Status Assessment 2022 - Coral Gardens
| Assessment of status | Distribution | Extent | Condition | Previous OSPAR status assessment | Status (overall assessment) | |
|---|---|---|---|---|---|---|
Region | I | ←→2,5 | ?5 | ↓3,5 | ● | Poor |
| II | ←→2,5 | ?5 | ↓5 | ● | Poor | |
| III | ● | NA | ||||
| IV | ←→2,5 | ?5 | ↓5 | ● | Poor | |
| V | ←→2,5 | ?5 | ↓4,5 | ● | Poor | |
| Assessment of threats | Fishing, particularly demersal trawling and long lining) | Climate change and ocean acidification | Threat or impact | |
|---|---|---|---|---|
Region | I | ←→1,5 | ?3 | |
| II | ↓1,5 | ?3 | ||
| III | ||||
| IV | ←→1,5 | ?3 | ||
| V | ←→1,5 | ?3 | ||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: If in QSR 2010 then enter Regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ) based on Chapter 10 Table 10.1 and Table 10.2 . If a more recent status assessment is available, then enter ‘poor’/’good’
Status*(overall assessment)
red – poor
green – good
? – status unknown
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressure
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threats or impacts (overall assessment)
red – significant threat or impact;
green–no evidence of a significant threat or impact
Blue cells – insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
Confidence
Overall: Low to Medium confidence is placed overall on the assessment.
Distribution: High confidence that the distribution has not changed.
Condition: Moderate confidence that the overall condition across the Regions is still declining. There is good evidence for declining condition, but it is limited to particular sites that have been studied, and broader evidence of decline is drawn from proxies such as the overlap of fishing pressure with the habitat.
Fishing Pressure: Low confidence in the scale of fishing pressure currently acting on the habitat. Available fishing data sets are not comprehensive, and analyses rely on a series of assumptions.
Because of data paucity, it was not possible to conclude whether the habitat extent was increasing or decreasing, and it was not possible to conclude whether the threat from ocean acidification is changing on short time scales.
Background Information
Year added to OSPAR List: 2007 https://www.ospar.org/site/assets/files/44271/coral_gardens.pdf
- The key criteria for their inclusion were:
- their probability of significant decline based on evidence of damage and lack of recovery from research surveys and bycatch reporting of commercial fishermen;
- that they are very sensitive based on longevity, unknown reproductive patterns, uncertain recovery and vulnerability to fishing impacts;
- and that they were threatened by relatively high fishing pressure in deep waters in the OSPAR area. It was considered that the probability of decline and the degree of threat might be higher than in other oceans.
- Major threats were considered to be fishing and climate change; listed in Table 10.3 of Quality Status Report (QSR) 2010 as pH changes and habitat damage. More specifically these threats stem from ocean acidification through its effects on aragonite structures, physical disturbance or damage to seabed, and extraction of, or mortality/injury to, species by fishing.
- In 2010, coral gardens were assessed as being very sensitive, of very important ecological significance, undergoing significant decline, and as currently threatened. https://www.ospar.org/documents?d=7217
Geographical Range and Distribution
The geographic range and distribution of coral gardens remains unchanged.
Coral gardens occur worldwide in depths between 200 m and several thousand meters, and occur shallower where low water temperatures prevail, such as in Norwegian Fjords. Coral gardens are known from all OSPAR Regions except Region III which is dominated by temperate water and continental shelf. Records of coral gardens in Region III (Figure 1) pertain to fouling communities on wrecks and do not meet the OSPAR definition of a coral garden.
Given the biological characteristics of the component species of coral gardens (e.g., slow growth, longevity), additions to the previously understood geographic range represent discovery of the habitat rather than new habitat.
The recorded range in the OSPAR Maritime Area has expanded northward since the last assessment but this reflects increased knowledge rather than a distribution change. Similarly, the number of known occurrences of the habitat has increased due to increased deployment of camera systems to the deep sea and targeted research on this vulnerable habitat.
