Status Assessment 2020 - Thick-billed murre or Brünnich‘s guillemot
| Assessment of status | Non-breeding distribution | Non-breeding Population size | Breeding distribution | Breeding Population size | Condition i.e. breeding productivity and adult survival | Previous OSPAR status assessment | Status | |
|---|---|---|---|---|---|---|---|---|
Region | I | ? | ? | ↔1,3,5 | ↓1,3,5 | ↔ | ● | Poor |
| II | NA | |||||||
| III | NA | |||||||
| IV | NA | |||||||
| V | NA | |||||||
| Assessment of key pressures | Climate change and indirect consequences (e.g., limited prey availability) | Hunting | Predation/ disturbance | Chemical pollution, i.e., mercury | Oil pollution | Threat or impact | |
|---|---|---|---|---|---|---|---|
Region | I | ↑1,3,5 | ↓3,5 | ? | ↑3,5 | ↓3,5 | |
| II | NA | ||||||
| III | NA | ||||||
| IV | NA | ||||||
| V | NA | ||||||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: If in QSR 2010 then enter regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ). If a more recent status assessment is available, then enter ‘poor’/’good’
Status* (overall assessment)
red – poor
green – good
blue cells –? status unknown, insufficient information available,
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key pressures
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threat or impact (overall assessment)
red – significant threat or impact;
green – no evidence of a significant threat or impact
blue cells – ? insufficient information available
NA – not applicable
1 – direct data driven,
2 – indirect data driven,
3 – third party assessment close-geographic match,
4 - third party assessment partial-geographic match
5 – expert judgement.
Confidence
There is high confidence in the status assessment because of the quantitative and current data used. But medium confidence in the threat assessment because determining future impacts of climate change and its consequences on BRGU populations (which is considered to be the main threat) are challenging, particularly during the non-breeding season when the birds are distributed over large offshore areas.
Background Information
Year added to OSPAR List: OSPAR (2008). The original evaluation of BRGUs referred to regional importance, decline and sensitivity criteria and it provided information on threats see https://www.ospar.org/site/assets/files/1887/thick_billed_murre.pdf.
Regional importance: BRGUs were listed because they are restricted to a relatively small number of populations within OSPAR Region I.
Decline: At the time of listing in 2008, BRGU populations in Svalbard were evaluated as broadly stable while breeding populations in Iceland and Greenland had decreased between 1990-2000.
Sensitivity: BRGUs were classified as sensitive because they have a low resistance to anthropogenic threats. They are long-lived and have a slow life-history and thus are likely to recover slowly if populations are reduced.
Anthropogenic and biological pressure: The key threats at the time of listing included hunting during the winter season in Greenland, disturbance from hunting activity, egg-harvesting, incidental kills in fishing gear, competition with commercial fisheries, chronic oil pollution and potentially climate change.
Last status assessment and brief outcome: OSPAR (2009) concluded that BRGUs still qualified under the OSPAR criteria of regional importance and decline. Except for some hunting regulations, there were no actions implemented to address threats BRGUs were facing. At the time of the last assessment, there was a concern of further population declines in Iceland and Greenland, which has been corroborated for the latter location in the current assessment. In addition, a population decline has also been observed in Norway (incl. Svalbard and Jan Mayen) since the last assessment (see population and abundance section below).
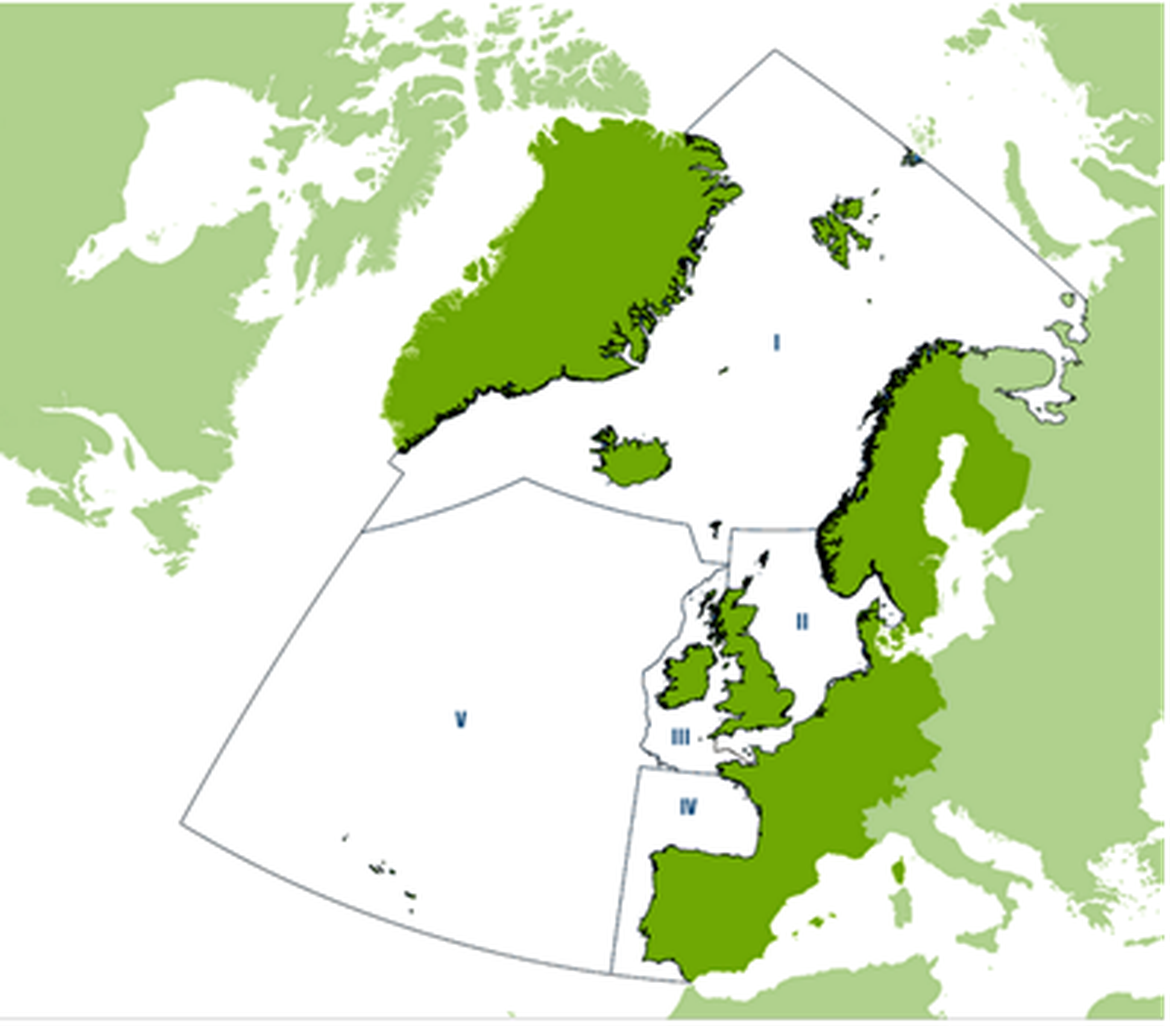
Figure 1: OSPAR Regions. BRGU are breeding only within Region I (Arctic waters). Copyright © OSPAR Commission
Geographical Range and Distribution
Within the Northeast Atlantic, the species breeds in Norway (incl. Svalbard and Jan Mayen), Russia, Greenland and Iceland (OSPAR region 1, Figures 1-2, Bakken and Pokrovskaya, 2000).Most of the population breeds in the Arctic at high latitudes (>60 °N).
