010 and 2020, productivity has been increasing in all Regions. However, the levels of productivity are still below what would be required to sustain growth and recovery in these depleted populations (Figure 5). The poor growth rate in Region I, which has persisted since 1990, reflects the species’ status there as Critically Endangered (under IUCN Red List Criteria). The recovery of productivity in Regions II and III is still insufficent to sustain population growth and the species would be considered to be Endangered (under IUCN Criteria) in both Regions (Figure 5). Productivity would need to rise above 0,99 chicks per pair in Region I, 1,15 in Region II and 1,02 in Region III for population growth to be achieved, given the prevailing levels of age-specific survival in each population (Figure 4).
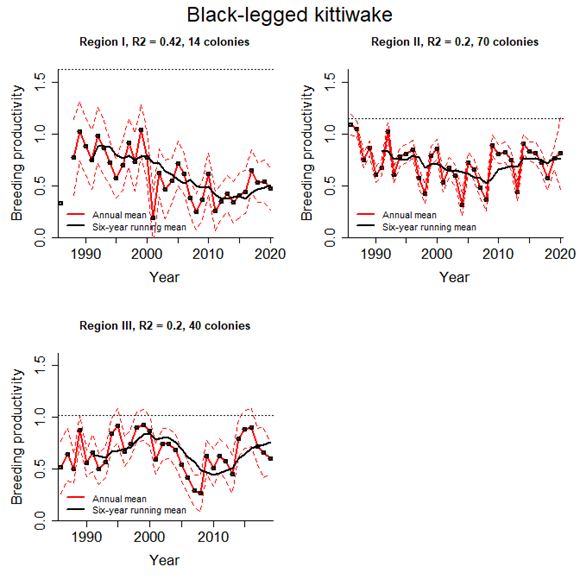
Figure 4 - Trends in annual breeding productivity (mean number of chicks fledged per pair) of black-legged kittiwake in each of OSPAR Regions I, II and III, from 1986 to 2019 or 2020 (depending on region). Dashed red lines indicate upper and lower 95% confidence limits around the annual mean productivity estimates (data points connected by red line). The black line represent the six-year rolling mean productivity. Horizontal dotted lines show the breeding productivity required to maintain a stable population, given the prevailing levels of age-specific survival: 1,62 in Region I, 1,15 in Region II and 1,02 in Region III. (Source: OSPAR 2023b).