Method of assessment: 3b
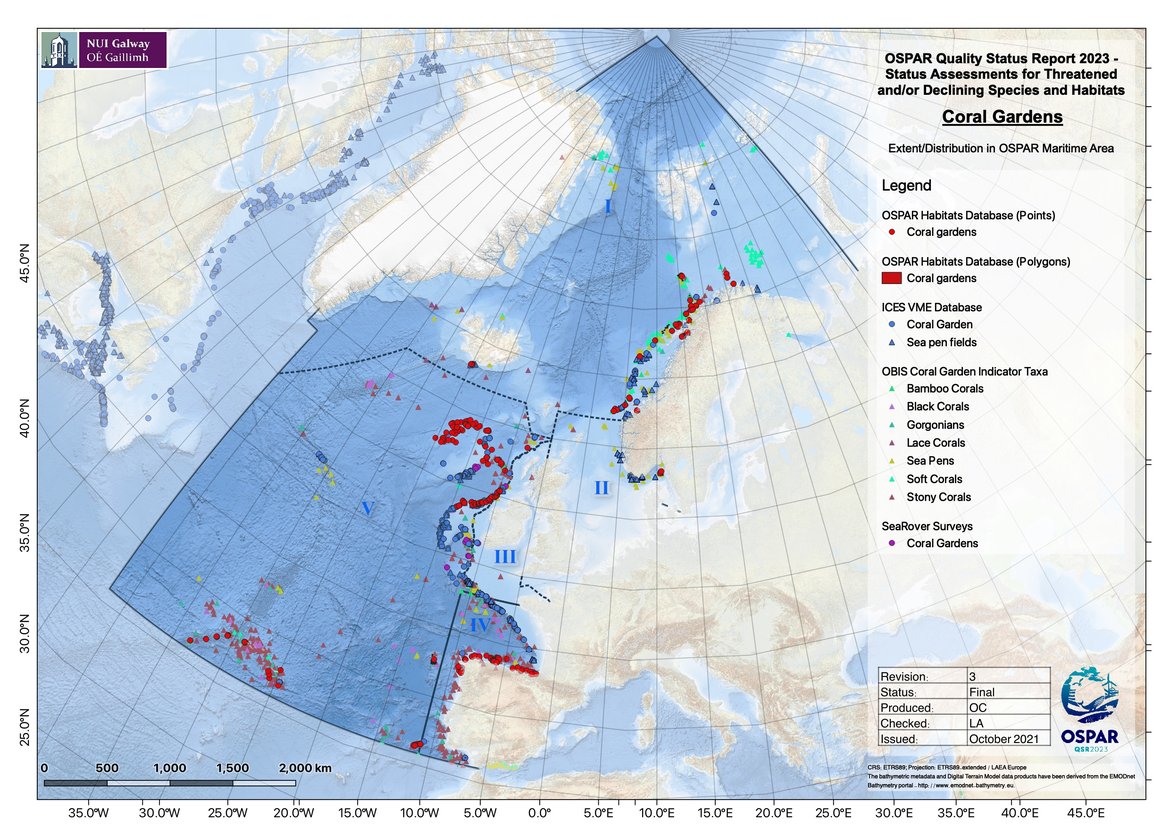
Figure 1: Distribution of known coral gardens in the OSPAR maritime area, and occurrence of coral species that could be indicative of coral garden presence.
Extent
There are insufficient data in any region to draw conclusions on current or past extent of this habitat, and therefore it is not possible to determine change in extent over the period assessed.
Most coral gardens are known from point location data only. While records now exist for their occurrence in many more locations, the extent of the habitats at these locations remains, for the most part, unknown. Transect data may reveal some additional data about habitat extent in some cases, but very few coral gardens have been comprehensively mapped. Exceptions are the coral gardens on the Galician Bank (Region V) and some coral gardens off northern Norway (Region I), represented as polygons on Figure 1. Following survey effort there has been a substantial increase in known records on the continental slope suggesting the habitat is likely to be widespread here.
Method of assessment: 1d
Condition
Overall habitat condition is considered to be declining in all regions.
The definition of coral gardens represents a range of habitats, each with varied characterizing species. Therefore, abundance and density are likely species-specific, without thresholds indicative of ‘good’ habitat condition. Due to data paucity, direct condition assessments are challenging. However, in the absence of in-situ measurements, condition could be inferred from the presence of pressure-causing activities (where pressure-receptor links are known).
