Feeder Report 2021 - Aquaculture
Distribution, Intensity and Trends since QSR 2010
Overall trends
2.1 QSR 2010 reported that almost 1,5 million tonnes of farmed fish and shellfish were produced in the OSPAR Maritime Area in 2006. It noted that production of finfish had grown by over 50% in the previous decade, mainly in Arctic Waters and the Greater North Sea, while shellfish farming had remained stable. It anticipated increased activity in all Regions other than the Wider Atlantic.
2.2 Subsequent developments in the volume of marine aquaculture varied according to individual countries, OSPAR Regions, and the type of species being farmed. Data from the Food and Agriculture Organisation of the United Nations (FAO) showed that production by weight in the North-East Atlantic (including the Baltic) increased from approximately 1,5 million tonnes to approximately 2,2 million tonnes between 2008 and 2018. Norway remained by far the largest producer. Norwegian production in Arctic Waters and the Greater North Sea accounted for well over half of total OSPAR countries’ production weight, and for the bulk of the overall increase since the QSR 2010 analysis (data from FAO, 2020a).
2.3 Trends over the decade to 2018 in countries other than Norway varied, with production increasing in some while staying roughly constant in others. In some countries, including France, the Netherlands, Germany and Ireland, aquaculture production in marine and brackish waters in the North-East Atlantic during the 2010s was lower than in some previous periods (FAO, 2020a).
2.4 In 2013, the European Commission sought to boost European Union (EU) aquaculture through new strategic guidelines, addressing four priority areas: simplifying administration; spatial planning to overcome lack of space; enhancing competitiveness; and promoting a level playing field (European Commission, 2013). Member states were to produce multiannual national strategic plans for aquaculture between 2014 and 2020. However, the EU’s Scientific, Technical and Economic Committee for Fisheries (STECF) considered that, for the EU as a whole, production goals in the multiannual plans would not be reached (STECF, 2018). The EU’s 2020 Blue Economy report noted that EU production by weight had been stagnant in previous decades, even though value had increased. It highlighted negative impacts on shellfish production (see below), while production of higher value species, such as salmon, sea bass and sea bream, had grown (European Commission, 2020).
Finfish aquaculture
2.5 Finfish aquaculture in the OSPAR Maritime Area is dominated by salmon production, particularly from Norway. In 2018, Norwegian production from all marine aquaculture (fish, shellfish and other organisms) was over 1,35 million tonnes, mainly of salmon, around 60% more than in 2008, though relatively little changed since 2012 (FAO, 2020a). Norway’s production represented around 1,65% of global aquaculture fish production in 2018; nearly 90% of production (freshwater and marine) was in Asia. Norway (second) and the United Kingdom (tenth) were among the largest global producers of marine and coastal finfish (FAO, 2020b). Norway is the largest global producer of farmed salmon; the United Kingdom is the third largest global salmon producer (FAO, 2020c).
2.6 Charts of production in the North-East Atlantic from 2008 to 2018 are shown below1 . Behind Norway, the United Kingdom was the second largest producer of finfish, almost entirely due to salmon production in the Celtic Seas and the northern part of the Greater North Sea. In the Celtic Seas, Ireland is also a salmon producer; the Faroe Islands and Iceland are producers of salmon in Arctic Waters. Sweden has no finfish aquaculture in the OSPAR Maritime Area; Sweden’s environmental courts have stated that open cage fish farming is not compatible with good environmental practice, effectively banning new sites.
Shellfish aquaculture
2.7 The largest producers of shellfish in OSPAR Regions were Spain in the Bay of Biscay and Iberian Coast (mainly mussels) and France in the Greater North Sea, the Celtic Seas, and the Bay of Biscay and Iberian Coast (predominantly oysters). Spain (sixth) and France (eighth) were among the largest global producers of marine and coastal molluscs (FAO, 2020b)2 .