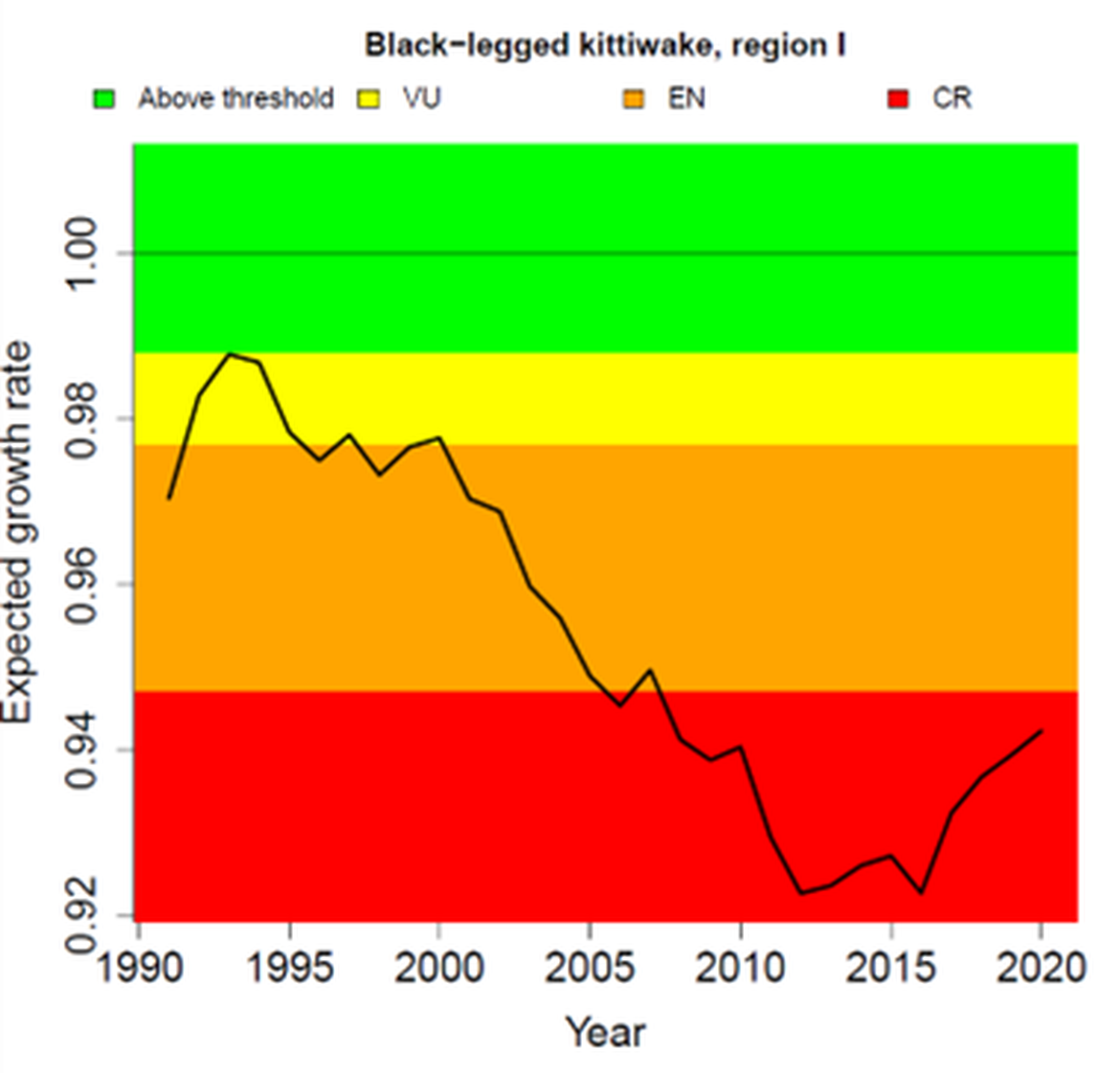
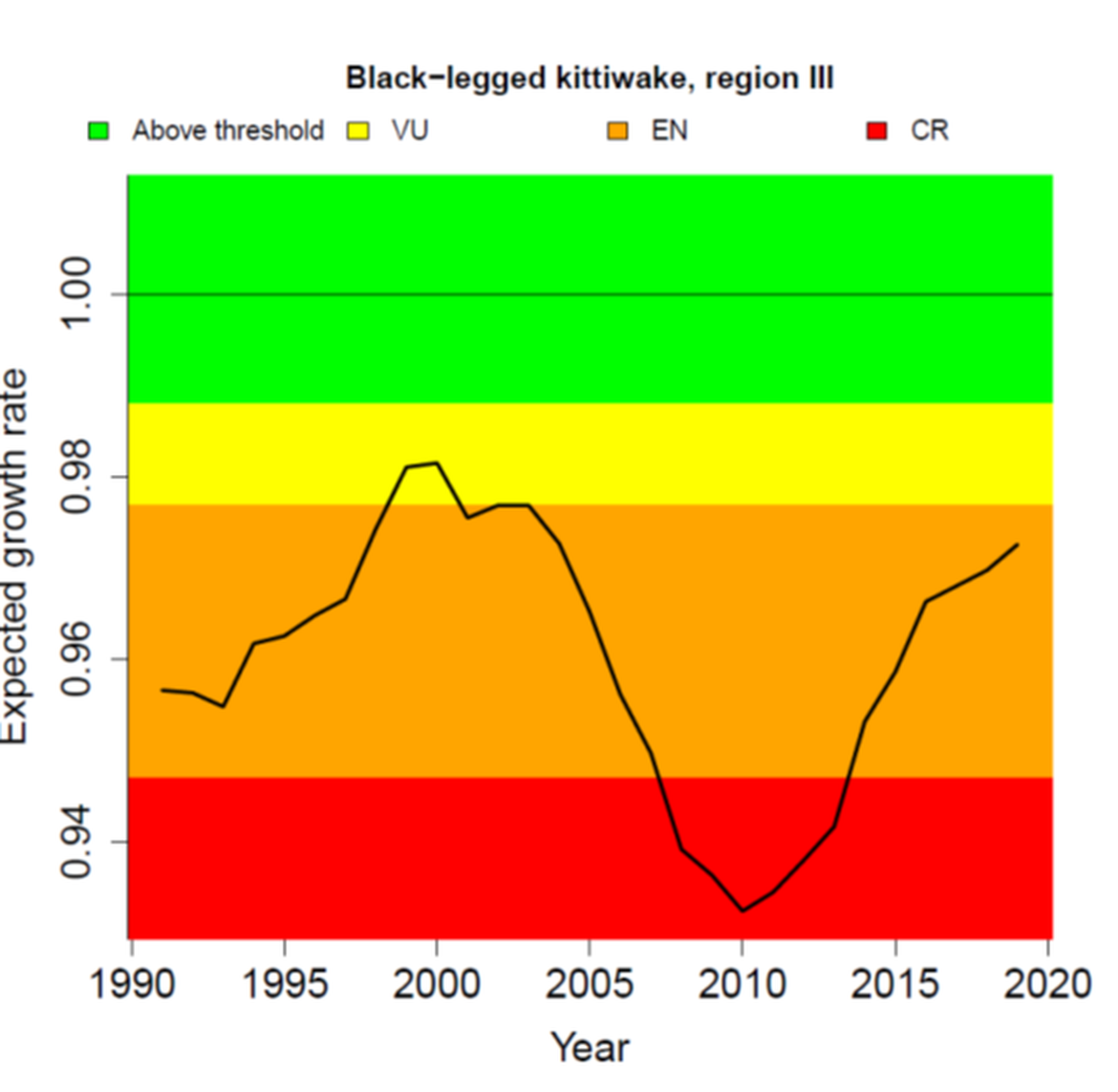
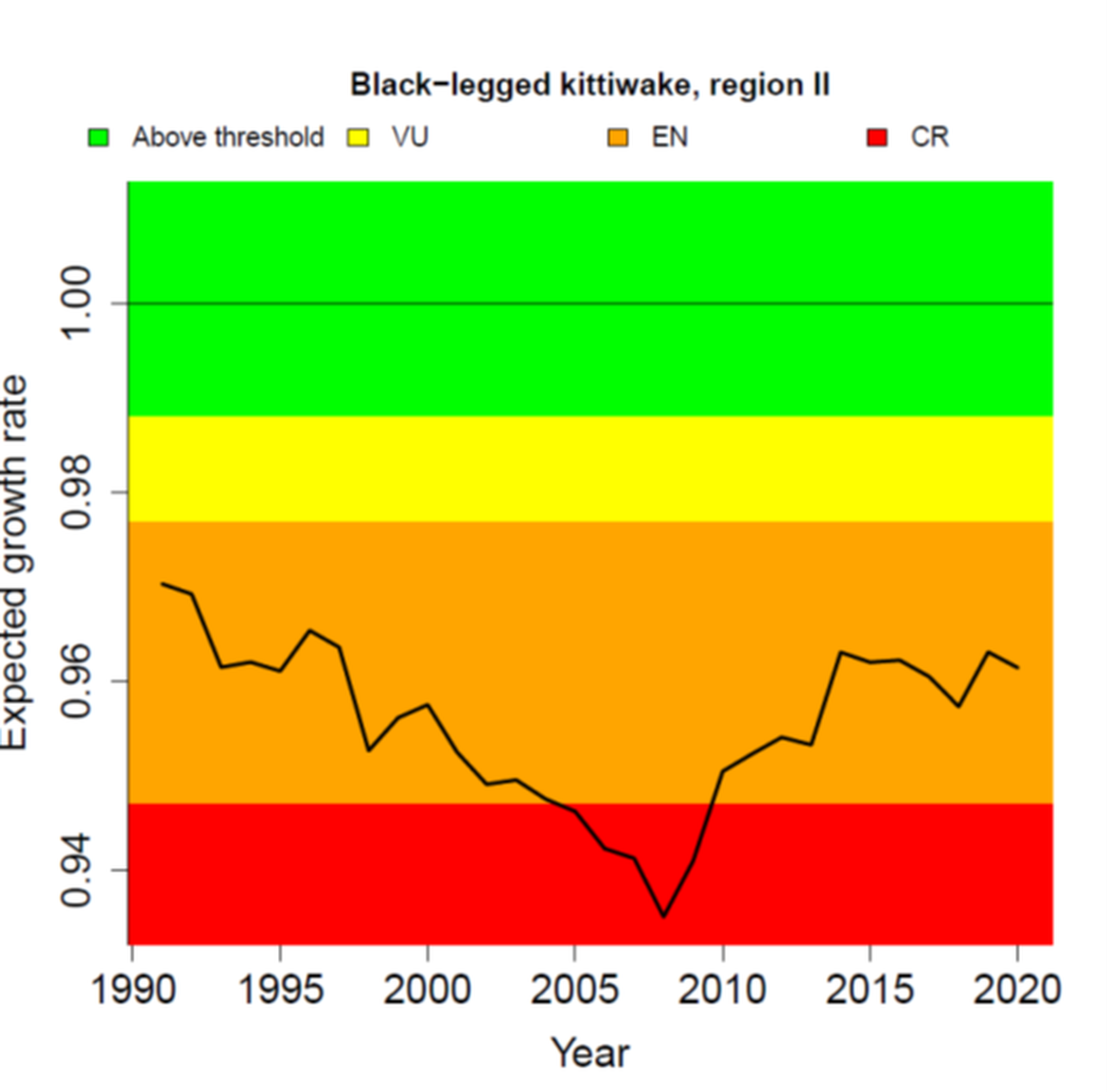
Figure 5: Expected annual population growth rate of Black-legged kittiwake in OSPAR Regions I, II and III, 1991-2020 (black line). The colour-coded background shows the OSPAR QSR2023 threshold values; values in the green zone indicate the threshold is achieved, whereas values in the other zones are below and indicate the threshold is not achieved. For illustration, the figure also shows a breakdown for the corresponding IUCN red list categories of Vulnerable (VU), Endangered (EN) and Critically Endangered (CR). Source: OSPAR (2023b).