An assessment of Nordic Sea VMEs (Buhl-Mortensen et al. 2019) showed overlap between fishing pressure and predicted range of optimal habitat of coral gardens. There is evidence of trawling impacts on coral gardens in all regions where the habitat occurs (e.g., Tong et al. 2012, Buhl-Mortensen et al. 2014, Vieira et al. 2015, González-García et al. 2020), and aggregated surface swept area ratio data suggests that fishing pressure between 2010 and 2016 has overlapped with coral gardens in all regions. In Region V, the condition of some coral gardens has been assessed as unfavorable in previously trawled Marine Protected Areas (JNCC unpublished data).
A proportion of coral gardens exist within Marine Protected Areas and/or NEAFC closures in all Regions, but the proportion protected varies from 2% in OSPAR Region I to 38% in OSPAR Region V (Figure 2). These figures do not account for other protective actions taken beyond the scope of the OSPAR measures.
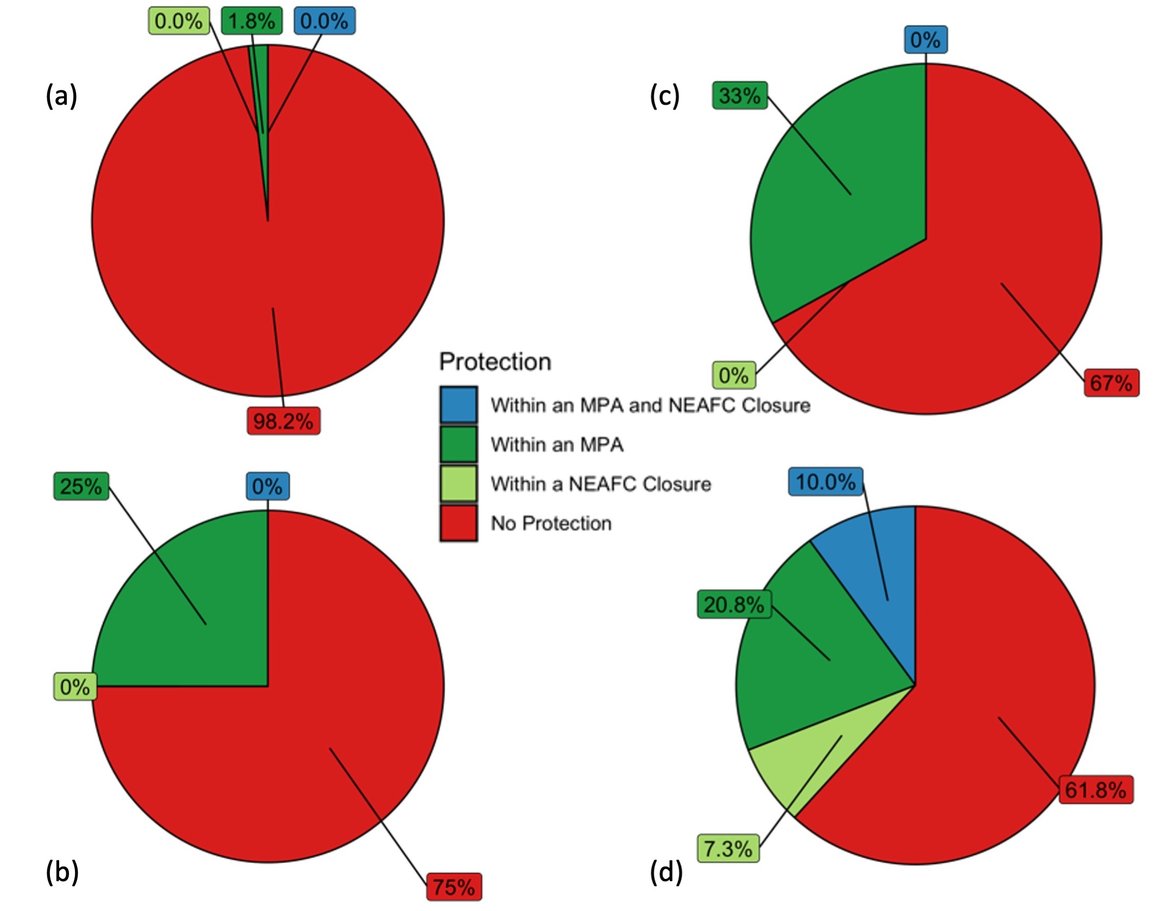
Figure 2: Proportion of coral gardens within an OSPAR Marine Protected Area and/or NEAFC Closure (a) OSPAR Region I; (b) OSPAR Region II; (c) OSPAR Region IV; (d) OSPAR Region V.