Recent tracking studies indicate that birds from breeding populations in the western sector of the Northeast Atlantic (Iceland, East Greenland, Jan Mayen, and West Svalbard) use primarily the waters off Southwest Greenland, as well as Iceland (Bjørnøya birds only) as wintering grounds (Frederiksen et al., 2016; Linnebjerg et al., 2018; Seatrack, 2020c), while individuals from the eastern sector of the Northeast Atlantic (northern Norway, East Svalbard and Russian populations) spend the non-breeding season in the Barents and Kara Seas (Seatrack, 2020a) (see Figures 3 and 4).
In addition, a sex-specific migration has been observed in some populations resulting in spatial separation during at least some of the non-breeding season. In West Greenland, male birds from Baffin Bay occur further north than females and started migrating later than females from the same colony (Frederiksen et al., 2016).
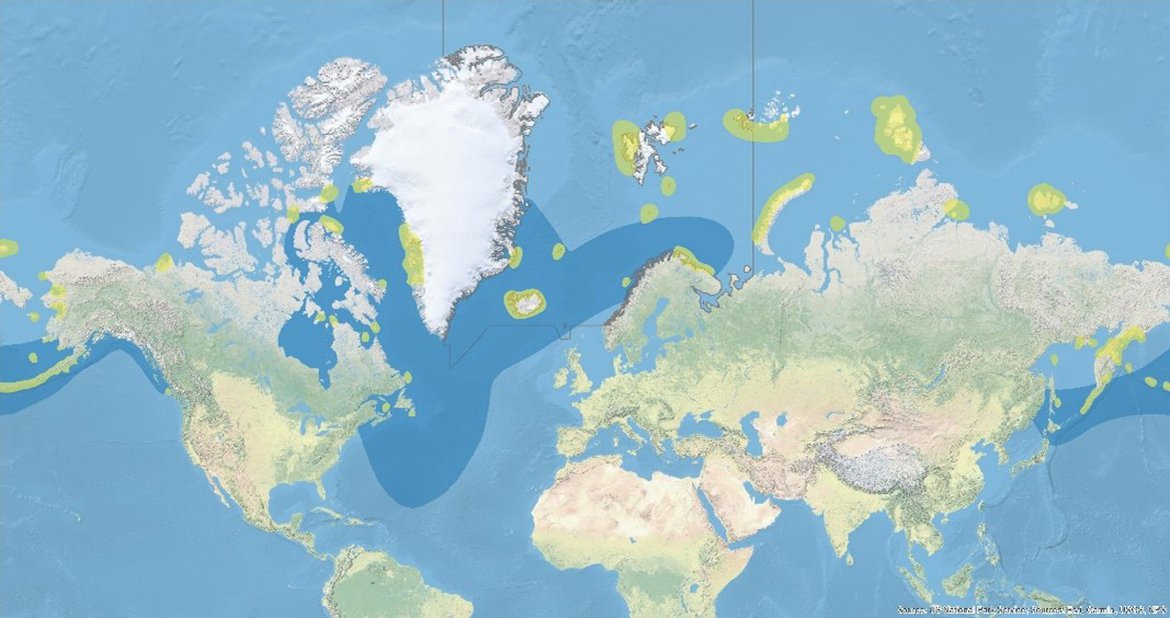
Figure 2: Global range of Brünnich‘s guillemots (map: BirdLife International, 2020). The species has a circumpolar breeding distribution in sub-Arctic and Arctic regions. Breeding areas are highlighted in yellow and the non-breeding distribution of BRGUs is indicated in blue. The grey line outlines OSPAR Region I.
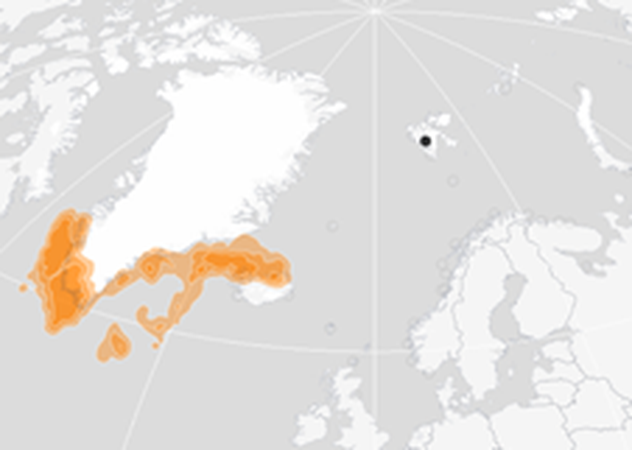
Figure 3: Winter (November – January) distribution of BRGUs from the Isfjorden colony in West Spitsbergen (indicated by black dot) 2013-2019 (Seatrack, 2020c).
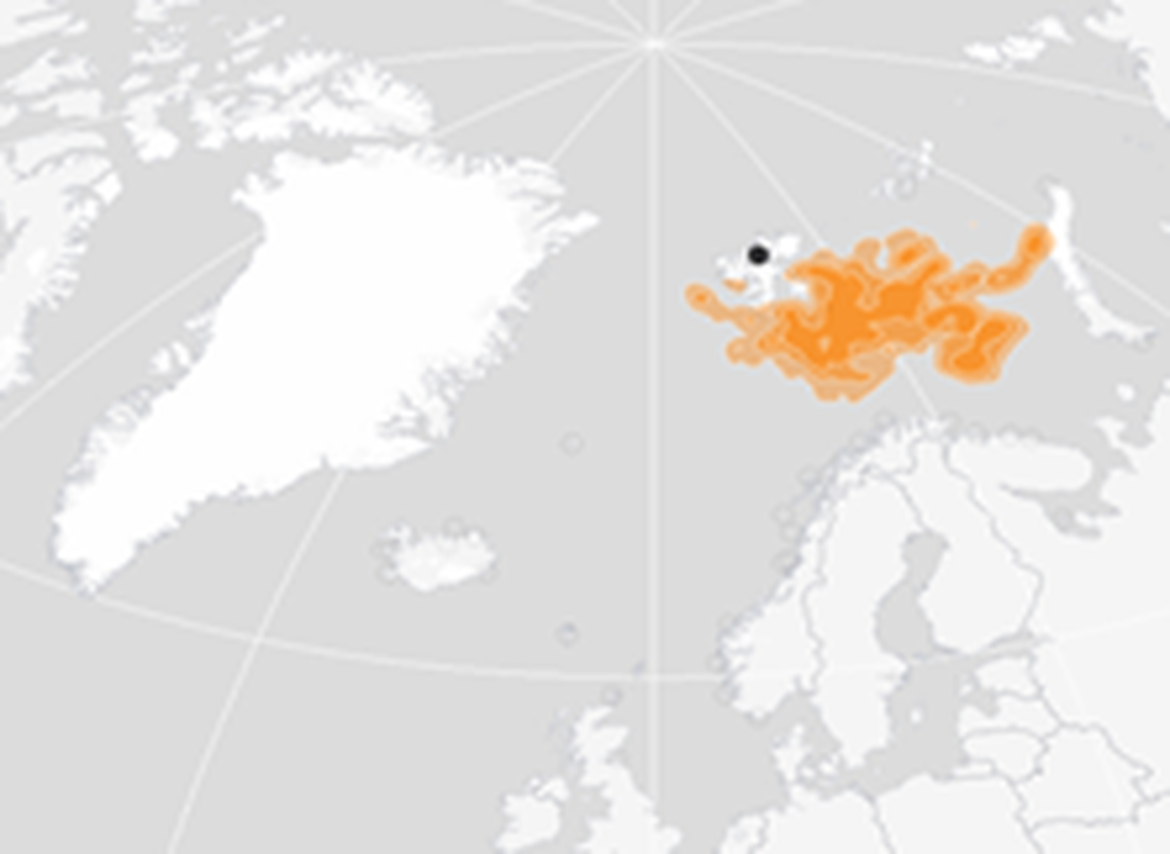
Figure 4: Winter (November – January) distribution of BRGUs from the Alkefjellet colony in East Spitsbergen (indicated as black dot) 2015-2019 (Seatrack, 2020a).