Method of assessment: 1 Data Driven – Source: OSPAR (2023b)
Threats and Impacts
Threats to the kittiwakes’ food supply of sandeels from climate change appear to be continuing in the Arctic and Greater North Sea (Cook et al. 2014; Mitchell et al. 2020). Threats from sandeel fishing in the North Sea appear to be decreasing due to a combination local closures and reduction in sandeel stocks and fishing effort (Mitchell et al. 2018). However, there is no evidence from the Celtic Seas to suggest climate change is driving reductions in food supply in the same way (Cook et al. 2014, Lauria et al. 2012). The southern edge of the breeding range in the Celtic Seas is constricting northwards, which is consistent with climate change predictions of Russell et al. (2015).
In Arctic Waters, some large kittwiake colonies in the Norwegian Sea have gone extinct since the last assessment due to a combination of poor food supply and increasing predation on chicks from White-tailed eagles (Haliaeetus albicilla) (Anker-Nilssen et al. in review). Post-larval herring from the Norwegian spring-spawning (NNS) stock that collapsed in the late 1960s, has proven to be a key driver of kittiwake breeding success in this part of Region I (Cury et al. 2011). The current low availbility of sandeels is more likely to be driven by competition for food with other larger fish and predation by larger fish than by climate change-induced temperature changes (Frederiksen et al. 2013, but see Hátún et al., 2017).
In the Bay of Biscay and Iberian Coast, a collapse in food supply (sardines) in 1991 caused the decline in Spain (Martínez-Abraín, 2019). The declines eslewhere in the Region may be part of northward shift in range, consistent with climate change predictions of Russell et al. (2015).
An emerging threat to kittiwakes may come from the expansion of offshore renewable energy generation in some areas, particularly in the North Sea.
Measures that address key pressures from human activities or conserve the species/habitat
In 2019, all but one Contracting Party within the breeding range of kittiwake (Ireland) reported on the implementation of national actions under OSPAR Recommendation 2011/5, as amended by OSPAR Recommendation 2020/1 (OSPAR, 2011). Most actions had been implemented by those Contracting Parties who reported, but in some cases it was difficult to assess effectiveness of the measure without more detailed standards (e.g. for monitoring).
Most Contracting Parties have legislation that bans deliberate killing or taking of chicks and eggs. However, regulated hunting of Black-legged kittiwake is permitted by Iceland and by Denmark with respect to Greenland.
OSPAR’s most recent MPA assessment (OSPAR 2022) concluded that kittiwake are sufficiently represented in MPAs accross their range (i.e. they are a feature of more than one MPA in each Region where they are declining or threatened). Furthermore, kittiwakes are included as a feature of the North Atlantic Current and Evlanov Sea basin MPA (NACES), which covers 600 000km2 of ocean in the western part of Region V and was recently nominated collectively (https://www.ospar.org/documents?v=43885).
However, protection measures in existing MPAs and measures to determine and address levels of bycatch appear to be under-implemented, according to the 2019 reporting (Table 2) and may need further attention.
Table 2: Summary of Contracting Party 2019 reports on implementation of national actions relating to Black-legged kittiwake (Recommendation 2011/5, OSPAR, 2011)
| National Action | Implementation | ||
| Full | Partial | None2 | |
| National legislation to protect the black-legged kittiwake | BE, DK, FR, DE, NL, NO, SE, UK | IS, DK(GL)1 | |
| Relevant conservation measures in key areas | DK, DE, IS, NO, SE, UK | FR, NL, DK(GL) | BE |
| Marine Protected Areas in OSPAR Network with management plans | FR, SE, UK | BE, DK, DK(GL), DE, IS, NL, NO | |
| Monitoring and assessment programmes | DK(GL), FR, IS, NO, SE, UK | NL, BE, DE, | DK |
| Raise awareness of the status of kittiwakes and threats | DK(GL), IS, NO, SE, UK | DE | BE, DK, FR, NL |
| Seabird bycatch National Plan of Action (NPOA) for longline fisheries | IS, (NO) | UK, NO | BE, DK, FR, DE, NL, SE |
| Support, promote and implement the priority actions identified in the Nordic Action Plan for Seabirds. | NO | BE, DK, FR, DE, NL, IS, SE, UK | |
1DK(GL) = DK Reporting on behalf of Greenland
2Grey text = action not applicable; Bold text = future plans for implementation
Conclusion (including management considerations)
Results from the QSR 2023 (OSPAR 2023a, b) show that the status of breeding populations of kittiwakes is poor in all four Regions where it breeds, and has declined further since the original assessment in 2008 and 09 (see OSPAR 2008, 2009).