On Le Danois Bank (Bay of Biscay, Region IV), habitat recovery is occurring following protection measure introduction (Prado et al. 2019), illustrating the importance of such measures.
Repeated monitoring efforts of known coral gardens, particularly those in MPAs, is essential for future assessment of the condition of this habitat.
Method of assessment: 3c
Threats and Impacts
Key threats to coral gardens in the QSR 2010 were fishing, particularly demersal trawling and long lining, and climate change and ocean acidification (QSR 2010). Marine litter, hydrocarbon and other chemical pollution from vessels and oil and gas operations, deep-sea mining, and the release of aquaculture waste pose potential additional threats.
Key pressure 1: Fishing
The pressure from fishing on this habitat appears to remain stable in all Regions. Bottom trawling has caused extensive damage to coral gardens (Fosså et al. 2000; Fosså et al. 2002; Clark et al. 2010), while coral garden taxa are commonly caught as by-catch in bottom longline fisheries (Sampaio et al. 2012). Under the Intermediate Assessment 2017, the OSPAR Common Indicator on the extent of physical damage to predominant and special habitats (BH3), found 86% of assessed areas (Regions II and III) had physical disturbance from bottom trawling, and 74% experienced consistent year-on-year pressure, likely affecting the ability of habitats to recover. Fishing pressure varied during the assessment period of 2010 to 2020 but there was no overall trend across regions. Analysis of the overlap of global fishing watch data with coral garden habitat records, an analysis which comes with various assumptions, suggests there has been little change on the overall impact of fishing on the habitat since 2016, when Regulation EU 2016/2336 was adopted, except in Region II where there appears to be an overall slight decrease in pressure.
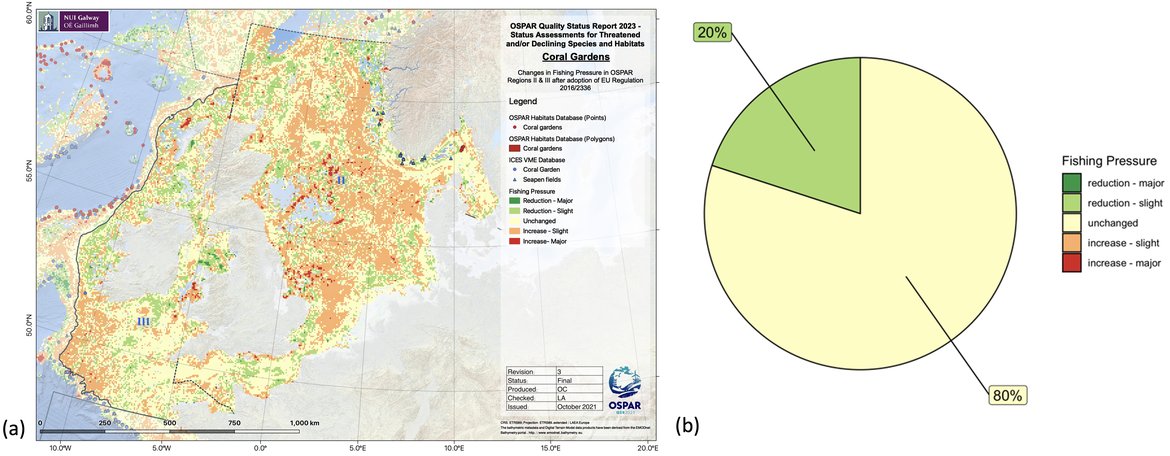
Figure 3: (a) Relative change in fishing pressure across OSPAR Region II after introduction of EU regulation 2016/2336 and (b) change in fishing pressure in those cells known to contain coral gardens.