Population/Abundance
BRGU populations in East Greenland Jan Mayen, northern Norway, West Spitsbergen, Bjørnøya, and along the coast of the Russian mainland (Murman) are decreasing (Descamps et al., 2013; Systad, G., personal communication; Merkel et al., 2014; Ezhov, 2019; Boertmann et al., 2020), with some populations facing potential extinction (Descamps et al., 2013; Systad, G., personal communication; Table 1). Population trends of monitored colonies in West Svalbard (incl. Bjørnøya and Jan Mayen, Figure 5), East Greenland (Figure 6), and along the Murman coast (Figure 7) are provided.
However, some populations in the eastern North Atlantic display a different trend. The colony at Alkefjellet in East Spitsbergen has been stable since 2015 (when the monitoring started; unpublished data, Descamps, S., NPI; Table 1; Figure 8) and a positive population trend has been documented in Franz Josef Land (Russia) (Krasnov and Gavrilo, 2018). The results in the eastern sectors of the Northeast Atlantic are based on short time-series so some caution is needed in their interpretation. In Iceland, breeding populations of BRGUs declined between 1983-1986 and 2006-2008 (Garðarsson et al., 2019) but recent monitoring suggests that populations have been stable since 2009 (Þórarinsson, Þ. L., personal communication).
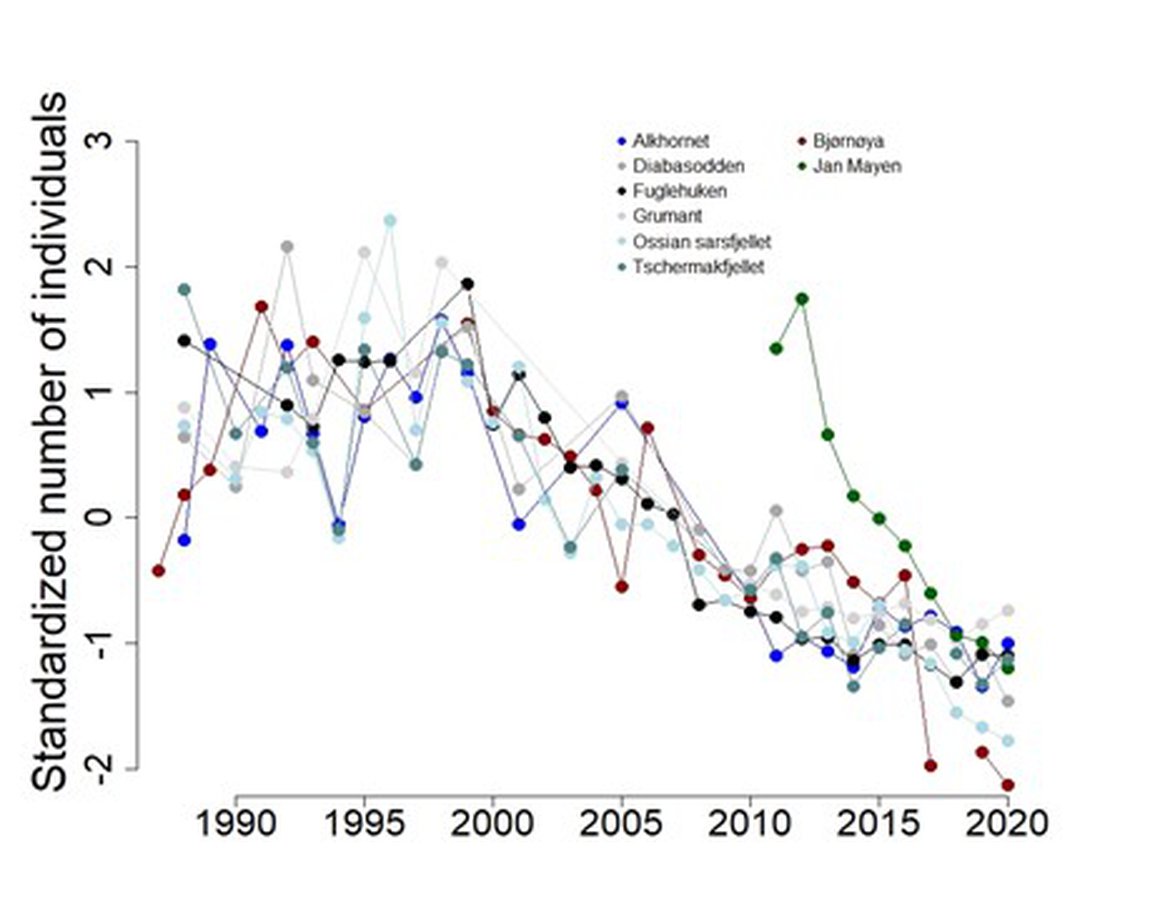
Figure 5: Population trends of BRGUs in monitored colonies in West Spitsbergen, Bjørnøya and Jan Mayen between 1987 – 2020. Data are standardized (deviation from mean divided by standard deviation). More details regarding BRGU breeding populations in Svalbard can be found on the Environmental Monitoring of Svalbard and Jan Mayen (MOSJ) (2020a). The numbers represent the total number of individuals in study plots during incubation/early chick-rearing.