Breeding numbers in all four Regions declined steadily during the 1990s and early 2000s. In Arctic Norway, which held the largest breeding population, numbers have declined by 88% since 1991. Declines of 64% and 58% also occured in the Greater North Sea and Celtic Seas, respectively. The declines occured during periods of poor productivity. In the last decade, productivity in all three Regions where they still breed has improved somewhat, and the declines in breeding numbers have slowed down or stopped since 2010, except in Arctic Waters (Norwegian part), where declines have continued. Despite the improvement, productivity remains at levels that are insufficient to stimulate population recovery.
Low productivity has most probably been caused by reduction in the kittiwakes’ food supply of small shoaling fish (e.g. sandeel, sprat, herring). Climate change appears to be the key driver of these changes in their prey-fish populations in the Arctic and Greater North Sea, but it is unclear what drivers are operating in the Celtic Seas. In the Bay of Biscay, few kittiwakes are left breeding, partly due to a collapse in the sardine stock in the south of the region in the 1990s. The declines elsewhere in the Region may be part of a northward contraction in range, which is also evident in the southern part of the Celtic Seas and is consistent with climate change predictions.
Black-legged kittiwakes are therefore still justified for inclusion in the OSPAR List of Threatened and Declining Species.
In view of the impacts of climate change on kittiwake populations, action under OSPAR Recommendation 2011/5, as amended by OSPAR Recommendation 2020/1, could focus on reducing other impacts as much as possible. This will enhance the resilience of kittiwake populations to the impacts of climate change, in parallel with global efforts to reduce carbon emissions. This may include adjusting existing actions such as the regulation of sandeel fishing in the North Sea, work to improve ecosystem-based management of key fisheries, regulation of hunting in the Arctic, management of MPAs and assessing risk from bycatch in fisheries.
Additional action may be required to monitor and address impacts from emerging threats. These threats include the loss of foraging habitat and increased mortality risk (i.e. collision with wind turbines) by offshore renewable energy generation in the North Sea and the spread of avian influenza following recent outbreaks at seabird colonies in the OSPAR Area.
In the future, kittiwakes can be re-assessed when the OSPAR Common Indicators for marine birds are updated, i.e. during future QSR or Intermediate Assessments.
Knowledge Gaps
The assessment for breeding populations of kittiwakes in Arctic Waters lacks data from Greenland, Iceland and the Faroes. The assessment in the Celtic Seas lacks data from Ireland.
This assessment was based on breeding populations of kittiwake. The QSR 2023 included a pilot assessment of their status at sea outside the breeding season (OSPAR 2023c), but this is limited to the southern part of the Greater North Sea.
The assessment of threats would also benefit from a greater understanding of the links between climate change, lower trophic levels and kittiwake population status, predation risks and movements.
Method used
Main source of information:
1. OSPAR data assessment
Assessment is based upon:
a) complete survey or a statistically robust estimate: The ability to assess kittiwake status in the Arctic Waters, Celtic Seas and Greater North Sea was greatly improved in this assessment, compared to the background document (OSPAR 2009), by the inclusion of common indicators from the QSR2023 (OSPAR 2023a, b, c,
d) These were constructed from a systematic collation of seabird data from across the OSPAR maritime area.
Anker-Nilssen, T., Fayet, A.L. & Aarvak, T. in review. Top-down control of endangered seabirds: native White-tailed eagles accelerated the extinction of a large kittiwake colony.