2.8 Shellfish cultivation also occurs elsewhere in Arctic Waters, the Greater North Sea and the Celtic Seas. Cultivation techniques vary: for example, mussel production in Spain is largely ‘off-bottom’, using ropes fixed to floating rafts (STECF, 2018). In the Netherlands and Germany, blue mussel seeds are collected with similar techniques using ropes fixed to poles or anchors. For maturing of the seeds, mainly ‘on-bottom’ techniques are used, using natural designated beds. These may have different environmental impacts – for example, during maintenance and extraction of species (for more detail see OSPAR, 2009 and European Commission, 2012).
2.9 The STECF analysis noted the impact of factors such as shellfish mortalities, weather conditions and diseases. The EU’s 2020 Blue Economy report referred to impacts of disease and lack of seed on mussels and other shellfish in 2013, although production had recovered subsequently (European Commission, 2020). For 2019, FAO reported that Spanish mussel production was likely to reach a new high; conversely, the hot summer in France had an impact on oyster mortality, with lower production expected in 2020 (FAO, 2019). Since 2008, significant mortality of juvenile oysters in France has been experienced, with the OsHV1 μvar virus being an important factor (STECF, 2018).
Other aquaculture
2.10 Other marine aquaculture products are small. FAO estimates that 346 tonnes of crustacea were produced in the North-East Atlantic in 2018, the majority in Spain. Around 700 tonnes of aquatic plants were farmed, mainly brown seaweeds, the majority in France (FAO, 2020a).
Recirculating Aquaculture Systems
2.11 Recirculating Aquaculture Systems (RAS) can be closed containment land-based facilities (the majority) or marine based floating closed / semi-closed containment systems (currently in development). Currently the main species cultured in RAS in the OSPAR region are finfish. According to Eurostat, the production of fish for human consumption in ‘recirculation systems’ has been relatively stable at between 1,5% and 2% of the total aquaculture production over the period, with an average yearly volume of nearly 24 000 tonnes (EUMOFA, 2020).
2.12 Since this is an emerging technology with potentially important implications for the marine environment, a fuller description of RAS and its implications for OSPAR is provided in Annex A to this report.
Distribution
2.13 The maps below illustrate the distribution of marine aquaculture in OSPAR regions.
Figure 6: Finfish production sites and locations, (Source EMODnet 2020) https://odims.ospar.org/layers/geonode:emodnet_finfish
Figure 7: Shellfish production sites and locations, (Source EMODnet 2020) https://odims.ospar.org/en/submissions/ospar_aquaculture_shellfish_2020_08/
2.14 The European Environment Agency’s indicator assessment on aquaculture includes total marine production as a function of coastline length as a rough indicator of the potential environmental impacts of marine aquaculture. However, it notes that these figures do not take account of the pressure exerted by different production systems or local conditions. Nor is aquaculture evenly distributed along the coast (EEA, 2019).
Economic value
Finfish
3.1 The total value of marine aquaculture production in the OSPAR area has risen sharply over the past decade, mainly driven by increases in higher value species, notably salmon. Like production volumes, the economic value is dominated by Norway, with salmon production worth over €6,7 billion in 2018, compared with €1,9 billion in 2008 (Eurostat, 2020). The value of aquaculture production in Norway exceeds that of the entire EU production of fish and shellfish (Eurostat, 2019). Salmon is the largest single fish commodity in world trade of fish and fish products, with growth driven by demand both in developed and developing markets (FAO, 2020b).
3.2 The United Kingdom had the second highest marine aquaculture production value among OSPAR countries, again dominated by salmon. The value of United Kingdom salmon production grew in the decade to 2018 from around €500 million to around €1 billion. Increases in production value of salmon were also reported in Ireland and Iceland (FAO, 2020a).