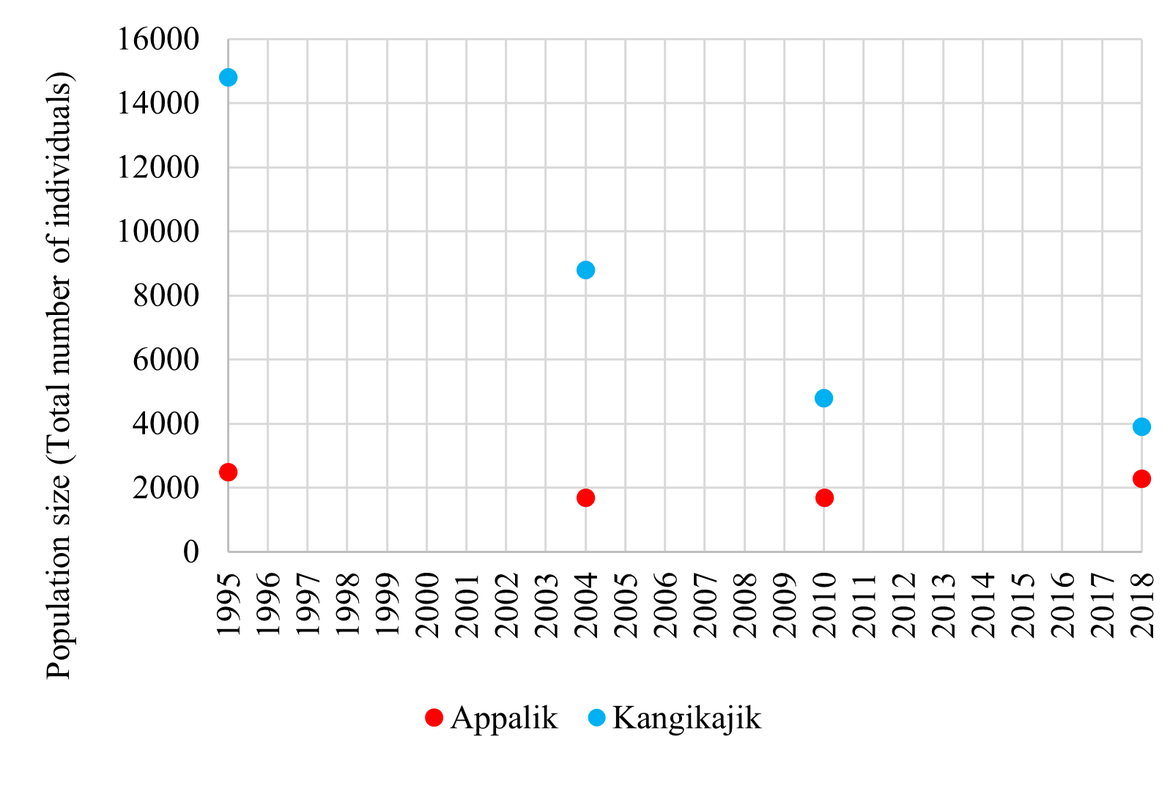
Figure 6: Population trends of BRGUs in East Greenland colonies (Appalik and Kangikajiik) 1995 – 2018. Population estimates for 1995, 2004, and 2010 were provided by Merkel, F. and reported as the number of individuals. For 2018, the number of breeding pairs was reported in Boertmann et al. (2020) and was converted into the number of individuals for comparison using a converting factor of 0.7 (number of pairs = 0.7 * number of individuals). The counts have not been adjusted for the potential diurnal variation in colony attendance and direct comparison of these counts should be done with caution (Merkel, F., personal communication)
| Location | Trend | Description |
|---|---|---|
| West Spitsbergen, Bjørnøya, Jan Mayen | ↓ | All monitored populations: Between the late 1990s until 2020, populations have been significantly and synchronously declining (2-5 % p.a.) and populations might become extirpated in the next 40-50 years (Descamps et al., 2013; unpublished data, Descamps, S., NPI) |
| East Spitsbergen | ? | Alkefjellet: The population is likely stable (monitored only between 2015-2020, unpublished data, Descamps, S., NPI). |
| Northern Norway | ↓ | Hjelmsøya: Between 2009-2019, this population was declining at 4% p.a. and may be extinct (some few birds may attend the colony but there has been no successful breeding since the late 1990s) (Anker-Nilssen et al., 2020; Systad, G., personal communication). |
| Hornøya: This population is likely declining but it is not monitored annually (Anker-Nilssen et al., 2020, Reiertsen, T.K., personal communication). | ||
| East Greenland | ↓ | Kangikajik and Appalik: Between 1990-2000, both of these populations were decreasing (7.1% and 2.4% p.a., respectively) (Merkel et al., 2014). However, the last estimates in 2018 suggest a shift to a stable population trend at Appalik (Boertmann et al., 2020). |
| Iceland | ↑ | Between 1983-1986 and 2006-2008,, the populations in Iceland were declining (Garðarsson et al., 2019) but recent monitoring indicates that they have been stable or have increased slightly since 2009 (Þórarinsson, Þ. L., personal communication). |
| Mainland Russia | ↓ ? | Overall: Interannual fluctuations are large and a negative trend is suspected in the most recent years. |
| Gorodetsky: The population decreased sharply after 2000 (Ezhov, 2019). | ||
| Franz-Josef Land | ↑ | Overall: The population trend has been positive between 2012-2017. |
| Rubini Rock: The number of individuals has increased more than 3 fold at Rubini Rock from 3 to 14 thousand individuals compared to counts reported in 1993 (Krasnov and Gavrilo, 2018). |
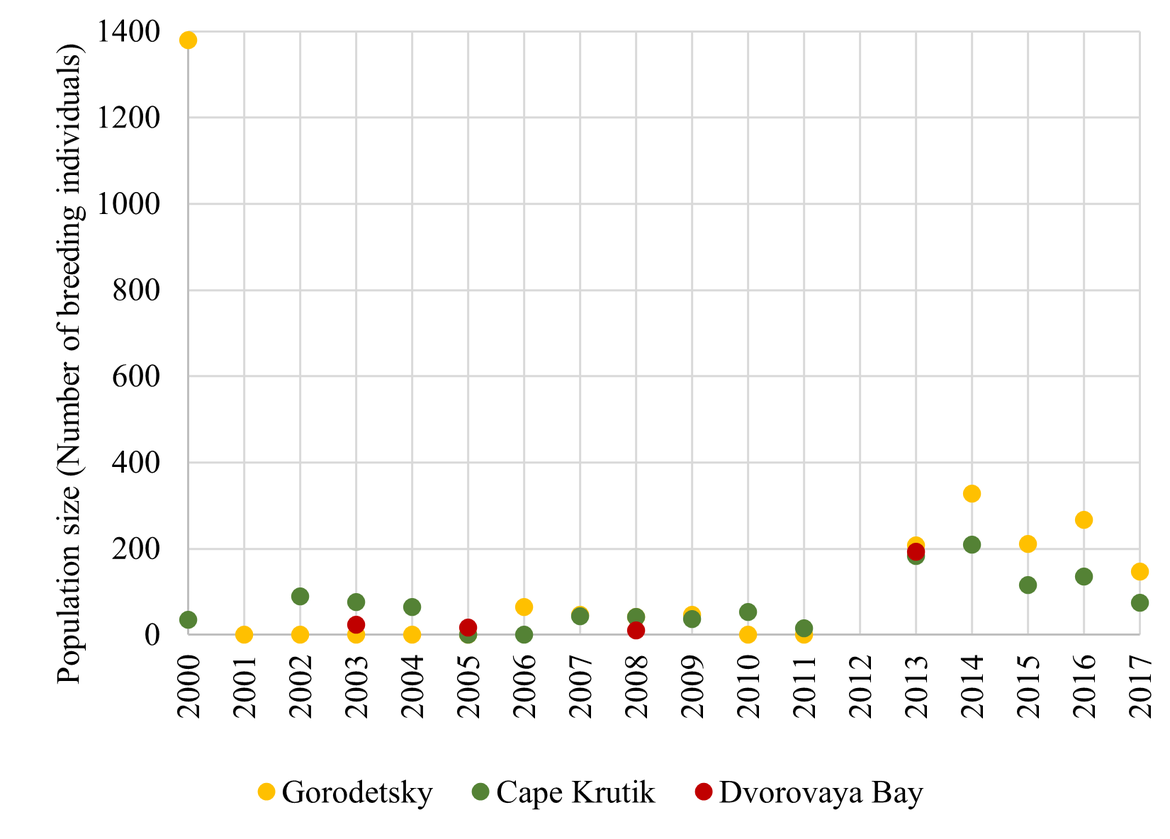
Figure 7: Population trends at the three BRGU colonies in Russia (Gorodetsky,, Cape Krutik and Dvorovaya Bay) along the Murman coast between 2000 – 2017. Numbers represent the total number of individuals in study plots. Population estimates are from Ezhov (2019).