Anker-Nilssen, T., Hanssen, S.A., Moe, B., Systad, G.H.R., Barrett, R., Bustnes, J.O., Christensen-Dalsgaard, S., Dehnhard, N., Descamps, S., Erikstad, K.E., Follestad, A., Langset, M., Layton-Matthews, K., Lorentsen, S.H., Lorentzen, E., Reiertsen, T.K., Strøm, H. 2022. Key-site monitoring in Norway 2021, including Svalbard and Jan Mayen. SEAPOP Short Report 1-2022, Norwegian Institute for Nature Research and Norwegian Polar Institute, Trondheim/Tromsø, 15 pp. https://seapop.no/wp-content/uploads/2022/10/seapop-short-report-1-2022.pdf
Bærum, K.M., Anker-Nilssen, T., Christensen-Dalsgaard, S., Fangel, K., Williams, T. & Vølstad, J.H. 2019. Spatial and temporal variations in seabird bycatch: incidental bycatch in the Norwegian coastal gillnet-fishery. PLoS ONE 14(3): e0212786
Busch, M., Garthe, S., 2018. Looking at the bigger picture: the importance of considering annual cycles in impact assessments illustrated in a migratory seabird species. ICES (Int. Counc. Explor. Sea) J. Mar. Sci. https://doi.org/10.1093/icesjms/fsx170.
Cabrero A, González-Nuevo G, Gago J, Cabanas JM (2019). Study of sardine (Sardina pilchardus) regime shifts in the Iberian Atlantic waters. Fish Oceanogr 28: 305−316Carroll, M. J., Butler, A., Owen, E., Ewing, S. R., Cole, T. J., Green, A., Soanes, L. M., Arnould, J. P. Y., Newton, S. F., Baer, J., Daunt, F., Wanless, S., Newell, M. A., Robertson, G. S., Mavor, R. A. & Bolton, M. 2015. Effects of sea temperature and stratification changes on seabird breeding success. Clim. Res. 66: 75–89.
Carroll, M. J., Bolton, M., Owen, E., Anderson, G. Q. A., Mackley, E. K., Dunn, E. K. & Furness, R. W. 2017. Kittiwake breeding success in the southern North Sea correlates with prior sandeel fishing mortality. Aquatic Conserv: Mar Freshw Ecosyst. 2017: 1–12.
Cook A.S. C. P., Dadam, D., Mitchell, I., Ross-Smith, V. H. and Robinson, R. A.. 2014. Indicators of seabird reproductive performance demonstrate the impact of commercial fisheries on seabird populations in the North Sea. Ecological Indicators 38: 1–11.
Cury, P.M., Boyd, I.L., Bonhommeau, S., Anker-Nilssen, T., Crawford, R.J.M, Furness, R.W., Mills, J.A., Murphy, E.J., Österblom, H., Paleczny, M., Piatt, P.F., Roux, J.-P., Shannon, L. & Sydeman, W.J. 2011. Global seabird response to forage fish depletion – one-third for the birds. Science 334: 1703-1706.
Christensen-Dalsgaard, S., Langset, M. & Anker-Nilssen, T. 2019. Offshore oil rigs – a breeding refuge for Norwegian Black-legged Kittiwakes? Seabird 32: 20-32.
EEA 2020. Breeding population and distribution trends. European Environment Agency. https://www.eea.europa.eu/themes/biodiversity/state-of-nature-in-the-eu/article-12-national-summary-dashboards/breeding-population-and-distribution-trends
Frederiksen M., Wanless S., Harris M.P., Rothery P., Wilson L.J. (2004). The role of industrial fisheries and oceanographic change in the decline of North Sea black-legged kittiwakes. J. Appl. Ecol. 41: 1129–1139
Frederiksen M., Wright P.J., Harris M.P., Mavor R.A., Heubeck M., Wanless S. 2005. Regional patterns of kittiwake Rissa tridactyla breeding success are related to variability in sandeel recruitment. Mar. Ecol. Prog. Ser. 300: 201–211
Frederiksen, M., Mavor, R.A. & Wanless, S. 2007. Seabirds as environmental indicators: the advantages of combining data sets. Mar. Ecol. Prog. Ser. 352: 205–211.
Frederiksen, M., Jensen, H., Daunt, F., Mavor, R.A. & Wanless, S. 2008. Differential effects of a local industrial sand lance fishery on seabird breeding performance. Ecol. Appl. 18: 701–710.
Frederiksen, M., B. Moe, F. Daunt, R.A. Phillips, R.T. Barrett, M.I. Bogdanova, T. Boulinier, J.W. Chardine, O. Chastel, L.S. Chivers, S. Christensen-Dalsgaard, C. Clément-Chastel, K. Colhoun, R. Freeman, A.J. Gaston, J. González-Solís, A. Goutte, D. Grémillet, T. Guilford, G.H. Jensen, Y. Krasnov, S.-H. Lorentsen, M.L. Mallory, M. Newell, B. Olsen, D. Shaw, H. Steen, H. Strøm, G.H. Systad, T.L. Thórarinsson & T. Anker-Nilssen (2012): Multicolony tracking reveals the winter distribution of a pelagic seabird on an ocean basin scale. Diversity Distributions 18: 530-542.