3.3 Eurostat figures show a general upward trend in the value per tonne of salmon over the period, particularly in earlier years. The FAO reports that farmed Atlantic salmon is one of the most profitable and technologically advanced aquaculture industries in the world, backed by coordinated international marketing and product innovation. Factors limiting supply, such as site availability and regulatory constraints, have led to price increases. Shorter term prices can be quite volatile – for example, the FAO’s Globefish information exchange reported steep falls in global salmon prices in the first half of 2019, linked to good production in Norway and Chile, but prices reaching near record levels later in the year, driven by strengthening demand in traditional and emerging markets (FAO, 2020b; FAO, 2020c).
3.4 In Spain and Portugal, turbot (land-based production in tanks) was the largest component of finfish production value in the North-East Atlantic region, but did not show a significant increase in value over the decade, reflecting relatively stable average prices over the period (STECF, 2018).
3.5 Finfish aquaculture is capital intensive, with relatively large investment in physical equipment and stocking of cages compared to the input of labour. Labour productivity in sea cage farms is high compared to other EU aquaculture segments (STECF, 2018).
Shellfish
3.6 Changes in shellfish production value have been less substantial than for finfish. France’s production of molluscs in the North-East Atlantic was worth over €400m annually across the decade, making France the third highest aquaculture producer, in terms of value (finfish and shellfish) in the OSPAR region. The value fluctuated year on year, with a dip in 2014 and 2015, but did not increase significantly across the period. Nearly three-quarters of the value in 2017 and 2018 was from oysters, with most of the remainder due to (blue) mussels. Shellfish production (mainly mussels) accounted for over half the production value in Spain (North-East Atlantic), and showed some increase over the decade (Eurostat, 2020).
Employment
3.7 The table below shows marine aquaculture employment statistics in OSPAR countries in 20183
| Employees in finfish aquaculture | Employees in shellfish aquaculture | |
|---|---|---|
| Belgium | 0 | 0 |
| Denmark | 150 | 10 |
| France | 152 | 13 841 |
| Germany | 0 | 127 |
| Iceland | ||
| Ireland | 180 | 1 719 |
| Netherlands | 5 | 257 |
| Norway | 7 903 | |
| Portugal | 257 | 2 362 |
| Spain | 2 379 | 14 465 |
| Sweden | 0 | 68 |
| United Kingdom | 1 900 | 700 |
3.8 For some activities, such as shellfish production in Spain and France, the sector has a significant social role as a local employer, often through small family-owned businesses, with a significant component of part-time and seasonal working. In Germany and the Netherlands, however, mussel production is based on a few relatively large companies (STECF, 2018).
Future trends
4.1 While growth in global aquaculture was lower in the past decade than the high rates of the 1980s and 1990s, it is still an expanding sector globally. The FAO’s 2020 report on the state of the world’s fisheries and aquaculture projected an increase of around 32% in world aquaculture between 2018 and 2030. This is a slower rate of growth than in the previous decade, due to factors such as limits on production sites, better environmental regulation, aquatic diseases, and decreasing productivity improvements. Nevertheless, aquaculture, rather than capture fisheries, is expected to remain the driving force behind overall increases in global fish production4 . The FAO also projected that fish prices generally would remain high, with increases in nominal terms albeit with some decline in real terms (FAO, 2020b).
4.2 Within the OSPAR region, overall projections of future aquaculture volumes are, as now, highly influenced by what happens in Norway. The FAO’s 2020 report projected an increase of nearly 20% in Norwegian aquaculture production between 2018 and 2030, to over 1,6 million tonnes (FAO, 2020b). For the longer term, the Norwegian Government’s 2017 ocean strategy refers to an estimate of the potential for a six-fold increase in revenue for Norwegian biomarine industries by 2050, with a large proportion of this growth from aquaculture. As well as salmon, this could include new species and algae such as seaweed and kelp (Norwegian Ministry of Trade, Industry and Fisheries, 2017). An update to the strategy refers to a new system for adjusting aquaculture production capacity to facilitate predictable and environmentally sustainable growth of salmon and trout farming. Norway is also refining its regulatory system to facilitate new developments in offshore aquaculture (Norwegian Ministry of Trade, Industry and Fisheries, 2019). Research informing the International Council for the Exploration of the Sea’s (ICES) Working Group on Scenario Planning in Aquaculture referred to a Norwegian expansion target of a fourfold increase by 2050 (Froehlich et al, 2020). The ICES ecosystem review of the Barents Sea notes that aquaculture is increasing along the coasts and in the fjords of northern Norway and Russia, with several commercial fish farms producing salmon, trout and shellfish (ICES, 2019). Furthermore, the number of applications to the local and regional authorities for land-based aquaculture in Norway is increasing (Norwegian Environment Agency, personal communication).