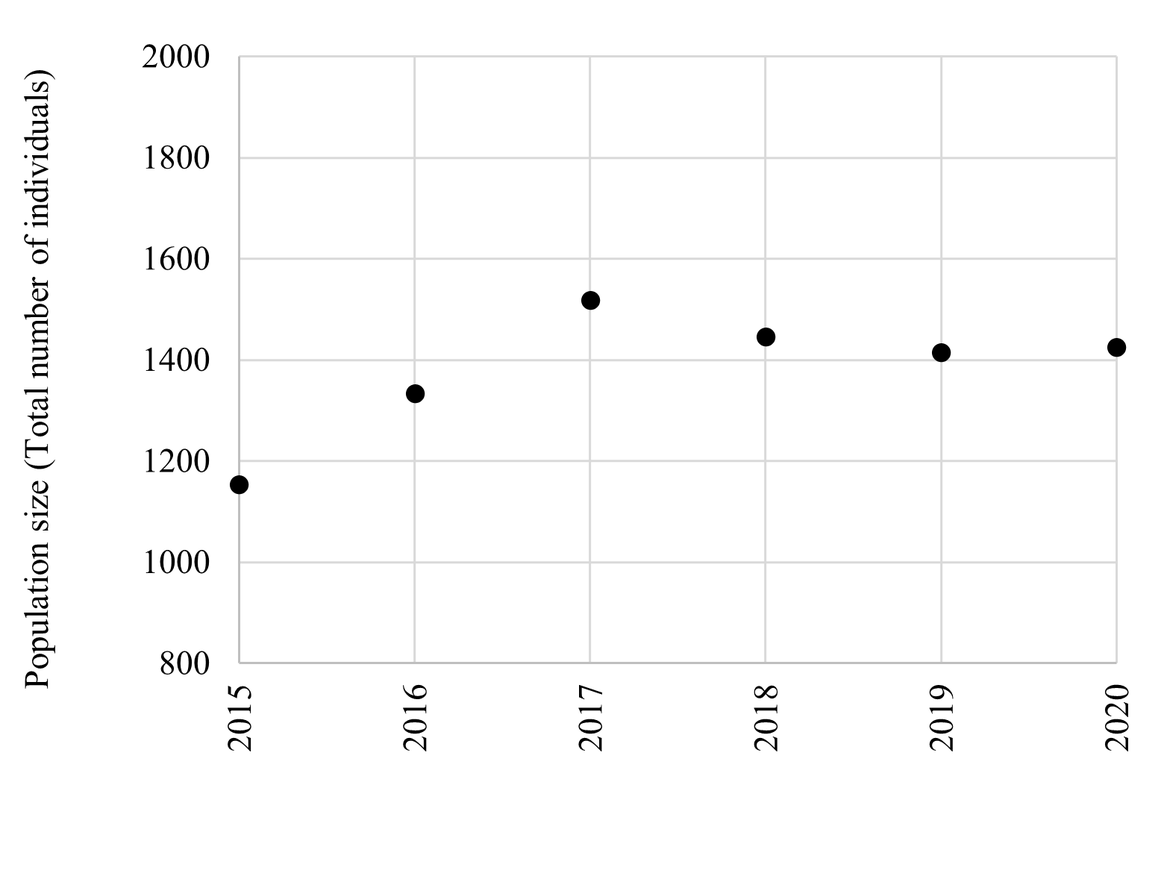
Figure 8: Population trends of BRGUs at the Alkefjellet colony in East Svalbard 2015 – 2020. The numbers represent the total number of individuals in study plots during the incubation period (unpublished data, Descamps, S., NPI).
Condition
Adult survival and breeding success were used to assess condition.
Data is scarce for populations in Greenland and Russia. The breeding success of populations from Svalbard was categorized as good, moderate or poor by the Norwegian SEAPOP monitoring programme (Seapop, 2020; Table 2).
Table 2: Breeding success of BRGUs in Svalbard (West Spitsbergen, Jan Mayen and Bjørnøya) was defined as the % of nests where the chick survived until ca. 15 days of age and was categorized as good (>80% in green), moderate (20-80% in yellow) or poor (<20% in red) by the SEAPOP monitoring programme. Unknown breeding success is shown in grey.
| Colony | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 |
| Spitsbergen | U | U | U | G | M | M | M | M | M | M | M | M | M | M | M | M |
| Jan Mayen | U | U | U | U | U | U | U | U | M | M | M | M | M | M | M | M |
| Bjørnøya | M | M | M | M | M | M | M | M | M | M | M | M | M | M | M | M |
The results indicate that breeding populations from Bjørnøya, Jan Mayen and West Spitsbergen have generally had a moderate breeding success (<80%) since 2011, 2012, and 2009, respectively. No obvious decline in breeding success was observed (Frederiksen et al. in prep). A previous study (Fluhr et al., 2017) found that adult survival rates on Bjørnøya have declined from 1995/1998 up to approx. 2010 by 9-12% . However, survival rates on Bjørnøya have increased in recent years and average survival on Bjørnøya and Spitsbergen since 2010 has been high (Frederiksen et al. in prep).
In Iceland, BRGUs have had a rather high breeding success and high adult survival in recent years (Frederiksen et al. in prep).
These indicators currently used to assess condition (breeding success and adult survival) provide only partial insights regarding BRGU condition. Changes may have occurred in other demographic parameters such as juvenile survival or breeding probability (Frederiksen et al. in prep).
Threats and Impacts
Climate change is likely the main threat to BRGU populations breeding in the Northeast Atlantic (Garðarsson, 2006; Descamps et al., 2013; Frederiksen et al., 2016; Fluhr et al., 2017). A climate-related oceanographic shift in the wintering grounds (associated with warming of the ocean) is thought to have resulted in reduced food supplies and concomitantly reduced survival and recruitment rates (Descamps et al., 2013; Fluhr et al., 2017).
Hunting: BRGUs are hunted in Canada and Greenland, and to a lower extent in Iceland (Frederiksen et al., 2016). The winter harvest only partly explains the observed population declines Frederiksen et al., 2019) The potential importance of spring harvest (allowed in Greenland until 2016) and/or illegal harvest during summer has not been investigated but could have played a role in BRGU decline (Merkel et al. 2014)
Predation/disturbance: Disturbance by predators, such as the white-tailed eagle, has likely contributed to the local population declines in northern Norway(Anker-Nilssen et al., 2020, Reiertsen, T.K. and Systad, G., personal communication). This is likely a marginal and very local phenomenon for the BRGU and such disturbance has not been reported elsewhere.