Key pressure 2: Climate change and ocean acidification
Although ocean acidification is clearly increasing year on year, the current and immediate future impacts on coral gardens are difficult to quantify.
Deep-sea corals, especially those with aragonite rather than calcite skeletons, are vulnerable to the effects of ocean acidification, with impacts predicted by the end of this century. ICES (2014) predicts that, under RCP4.5, waters surrounding the Reykjanes Ridge, the southern Iceland shelf, and edge of the Hatton and Porcupine Banks will be approaching aragonite undersaturation by 2100. Under RCP8.5 most of the North Atlantic will be undersaturated except for the Celtic margin and Bay of Biscay. Other models of aragonite saturation states under RCP8.5 have predicted similar results (e.g., Puerta et al. 2020), including for specific coral garden taxa such as Acanella and Paragorgia (Morato et al. 2020). Predictions tend not to take into account the potential increased vulnerability of early life stages to ocean acidification.
Other effects of climate change likely to impact coral gardens by the end of the century include increases in temperature which could affect distribution, decreased particulate organic carbon inputs, and slowing of the Atlantic Meridional Overturning Circulation which could have broadscale impacts on the whole of the North Atlantic.
Measures that address key pressures from human activities or conserve the species/habitat
OSPAR measures:
3.1 a Iceland has closed all areas where coral gardens are known to exist; Norway has prohibited fishing below 1 000 m; UK, France and Sweden have introduced legislation that allows protection of coral gardens.
3.1 b Mostly the effectiveness of measures implemented has not been assessed, except in Norway where this assessment led to further protective measures, and in Spain where the management plan for El Cachucho MPA is considered effective.
3.1 c Iceland, Norway and Ireland have implemented programmes mapping the distribution of coral gardens; UK and Spain have located some additional sites.
3.1 d In Iceland, fisheries observers record coral by-catch, and companies attempt to record coral by-catch information from their vessels; Ireland is broadening its knowledge through research; the UK is integrating data from fisheries surveys. In the EU, fisheries vessels are obliged to report VME bycatch between 400 and 800 m depth.
3.1 e Iceland, Ireland, Norway, Sweden and UK make all VMS data available to regulators.
3.1 f France, Norway, Spain, Sweden and UK all regularly report new data on the distribution, quality and extent of coral gardens habitat to the OSPAR habitat mapping database.
3.1 g Denmark, France, Iceland, Spain, Sweden and UK have all designated MPAs for coral gardens.
3.1 h Sweden and Iceland have specific management plans for MPAs designated for coral gardens.
3.1 i Iceland, Ireland, Spain, Sweden and the UK report various steps to address significant adverse impacts on coral gardens arising from human activities in their waters, for example through fisheries legislation, including the CFP, through MSFD, and through the Habitats Directive.
Actions taken beyond the scope of the OSPAR measures:
Within the North-East Atlantic Fisheries Commission (NEAFC) Regulatory Area, measures to protect VMEs include the implementation of bottom fishing closure areas and the use of “move-on” rules for fishing vessels, related to encounters of specific quantities of VME indicators within bycatch from bottom-contacting gears (NEAFC Recommendation 19 2014).
Within EU waters, Regulation (EU) 2016/2336 restricts bottom fishing >400 m to the 2009-2011 fishing footprint, prohibits bottom fishing >400 m where VMEs are known or likely to occur (through designation of VME closures) and places a complete ban on bottom trawling deeper than 800 m. Within UK waters, regulation (EU) 2016/2336 is transposed into the Common Fisheries Policy and Aquaculture (Amendment etc.) (EU Exit) Regulations 2019, with the same fishing restrictions and prohibitions in place. In Norwegian waters, Regulation 2011/755 restricts fishing with bottom gears on vulnerable benthic habitats in areas deeper than 1000 m in the Norwegian Economic Zone, including the Fisheries Protection Zone near Svalbard, and the Fisheries Zone at Jan Mayen.