4.3 Other salmonid producers also have growth ambitions. For example, the Scottish Government is supporting the aquaculture industry in its growth strategy, which aims to double the economic contribution of the aquaculture sector by 2030 (Aquaculture Industry Leadership Group, 2017). The Icelandic Government has stated an expectation that its production will double over the next few years from 2017 levels, and there is scope for further expansion (Government of Iceland, 2020). Some growth in salmon aquaculture was also envisaged in Ireland’s national plan for aquaculture development (Department of Agriculture, Food and the Marine, 2015). A growth strategy for English aquaculture (finfish and shellfish) is being prepared under the Seafood 2040 initiative (Seafish, 2018).
4.4 In relation to shellfish production, the FAO has reported an increase in current demands for bivalves, helping to drive increases in Spanish mussel production (FAO, 2019). Offshore production of shellfish is also a possibility (Collins et al, 2020, EU TAPAS, 2020).
4.5 Within the EU as a whole, prospective increases in demand within and beyond Europe are seen as an opportunity to expand European aquaculture. The EU’s 2018 Economic Report on the EU Aquaculture Sector anticipated higher future demand for fish driven by population increase and income, and by awareness of health benefits of fish consumption (STECF, 2018). The FAO projected an increase of 13% between 2018 and 2030 in EU aquaculture (including freshwater as well as marine) (FAO, 2020b). The EU’s Blue Economy report 2020 considered it realistic to expect a growth of EU aquaculture products with a high degree of control, such as finfish in closed systems, while shellfish production in open waters would be more influenced by environmental factors (European Commission, 2020). At present, most planning for expansion is relatively short-term; ICES work on scenario planning in aquaculture reported that, of OSPAR countries, only Spain (a tripling of production by 2030) and Norway had strategic plans for aquaculture growth out to 2030-2050 (Froehlich et al, 2020).
4.6 The scale of any growth will in practice be influenced by several factors. Future economic developments globally and in Europe will have an impact. For example, the FAO’s January 2020 update on world seafood markets notes that these markets are highly sensitive to wider economic conditions, and reported that in 2019, Brexit-related economic challenges and trade issues had contributed to a slowdown in seafood trade in the EU. Global economic uncertainties remain, including the impact of COVID-19 (FAO, 2020c). International competitiveness will also influence the sector’s economic performance. The EU-funded Aquaspace project noted that a strong competitive advantage of EU aquaculture is related to the quality and sustainability of its products, and future success depends in part on maintaining healthy marine environments (O’Hagan et al, 2017). For example, Spanish mussel producers now have a certification of Protected Denomination of Origin in the EU, which will help with market image (FAO, 2019).
4.7 There are also uncertainties or constraints specific to aquaculture. Aquaculture may be competing for space with other uses such as tourism and recreation, shipping, fishing, aggregate extraction and energy production, although there may be some synergies with offshore wind structures (European Commission, 2020). The Aquaspace project advised that while improvements in nutrition and feed, species growth, disease treatments and production methods can achieve some production expansion, more space for aquaculture would be necessary if growth potential were to be achieved. The project looked at how to optimise and increase the area available for both marine and freshwater environments, through an ecosystem approach to aquaculture. For example, it noted that development of maritime spatial planning would be important if production is to take place offshore (O’Hagan et al, 2017). Similar issues are identified by ICES work on aquaculture (Froehlich et al, 2020). A review of global experience of using an ecosystem approach concludes that it has promoted greater sustainability in aquaculture but has had varying degrees of uptake (Brugère et al, 2019).