Chemical pollution: Mercury has been detected in feather samples of BRGUs in the Northeast Atlantic (Albert et al., 2021). Higher mean concentrations were detected during the non-breeding season compared to the breeding season, with one population (Isfjorden) exceeding the Hg threshold for seabirds, which may adversely impact health and reproduction (Eisler, 1987).
Oil Pollution: Oiling mortality, which primarily occurs off Newfoundland where only a small proportion of OSPAR breeding populations winter, has decreased substantially in recent decades (Wilhelm et al., 2009; Robertson et al., 2014) and the current impact of this threat on populations in the Northeast Atlantic region is small (Frederiksen et al., 2019).
Bycatch: In the OSPAR region, the impact of fishing activity on BRGUs is negligible as incidental catches are rare (Merkel, 2011; Pálsson et al., 2015; Skarphéðinsson et al., 2016; Bærum et al., 2019; Christensen-Dalsgaard et al., 2019).
Other potential threats may include ingestion of plastics (Provencher et al., 2010; Bond et al., 2013) and disturbance due to hydrocarbon extraction (McFarlane Tranquilla et al., 2013).
More details are provided in the additional information section below.
Measures that address key pressures from human activities or conserve the species/habitat
National legislation and relevant conservation measures in key areas
Legislation is in place in Norway, Greenland and Iceland to regulate guillemot hunting. BRGU hunting has been forbidden in Norway since 2015. In Greenland, spring hunting was allowed until 2016 but BRGUs are now protected during the breeding season(Boertmann et al., 2020) and the hunting period during the non-breeding season was shortened from 4.5 months to 1.5 months in 2017 in East Greenland (Enoksen and Olsen, 2017). In Iceland, the hunting season is regulated from 1st of September to 25th of April (i.e. outside breeding season) with some harvesting of eggs allowed under licence.
Marine Protected Area in OSPAR network
No MPA is currently implemented or planned in OSPAR network to specifically protect BRGU marine habitats.
Monitoring programs
In mainland Norway, Svalbard, Jan Mayen, Iceland, Greenland and partly Russia, monitoring of BRGU populations is undertaken (irregularly in Russia and Greenland) (see additional information).
Raise awareness of the status and threats
Globally, the BRGU is categorized as a least concern species on the IUCN Red List (BirdLife International, 2018). There are several international measures to conserve and protect the species, which are listed in the additional information section.
The BRGU was listed as near threatened in Svalbard and as endangered in mainland Norway in 2015 (Kålås et al., 2020). In Iceland, the species has been listed as endangered since 2018 (Icelandic Institute for Natural History, 2018) and in Greenland the species was listed as vulnerable in 2007 (and the category was kept in the last red-list assessment in 2018 (Boertmann and Bay, 2018).
CAFF BRGU programme
The Circumpolar Seabird Expert Group, CBird, within CAFF has developed a Conservation and Action Plan for the BRGU (and COGU) species (see https://www.caff.is/species-conservation-strategy/murre-conservation-strategy-and-action-plan ).
Seabird bycatch programme
Norway has an ongoing project on bycatch of seabirds in Norwegian fisheries since 2009. Reports on bycatch of seabirds in Greenland are part of the Greenland harvest statistics (and are made available by the Greenland Home Rule Department of Hunting and Fisheries). No such program is ongoing in Iceland but recent efforts have been undertaken to estimate bycatch of seabirds and marine mammals in lumpsucker gillnets (details in Christensen-Dalsgaard et al. 2019).
Support, promote and implement the priority actions identified in the Nordic Action Plan for seabirds
Norway is working on a national action plan for seabirds, where the measures in the Nordic Action Plan will be dealt with.
No such action plan currently exists in Iceland and Greenland.
Conclusion (including management considerations)
The best data available (long-term monitoring time-series) suggest that many breeding populations within the OSPAR region are declining (Descamps et al., 2013; Systad, G., personal communication; Merkel et al., 2014; Ezhov, 2019; Boertmann et al., 2020), although some colonies are showing stable or positive population trends since the last assessment (unpublished data, Descamps, S., NPI, Krasnov and Gavrilo, 2018; Þórarinsson, Þ. L., personal communication). However, some of these stable/positive trends are based on only a few population estimates. As BRGU population counts usually show large interannual fluctuations (due for example to diurnal fluctuations in colony attendance, Huffeldt et al. 2013), long-term data are needed to reliably assess the population trend.
Due to its life-history (long-lived and slowly reproducing) characteristics, it is likely that BRGU populations take a long time to recover from population reductions, which makes it important to minimize anthropogenic effects. Special focus should be placed on the declining, red-listed populations in West Svalbard (incl. Bjørnøya), Jan Mayen, northern Norway and East Greenland.
Climate associated effects impacting food availability and concomitantly survival rates in the common wintering grounds of birds from declining populations are considered to be the main threat (Descamps et al., 2013; Frederiksen et al., 2016; Fluhr et al., 2017). In addition, these birds are exposed to hunting (mainly in Greenland) (Frederiksen et al., 2016). In northern Norway, disturbance by predators is also a potential threat (Anker-Nilssen et al., 2020, Reiertsen, T.K. and Systad, G., personal communication). Necessary measures may include a suspension of hunting in the common wintering grounds until currently declining populations have stabilized (Frederiksen et al., 2019). However, recent data suggests that other factors played a role in the observed trends as the population from Iceland, which partly uses the same wintering grounds, has been stable or even slightly increasing in the past years (Þórarinsson, Þ. L., personal communication).
Additionally, efforts are required to reduce illegal hunting and underreporting of harvesting (Frederiksen et al., 2019).
Future measures could also include the implementation of marine conservation areas for seabirds, i.e., key foraging or overwintering areas for BRGUs.
As recent research reported high Hg levels in some BRGUs (Albert et al., 2020), more contaminant monitoring efforts are needed to gain a better understanding of potentially harmful contaminants.
Based on the results of this assessment, BRGUs should remain on the OSPAR list.
Knowledge Gaps
Population monitoring data are lacking or are insufficient for some important breeding populations within the Northeast Atlantic region (e.g., East Greenland, Novaya Zemlya and Franz Josef Land). On Novaya Zemlya no data on population trends are available since the mid-1990s (more details are provided in the additional section below). In East Svalbard, currently, only one colony is monitored annually.
An in-depth understanding of how climate change affects BRGU population status is needed. However, the future effects of climate change, and its indirect consequences (such as lower food availability), are difficult to assess.
Albert C, Helgason HH, Brault-Favrou M, Robertson GJ, Descamps S, et al. 2021. Seasonal variation of mercury contamination in Arctic seabirds: A pan-Arctic assessment. Science of the Total Environment. 750. https://doi.org/10.1016/j.scitotenv.2020.142201 .
Anker-Nilssen T, Barrett R, Christensen-Dalsgaard S, Dehnhard N, Descamps S, et al. 2020. Key-site monitoring in Norway 2019, including Svalbard and Jan Mayen. SEAPOP Short Report 1-2020: 15 pp. Retrieved from www.seapop.no .