Frederiksen, M., Anker-Nilssen, T., Beaugrand, G. & Wanless, S. (2013). Climate, copepods and seabirds in the boreal Northeast Atlantic – current state and future outlook. Global Change Biology (2013) 19, 364–372.
Furness, R. W. 1997. The impact of predation by great skuas on other species with particular reference to Special Protection Areas in Shetland. Report to Scottish Natural Heritage.
Furness R.W. & Tasker M.L. 2000. Seabird-fishery interactions: quantifying the sensitivity of seabirds to reductions in sandeel abundance, and identification of key areas for sensitive seabirds in the North Sea. Mar. Ecol. Prog. Ser. 202: 253–264.
García-Barón, I., Santos, M.B., Uriarte, A., Inchausti, J.I., Escribano, J.M., Albisu, J., Fayos, M., Pis-Millán, J.A., Oleaga, Á., Alonso Mier, F.E., Hernández, O., Moreno, O.; Louzao, M. 2019. Which are the main threats affecting the marine megafauna in the Bay of Biscay? Continental Shelf Research, 186: 1-12
Hamer, K. C., Monaghan, P., Uttley, J. D., Walton, P. & Burns, M. D. 1993. The influence of food supply on the breeding ecology of Kittiwakes Rissa tridactyla in Shetland. Ibis 135: 255-263.
Hátún, H., Azetsu-Scott, K., Somavilla, R., Rey, F., Johnson, C., Mathis, M., Mikolajewicz, U., Coupel, P., Tremblay, J-É., Hartman, S., Pacariz, S.V., Salter, I. & Ólafsson, J. 2017. The subpolar gyre regulates silicate concentrations in the North Atlantic. Scientific Reports 7: 14576
Heubeck, M., Mellor, R.M., Harvey, P.V., Mainwood, A.R. & Riddington, R. 1999. Estimating the population size and rate of decline of Kittiwakes Rissa tridactyla breeding in Shetland, 1981-97. Bird Study 46: 48-61.
ICES 2017. Sandeel (Ammodytes spp.) in Division 4.a, Sandeel Area 7r (northern North Sea, Shetland). ICES Advice on fishing opportunities, catch, and effort Greater North Sea Ecoregion (san.sa.7r). Available at: http://ices.dk/sites/pub/Publication%20Reports/Advice/2017/2017/san.sa.7r.pdf
ICES. 2015. Report of the Joint ICES/OSPAR Working Group on Seabirds (JWG-BIRD), 17–21 November 2014, Copenhagen, Denmark. ICES CM 2014/ACOM:30. 115 pp.
JNCC. 2016. Seabird Population Trends and Causes of Change: 1986-2015 Report (http://jncc.defra.gov.uk/page-3201). Joint Nature Conservation Committee. Updated September 2016.
Kelsey, E.C.; Felis, J.J.; Czapanskiy, M.; Pereksta, D.M.; Adams, J. 2018. Collision and displacement vulnerability to offshore wind energy infrastructure among marine birds of the Pacific Outer Continental Shelf. Ibis, 148: 90-109
Lauria V., Attrill M.J., Pinnegar J.K., Brown A., Edwards M. & Votier S.C. 2012. Influence of climate change and trophic coupling across four trophic levels in the Celtic Sea. PLoS ONE 7(10): e47408.
Legroux, N., 2019. Bilan de la saison de reproduction de la Mouette tridactyle sur les colonies témoins des sous-régions marines Manche/mer du Nord et mers Celtiques. Année 2019. 12p.
Martínez-Abraín, A., Santidrián Tomillo, P., Mouriño, J., Tenan, S. & Oro, D. 2019. Delayed predator−prey collapses: the case of black-legged kittiwakes and Iberian sardines. Mar Ecol Prog 631: 201–207.
McMurdo-Hamilton, T., Brown, A. & Lock, L. 2016. Kittiwake declines extend to southern England and beyond: an update on colonies at the southern edge of the species’ Northeast Atlantic range. British Birds 109: 199–210.
Miles, J., Parsons, M. and O’Brien, S. 2020. Preliminary assessment of seabird population response to potential bycatch mitigation in the UK-registered fishing fleet. Report prepared for the Department for Environment Food and Rural Affairs (Project Code ME6024).
Mitchell, P. I., Newton, S. F., Ratcliffe, N. and Dunn, T. E. (Eds.). 2004. Seabird Populations of Britain and Ireland: results of the Seabird 2000 census (1998-2002). Published by T and A.D. Poyser, London.