4.8 The FutureEUAqua project, supported by the EU’s Horizon 2020 programme, is working on ways to promote the sustainable growth of climate-change resilient, environmentally-friendly aquaculture of fish and low level trophic organisms. Areas of interest include genetic selection, ingredients and feeds, monitoring technologies, innovative products, and optimal production systems (more details at FutureEuAqua, 2020). In the case of feeds, while fish meal and fish oil were traditionally the bulk of feed, recent years have seen a shift towards use and development of other sources including terrestrial plant- or animal-based proteins, seafood processing waste, microbial ingredients, insects, algae and genetically modified plants (e.g. Costello et al, 2020). According to the Norwegian Government, around 70% of ingredients in salmon feed are vegetable, while the remainder is from marine raw materials, including trimmings and by-products from fisheries (Norwegian Ministry of Trade, Industry and Fisheries, 2019).
4.9 The EU’s TAPAS project has looked at the potential for offshore aquaculture of species such as of salmon, blue mussel and oyster. It suggests that large areas of those seas could be used for aquaculture, if logistical and administrative issues could be overcome (Wallhead et al, 2020). An ICES working group on open ocean aquaculture (WGOOA) is looking at issues such as environmental influences, technical challenges, system design, site selection, and economic aspects, with the aim of developing a roadmap for future open ocean aquaculture.
4.10 Historically, marine aquaculture in the OSPAR region has been dominated by a few species. The EU Aquaspace project noted that more understanding and new strategies would be needed if this were to change, and that failed attempts to produce Atlantic cod (Gadus morhua) in Norway, United Kingdom and Iceland illustrate the difficulty of introducing new species economically to aquaculture. In addition, new species may not deliver the same commercial returns as existing species (O’Hagan et al, 2017). As well as aquaculture of fish species, the idea of managed cultivation of seaweed is a potential new development in areas such as the Netherlands and Greenland. Expansion of seaweed aquaculture has potential as a source for food, animal feed, fuel, cosmetics, and pharmaceuticals, but understanding of possible impacts such as disease, alteration in population genetics and alterations to the physio-chemical environment still needs to be developed (e.g. Campbell et al, 2019).
4.11 Climate change is a significant uncertainty. Possible negative impacts include loss of production or infrastructure due to extreme events, diseases, toxic algae and parasites; decreased productivity due to suboptimal farming conditions; environmental factors affecting natural production of oyster spat or mussel seed; the ability to control issues with salmon farming such as sea lice and escapees; limited access to feeds from marine and terrestrial sources; loss of sites due to sea level rise; or the effect of ocean acidification on shellfish. However, there is also the possibility of positive impacts (e.g. FAO, 2018; STECF, 2018).
4.12 The EU-funded CERES project has reviewed the potential impacts of climate change on European aquaculture, including direct effects of changes in temperature, pH, dissolved oxygen concentration and salinity, as well as developing tools to project the occurrence and risk of indirect effects such as disease, algal blooms, and jellyfish (synthesis report at Peck et al, 2020). The synthesis report also summarises the impact of scenarios in the productivity by mid-century and end-century of certain finfish and bivalve species used in aquaculture, in the economic performance of aquaculture sectors, and in the vulnerability to climate change of aquaculture in individual countries. Projected impacts vary according to different scenarios and sectors; this is illustrated further in individual case studies produced by the project (CERES, 2020).