Regulation (EU) 2019/1241 Annex II provides closed areas for the protection of sensitive habitats. It prohibits deployment of bottom trawls, bottom set gillnets, entangling nets and trammel nets within specified areas around El Cachucho, Madeira and the Canary Islands, and the Azores. It further prohibits deployment of bottom set longlines within El Cachucho. It specifies five areas closed to bottom trawls, bottom set gillnets, entangling nets, trammel nets and bottom set longlines in the Porcupine Seabight and Porcupine Bank, and prohibits bottom trawling around the Darwin Mounds.
Conclusion (including management considerations)
An increase in survey effort has led to an increase in our knowledge of the location of coral gardens. Recruitment, which indicates recovery towards a more favorable condition status, has been reported in one MPA, evidencing the positive impact of protected area designation. However, the overall status of the habitat is considered to be poor due to the impact of ongoing fishing pressures in the extensive distribution outside areas protected by some form of legislation. While confidence in the assessment of fishing pressure is low, there is no doubt that fishing pressures remain throughout much of the habitat range, and that this habitat is extremely sensitive to any fishing pressure.
OSPAR implementation reporting demonstrates efforts by Contracting Parties to survey coral garden distribution which are reflected in the increased occurrence records. MPAs protect some of these coral gardens, but new discoveries are not yet protected, and not all MPAs have management plans and this should be rectified. Further work is required to understand the quality and extent of this habitat; some repeat monitoring by Contracting Parties is essential to fill highlighted knowledge gaps and allow assessment of the status and recovery of this habitat. Further implementation of the measures will be required to recover this habitat to good status.
Given the slow growth rate of coral garden taxa, reassessment of recovery/improvement could be on a longer-term (e.g., 10-year) cycle in order to detect any change and assess effectiveness of management measures. However, detection of damage/deterioration from pressures such as fishing may require a more frequent assessment cycle to ensure timely intervention.
Knowledge Gaps
Inaccessibility of habitat and associated cost of monitoring affects the availability of data to access high levels of confidence in assessment. The expected slow change in deep-sea habitats also means that data over longer time frames may be required to assess drivers of change.
‘Coral gardens’ encompasses a variety of very different taxa, from bamboo corals to deep-water sea pens, that are likely to be impacted differently by the pressures acting upon them. Further subdivision into more precise habitat descriptors would aid the assessment of distribution, extent, condition and threats to these habitats.
Most habitats are reported by point data only. To obtain a realistic estimate of areal extent, mapping of known habitats is required.
Repeat monitoring is very limited. Repeat monitoring of coral gardens in protected areas would better inform future prospects of this habitat.
Levin (2021) notes our limited understanding of the effects of climate change on deep-sea ecosystems and recommends distributed observations of climate effects in the deep sea, monitoring both the environmental change and the biological responses.
Together, these data gaps have meant that expert judgement has been used alongside data in most cases.
Method used
Main source of information:
- OSPAR data assessment only
- Assessment derived from third party assessment
- Assessment derived from a mix of OSPAR data assessment and assessments from third parties
Assessment is based upon:
a) complete survey or a statistically robust estimate (e.g. a dedicated mapping or survey or a robust predictive model with representative sample of occurrence data, calibration and satisfactory evaluation of its predictive performance using good data on environmental conditions across entire species range);
b) based mainly on extrapolation from a limited amount of data (e.g. other predictive models or extrapolation using less complete sample of occurrence and environmental data);
c) based mainly on expert opinion with very limited data;
d) insufficient or no data available.