Bærum KM, Anker-Nilssen T, Christensen-Dalsgaard S, Fangel K, Williams T, Vølstad JH. 2019. Spatial and temporal variations in seabird bycatch: Incidental bycatch in the Norwegian coastal gillnet-fishery. PloS One. 14. https://doi.org/10.1371/journal.pone.0212786 .
Bakken V, Pokrovskaya IV. 2000. Brünnich‘s guillemot Uria lomvia. Pp. 119-124. In: Anker-Nilssen T, Bakken V, Strøm H, Golovkin A, Bianki VV, Tatarinkova IP (Eds.). The Status of marine birds breeding in the Barents Sea region. Norsk Polarinstitutt Rapportserie. Nr.113. Tromsø.
BirdLife International. 2018. Uria lomvia. The IUCN Red List of Threatened Species 2018: e.T22694847A132066134. https://dx.doi.org/10.2305/IUCN.UK.2018-2.RLTS.T22694847A132066134.en. Downloaded 28/10/20.
BirdLife International. 2020. Species factsheet: Uria lomvia. Downloaded from http://datazone.birdlife.org/species/factsheet/black-guillemot-cepphus-grylle , on 20/10/20.
Boertmann D, Bay C. 2018. Grønlands Rødliste 2018: Fortegnelse over grønlandske dyr og planters trusselstatus (Greenland Red List 2018: List of Greenlandic animals and plants' threat status). Grønlands Naturinstitut, Nuuk. https://natur.gl/raadgivning/roedliste/1-roedliste/ , last accessed 28/10/20.
Boertmann D, Merkel F, Gilg O. 2020. Seabird Breeding Colonies in East and North Greenland: A Baseline. Arctic. 73: 20-39. https://doi.org/10.14430/arctic69885 .
Bond AL, Provencher JF, Elliot RD, Ryan PC, Rowe S, Jones IL, Robertson GJ, Wilhelm SI. 2013. Ingestion of plastic marine debris by common and thick-billed Murres in the northwestern Atlantic from 1985 to 2012. Marine Pollution Bulletin. 77: 192–195. https://doi.org/10.1016/j.marpolbul.2013.10.005 .
CAFF. 2020. Murre Conservation Strategy and Action Plan. https://www.caff.is/caff-webb/439-caff-is/strategies/murre-conservation-strategynew .
Christensen-Dalsgaard S, Anker-Nilssen T, Crawford R, Bond A, Sigurðsson GM, et al. 2019. What’s the catch with lumpsuckers? A North Atlantic study of seabird bycatch in lumpsucker gillnet fisheries. Biological Conservation. 240. https://doi.org/10.1016/j.biocon.2019.108278 .
Convention on Biological Diversity. 2015. Ecologically or Biologically Significant Areas (EBSAs). Seabird Foraging Zone in the Southern Labrador Sea. https://chm.cbd.int/database/record?documentID=204102.
Descamps S, Strøm H, Steen H. 2013. Decline of an arctic top predator: synchrony incolony size fluctuations, risk of extinction and the subpolar gyre. Oecologia. 173: 1271–1282. https://doi.org/10.1007/s00442-013-2701-0.
Egevang C, Merkel F, Labansen A. 2017. Grønlands lomvie-kolonier—en opsummering af polarlomvieoptællinger (Greenland’s guillemot colonies—a summary of the surveys). Pinngortitaleriffik. Greenland Institute of Natural Resources, Nuuk. http://www.natur.gl/fileadmin/user_files/Dokumenter/PAFU/Poster_Lomvietal_juni2017_dk.pdf.
Eisler R. 1987. Mercury hazards to fish, wildlife, and invertebrates: a synoptic review. U.S. Fish and Wildlife Service Biological Report 85(1.10). https://www.pwrc.usgs.gov/eisler/CHR_10_Mercury.pdf.
Enoksen H, Olsen JI. 2017. Selvstyrets bekendtgørelse nr. 1 af 5. januar 2017 om beskyttelse og fangst af fugle (Government Executive Order no. 1 of 5 January 2017 on protection and capture of birds). Gouvernement of Greenland, Law Department, Nuuk. http://lovgivning.gl/da-DK/Lov?rid=56675241-A0B5-4D4E-89F9-C34D78417539 , last accessed 28/10/20.
Environmental Monitoring of Svalbard and Jan Mayen (MOSJ). 2020a. Brünnich’s guillemot. http://www.mosj.no/en/fauna/marine/brunnichs-guillemot.html , last accessed 27/10/20.
Environmental Monitoring of Svalbard and Jan Mayen (MOSJ). 2020b. Pollutants in Brünnich’s guillemots. http://www.mosj.no/en/influence/pollution/pollution-brunnich-guillemot.html , last accessed 25/10/20.
Ezhov AV. 2019. Реакция моевок (Rissa Tridactyla) и кайр (Uria Aalge & U. Lomvia) мурмана на многолетнюю нестабильность кормовой базы в Баренцевом море (Murman kittiwake (Rissa Tridactyla) and guillemot (Uria Aalge & U. Lomvia) reaction on the long-term instability of food avaibility in Barents Sea)). Вестник ТвГУ. Серия "Биология и экология". (TvSU Bulletin. Series "Biology and Ecology"). № 1(53). 5: 72-82. https://doi.org/10.26456/vtbio52.
Fluhr J, Strøm H, Pradel R, Duriez O, Beaugrand G, Descamps S. 2017. Weakening of the subpolar gyre as a key driver of North Atlantic seabird demography: a case study with Brünnich’s guillemots in Svalbard. Marine Ecology Progress Series. 563: 1–11. https://doi.org/10.3354/meps11982.
Frederiksen M, Descamps S, Erikstad KE, Gaston AJ, Gilchrist HG, et al. 2016. Migration and wintering of a declining seabird, the thick-billed murre Uria lomvia, on an ocean basin scale: Conservation implications. Biological Conservation. 200: 61826–61835. https://doi.org/10.1016/j.biocon.2016.05.011 .
Frederiksen M, Linnebjerg JF, Merkel FR, Wilhelm SI, Robertson GJ. 2019. Quantifying the relative impact of hunting and oiling on Brünnich’s Guillemots in the North-west Atlantic. Polar Research. 38: 3378. https://doi.org/10.33265/polar.v38.3378 .
Frederiksen M, Descamps S, Elliott KH, Gaston AJ, Huffeldt NP, Kolbeinsson Y, Linnebjerg JF, Lorentzen E, Merkel FR, Strøm H, Thórarinsson TL (2021) Spatial variation in vital rates and population growth of thick-billed murres in the Atlantic Arctic. Marine Ecology Progress Series 672:1-13.
Garðarsson A. 2006. Nýlegar breytingar á fjölda íslenskra bjargfugla (Recent changes in numbers of cliff-breeding seabirds in Iceland). Bliki. 27: 13-22.
Garðarsson A, Guðmundsson GA, Lilliendahl K. 2019. Svartfugl i islenskum fuglabjorgum 2006-2008 (The numbers of large auks on the cliffs of Iceland in 2006-2008). Bliki. 33: 35-46.