Mitchell, I., Cook, A., Douse, A., Foster, S., Kershaw, M., McCulloch, N., Murphy, M. & Hawkridge, J. 2018. Kittiwake breeding success. UK Marine Online Assessment Tool. Available at: https://moat.cefas.co.uk/biodiversity-food-webs-and-marine-protected-areas/birds/kittiwake-breeding-success/
Mitchell, I., Daunt, F., Frederiksen, M. and Wade, K. (2020) Impacts of climate change on seabirds, relevant to the coastal and marine environment around the UK. MCCIP Science Review 2020, 382–399
Monaghan, P., Uttley, J. D. & Okill, J. D. 1989. Terns and sandeels: seabirds as indicators of changes in marine fish populations. J. Fish Biol. 35, 339–40.
Northridge, S., Kingston, A., and Coram, A. 2020. Preliminary estimates of seabird bycatch by UK vessels in UK and adjacent waters. Report prepared for the Department for Environment Food and Rural Affairs (Project Code ME6024).
OSPAR (2009). Background Document for Black-legged kittiwakes Rissa tridactyla tridactyla. https://www.ospar.org/work-areas/bdc/species-habitats/list-of-threatened-declining-species-habitats
OSPAR (2011). OSPAR Recommendation 2011/5 on furthering the protection and conservation of the Black-legged kittiwake (Rissa tridactyla tridactyla). https://www.ospar.org/work-areas/bdc/species-habitats/list-of-threatened-declining-species-habitats
OSPAR (2022). Report and assessment of the status of the OSPAR network of Marine Protected Areas in 2021. Available at: https://oap.ospar.org/en/ospar-assessments/committee-assessments/biodiversity-committee/status-ospar-network-marine-protected-areas/assessment-reports-mpa/mpa-2021/
OSPAR (2023a). Marine Bird Abundance [Online]. London, UK: OSPAR. Available at: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/indicator-assessments/marine-bird-abundance/
OSPAR (2023b). Marine Bird Breeding Productivity [Online]. London, UK: OSPAR. Available at: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/indicator-assessments/marine-bird-breeding-productivity/
OSPAR (2023c). Pilot Assessment of Marine Bird Abundance - Non-Breeding Offshore Birds [Online]. London, UK: OSPAR. Available at: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/indicator-assessments/bird-abundance-pilot/
OSPAR (2023d). Pilot Assessment of Marine Bird Habitat Quality. [Online]. London, UK: OSPAR. Available at: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/indicator-assessments/marine-bird-habitat-quality-pilot/.
Peschko, V., B. Mendel, S. Müller, N. Markones, M. Mercker & S. Garthe (2020): Effects of offshore windfarms on seabird abundance: Strong effects in spring and in the breeding season. Mar. Environ. Res. 162: 105157.
Russell, D.J.F., Wanless, S., Collingham, Y.C. and Hamer, K.C. (2015) Predicting future European breeding distributions of British seabird species under climate change and unlimited/no dispersal scenarios. Diversity 7, 342-359
Searle, K.R., Mobbs, D.C., Butler, A., Furness, R.W., Trinder, M.N. and Daunt, F. 2018. Finding out the fate of displaced birds. Scottish Marine and Freshwater Science, 9(8): 2043-7722
SEAPOP (2017). http://www.seapop.no/opencms/export/sites/SEAPOP/no/filer/pdf/arsbrosjyrer/SEAPOP-arsbrosjyre-2017-web.pdf_511166643.pdf).
Simian G., Artero C., Cadiou B., Authier M., Bon C. & Caillot E. 2018. Évaluation de l’état écologique des oiseaux marins en France métropolitaine. Rapport scientifique pour l’évaluation 2018 au titre de la DCSMM. Muséum National d’Histoire Naturelle, Service des stations marines de Dinard, 161 p.
Votier, S. C., Furness, R. W., Bearhop, S., Crane, J. E., Caldow, R. W. G., Catry, P., Ensor, K., Hamer, K. C., Hudson, A. V., Kalmbach, E., Klomp, N. I., Pfeiffer, S., Phillips, R. A., Prieto, I. & Thompson, D. R. 2004. Changes in fisheries discard rates and seabird communities. Nature 427: 727-730.
Wright, P. J. & Bailey, M. C. 1993. Biology of sandeels in the vicinity of seabird colonies at Shetland. Fisheries Research Report No. 15/93. SOAFD Marine Laboratory, Aberdeen
1In case of a significant trend over the first ten years of the time series (regression p value ≤ 0,05), the abundance in the first year (1991) was predicted from the trend regression and used as the baseline value. If no significant trend existed in the first ten years, the mean abundance of the first ten years (ignoring missing years) was used as the baseline value (OSPAR 2023a).
Sheet reference:
BDC2023/black_legged_kittiwake