Buhl-Mortensen, P. and Buhl-Mortensen, L. (2014). Diverse and vulnerable deep-water biotopes in the Hardangerfjord, Marine Biology Research, 10:3, 253-267, doi: https://doi.org/10.1080/17451000.2013.810759
Buhl-Mortensen, L. Burgos, J.M., Steingrund, P., Buhl-Mortensen, P., Ólagsdóttir, S.H. and Ragnarsson, S.Á. (2019). Vulnerable marine ecosystems (VMEs): Coral and sponge VMEs in Arctic and sub-Arctic waters – distribution and threats. Nordic Council of Ministers. doi: https://doi.org/10.6027/TN2019-519
Clark, M. R., Rowden, A. A., Schlacher, T., Williams, A., Consalvey, M., Stocks, K. I., Rogers, A. D., O'Hara, T. D., White, M., Shank, T. M., Hall-Spencer, J. M. (2010). The Ecology of Seamounts: Structure, Function, and Human Impacts. Annual Review of Marine Science 2010 2:1, 253-278
FAO. 2009. The FAO International Guidelines for the Management of Deep-sea Fisheries in the High Seas. Activities pages. In: FAO Fisheries and Aquaculture Department [online]. Rome. Updated 30 April 2013. http://www.fao.org/fishery/topic/166308/en
Fosså, J. H., Mortensen, P. B. & Furevik, D. M. 2000 Lophelia-korallrev langs norskekysten forekomst og tilstand. Fisken Havet 2, 1–94.
Fossa˚ J.H., Mortensen P. B. and Furevik D. M. (2002) The deep-water coral Lophelia pertusa in Norwegian waters: distribution and fishery impacts. Hydrobiologia 471, 1–12.
González-García, E., Mateo-Ramírez, Á., Urra, J., Farias, C., Marina, P., et al. (2020). Composition, structure and distribution of epibenthic communities within a mud volcano field of the northern Gulf of Cádiz in relation to environmental variables and trawling activity. Journal of Sea Research, 160-161, 101892. doi: 10.1016/j.seares.2020.101892
ICES. 2014. Final Report to OSPAR of the Joint OSPAR/ICES Ocean Acidification Study Group (SGOA). ICES CM 2014/ACOM:67. 141 pp.
Kazanidis, G., Orejas, C., Borja, A., Kenchington, E., Henry, L.-A., et al. (2020). Assessing the environmental status of selected North Atlantic deep-sea ecosystems. Ecological Indicators, 119, 106624. https://doi.org/10.1016/j.ecolind.2020.106624
Levin LA (2021) IPCC and the Deep Sea: A Case for Deeper Knowledge. Frontiers in Climate, 3:720755. doi: 10.3389/fclim.2021.720755
Morato, T, González-Irusta, J-M, Dominguez-Carrió, C, et al. (2020). Climate-induced changes in the suitable habitat of cold-water corals and commercially important deep-sea fishes in the North Atlantic. Global Change Biology, 26: 2181– 2202. doi: 10.1111/gcb.14996
Prado, E., Sánchez, F., Rodríguez-Basalo, A., Altuna, Á., Cobo, A. (2019). Analysis of the population structure of a gorgonian forest (Placogorgia sp.) using a photogrammetric 3D modeling approach at Le Danois Bank, Cantabrian Sea. Deep-sea Research Part I, 153, 103124. doi: 10.1016/j.dsr.2019.103124
Puerta P, Johnson C, Carreiro-Silva M, Henry L-A, Kenchington E, Morato T, Kazanidis G, Rueda JL, Urra J, Ross S, Wei C-L, González-Irusta JM, Arnaud-Haond S and Orejas C (2020) Influence of Water Masses on the Biodiversity and Biogeography of Deep-Sea Benthic Ecosystems in the North Atlantic. Frontiers in Marine Science, 7:239. doi: 10.3389/fmars.2020.00239
Sampaio, Í, Braga-Henriques, A., Pham, C., Ocaña, O., De Matos, V., Morato, T., & Porteiro, F. (2012). Cold-water corals landed by bottom longline fisheries in the Azores (north-eastern Atlantic). Journal of the Marine Biological Association of the United Kingdom, 92(7), 1547-1555. doi: 10.1017/S0025315412000045
Tong R, Purser A, Unnithan V, Guinan J (2012) Multivariate Statistical Analysis of Distribution of Deep-Water Gorgonian Corals in Relation to Seabed Topography on the Norwegian Margin. PLoS ONE 7(8): e43534. doi: 10.1371/journal.pone.0043534
Vieira RP, Raposo IP, Sobral P, Gonçalves JMS, Bell KLC, Cunha MR (2015). Lost fishing gear and litter at Gorringe Bank (NE Atlantic). Journal of Sea Research, 100, 91-98. doi: 10.1016/j.seares.2014.10.005
Sheet reference:
BDC2022/Coral gardens