Gaston AJ, Mallory M, Gilchrist HG. 2012. Populations and trends of Canadian Arctic seabirds. Polar Biology. 35: 1221-1232. https://doi.org/10.1007/s00300-012-1168-5.
Huffeldt NP, Merkel FR (2013). Remote time-lapse photography as a monitoring tool for colonial breeding seabirds: A case study using thick-billed murres (Uria lomvia). Waterbirds 36: 330-341.
Icelandic Institute for Natural History. 2018. Válisti fugla (Bird list). Icelandic Institute of natural history (Náttúrufræðistofnun Íslands). Garðabær, Iceland. https://en.ni.is/resources/publications/red-lists/red-list-birds , last accessed 28/10/20.
Kålås JA, Lislevatn T, Gjershaug JO, Strann K-B, Husby M, Dahle S, Strøm H. 2020. Fugler Aves - in: Henriksen S. and Hilmo O. (red.) 2015. Norsk rødliste for arter 2015 (Norwegian red list for species 2015). Artsdatabanken, Norge. Document: https://www.artsdatabanken.no/Files/13973/Norsk_r_dliste_for_arter_2015.pdf
Krasnov YV, Gavrilo MV. 2018. Current status of Brunnich’s Guillemot (Uria lomvia) population on the Franz-Josef Land. Biological Problems of the North: The Materials of International Scientific Conference, dedicated to Academiсian V. L. Kontrimavichus (Magadan, September, 18–22 2018); [ed. by E. V. Khamenkova]. Magadan, 2018. P. 333.
Linnebjerg JF, Frederiksen M, Kolbeinsson Y, Snaethórsson AÖ, Thórisson B, Thórarinsson TL. 2018. Non‐breeding areas of three sympatric auk species breeding in three Icelandic colonies. Polar Biology. 41: 1951–1961. https://doi.org/10.1007/s00300-018-2334-1.
McFarlane Tranquilla LA, Montevecchi WA, Hedd A, Fifield DA, Burke CM, et al. 2013. Multiple-colony winter habitat use by murres Uria spp. in the Northwest Atlantic Ocean: implications for marine risk assessment. Marine Ecology Progress Series. 472: 287-303. https://doi.org/10.3354/meps10053.
Merkel F. 2016. Status for lomvien i Grønland, 2016 (Status of Guillemots in Greenland, 2016). Nuuk: Pingortitaleriffik, Grønlands Naturinstitut, Nuuk. http://www.natur.gl/fileadmin/user_files/Dokumenter/Overblik/Lomviestatus_2016_05_31.pdf.
Merkel F, Labansen AL, Boertmann D, Mosbech A, Egevang C, Falk K, Linnebjerg JF, Frederiksen M, Kampp K. 2014. Declining trends in the majority of Greenland’s thick-billed murre (Uria lomvia) colonies 1981–2011. Polar Biology. 37: 1061–1071. https://doi.org/10.1007/s00300-014-1500-3.
Merkel FR. 2011. Gillnet bycatch of seabirds in Southwest Greenland, 2003–2008. Technical report no. 85. Pinngortitaleriffik, Greenland Institute of Natural Resources, Nuuk. Document: https://natur.gl/wp-content/uploads/2019/07/85-GN_TR_85_Seabird-Bycatch_Merkel__2011.pdf.
OSPAR. 2008. Nomination Uria lomvia, Thick-billed Murre. Case Reports for the OSPAR List of Threatened and/or Declining Species and Habitats. OSPAR, London UK. 3pp. Document: https://www.ospar.org/site/assets/files/1887/thick_billed_murre.pdf.
OSPAR. 2009. Background Document for Thick-billed Murre (Uria lomvia), Biodiversity Series. OSPAR, London UK, 20pp. ISBN 978-1-906840-56-3. Document: https://www.ospar.org/documents?v=7181 .
Pálsson ÓK, Gunnlaugsson Þ, Ólafsdóttir D. 2015. Meðafli sjófugla og sjávarspendýra ífiskveiðum á Íslandsmiðum (By-catch of seabirds and marine mammals in Icelandic fisheries). Hafrannsóknir. 178: 21.
Provencher JF, Gaston AJ, Mallory ML, O’hara PD, Gilchrist HG. 2010. Ingested plastic in a diving seabird, the thick-billed murre (Uria lomvia), in the eastern Canadian Arctic. Marine Pollution Bulletin. 60: 1406–1411. https://doi.org/10.1016/j.marpolbul.2010.05.017 .
Robertson GJ, Wiese FK, Ryan PC, Wilhelm SI. 2014. Updated numbers of murres and dovekies oiled in Newfoundland waters by chronic ship-source oil pollution. Pp 265-275. In: Proceedings of the 37th AMOP Technical Seminar on Environmental Contamination and Response. Environment Canada, Ottawa.
Seapop. 2020. SEAbird POPpulation monitoring programme (SEAPOP) https://www2.nina.no/seapop/seapophtml/ .
Seatrack. 2020a. Winter distribution of Brünnich's guillemots from the Alkefjellet colony. Seabird Tracking Project. Map: http://seatrack.seapop.no/map/?species=uria_lomvia&?season=winter&?colony=alkefjellet , last accessed 28/10/20.
Seatrack. 2020b. Winter distribution of Brünnich's guillemots from the Grimsey colony. Seabird Tracking Project. Map: http://seatrack.seapop.no/map/?species=uria_lomvia&?season=winter&?colony=grimsey , last accessed 29/10/20.
Seatrack. 2020c. Winter distribution of Brünnich's guillemots from the Isfjorden colony. Seabird Tracking Project. Map: http://seatrack.seapop.no/map/?species=uria_lomvia&?season=winter&?colony=isfjorden , last accessed 28/10/20.
Skarphéðinsson KH, Katrínardóttir B, Guðmundsson GA, Auhage SNV. 2016. Mikilvæg fuglasvæði á Íslandi (Important bird areas in Iceland). Fjölrit Náttúrufræðistofnunar. 55: 295.
Speer L, Nelson R, Casier R, Gavrilo M, von Quillfeldt C, Cleary J, Halpin P, Hooper P. 2017. Natural marine world heritage in the Arctic Ocean. Report of an expert workshop and review process. IUCN, Gland, Switzerland.
Stiansen JE, Korneev O, Titov O, Arnberg P, Filin A, Hansen JR, Høines Å, Marasaev SE. 2009. Joint Norwegian–Russian Environmental Status 2008. Report on the Barents Sea ecosystem. Part II. IMR/PINRO JointReport Series: 375 pp. https://imr.brage.unit.no/imr-xmlui/handle/11250/106940 .
Strøm H, Øien IJ, Opheim J, Kuznetsov EA, Khakhin GV. 1994. Seabird censuses on Novaya Zemlya 1994. NOF (Norwegian Ornithological Society). Rapportseri. No. 2-1994. 38 pp.Wilhelm SI, Robertson GJ, Ryan PC, Tobin SF, Elliot RD. 2009. Re-evaluating the use of beached bird oiling rates to assess long-term trends in chronic oil pollution. Marine Pollution Bulletin. 58: 249–255. https://doi.org/10.1016/j.marpolbul.2008.09.018 .
Sheet reference:
BDC2020/ Thick-billed murre or Brünnich‘s guillemot