4.13 This complexity is also evident in other reports, for example:
- in Norway, salmon aquaculture may be vulnerable to temperature rise exceeding the thermal optima, including in extreme events such as heat waves, although there could be positive effects on growth rates in some areas. The socio-economic outcomes as well as adaptation strategies are difficult to assess, however, given the diversity of farming sites and evolving technology (Climefish, 2019a);
- temperature increase was likely to increase growth rates for most UK farmed species, but issues such as sea lice and gill disease in salmon, or harmful algal blooms and jellyfish swarms, are likely to get worse. Overall, technical and management changes in the rapidly evolving aquaculture industry make long-term impacts of climate change difficult to forecast (Collins et al, 2020);
- higher temperatures and increasing summer northerly winds could have positive effects for mussel aquaculture in north-west Spain, but could also increase harmful algae. Extreme weather events could cause detachment of mussels or loss of mussel rafts. Predicting the socio-economic consequences is challenging (Climefish, 2019b).
Quality Status Report 2010 and Intermediate Assessment 2017
5.1 QSR 2010 outlined several environmental issues linked to marine aquaculture, such as:
- genetic interactions between farmed fish and wild stocks;
- transfer of parasites and diseases;
- the spread of non-indigenous species;
- the dependence on industrial catches of wild fish to feed fish in aquaculture;
- eutrophication as a result of nutrient enrichment from feeds and effluents;
- competition between escaped farmed fish and wild stocks for spawning grounds;
- release of chemicals used to prevent equipment fouling or to treat parasites and diseases;
- displacement of bird and seal populations as a result of scaring devices;
- impacts from shellfish harvesting and from mussel seed collection;
- litter.
5.2 The QSR referred to the use of best environmental practice to reduce inputs of chemicals, and measures under OSPAR’s Eutrophication, Hazardous Substances and Biodiversity and Ecosystems Strategies as a way to monitor, assess and regulate impacts. It also noted the existence of national and EU measures, and risk assessment protocols from ICES on the use of non-indigenous species in aquaculture.
5.3 IA 2017 did not specifically cover aquaculture, but its discussion of eutrophication in OSPAR’s Third Integrated Report on the Eutrophication Status of the OSPAR Maritime Area commented on nutrient inputs from aquaculture - see paragraph 6.13 (OSPAR, 2017).
Analysis of specific pressures, impacts and measures
Cross-cutting issues and measures
6.1 The potential environmental impacts of finfish and shellfish aquaculture listed in paragraph 5.1 were described in OSPAR (2009). The potential impacts remain substantially the same, and so that analysis is not repeated in depth here. The existence of a potential pressure does not necessarily mean that harmful impacts will occur, but means that assessment of the location and management of individual facilities is important in limiting pressures.
6.2 Environmental pressures from aquaculture vary considerably depending on the species being cultivated and the production methods being used. For example, at a general level, the pressures from finfish farming are very different to those from shellfish cultivation. The scale and location of impacts can be particularly important to consider for aquaculture, and the impact of potential pressures is strongly influenced by local environmental circumstances, including hydrographic conditions, and the nature and sensitivity of local ecosystems. This means that assessment of the location and management of individual facilities is important in limiting pressures (OSPAR, 2009; European Commission, 2012; ICES, 2014).
6.3 Measures to mitigate environmental impacts, including modelling approaches, have moved forward, although knowledge gaps remain. The ICES Working Group on the Environmental Impacts of Aquaculture (WGEIA) reported on its work in December 2020 (ICES, 2020). Its report contains an extensive summary of different environmental impacts and how these are regulated and monitored in different countries. The report highlighted areas where improved aquaculture management can lead to better environmental performance, and made recommendations for prioritised research. The report is referred to further below. HELCOM has developed recommendation 37-7 on sustainable aquaculture in the Baltic Sea region and is currently in the process of elaborating BAT / BEP in this context.
OSPAR measures
6.4 OSPAR has taken few specific measures on aquaculture. OSPAR guidelines on reporting nutrient discharges/losses from marine and freshwater aquaculture plants were issued in 2004 and revised in 2018 (OSPAR, 2018). Under the Riverine Inputs and Direct Discharges (RID) programme there is some reporting of inputs from aquaculture (nutrients, heavy metals and certain organic pollutants). PARCOM Recommendation 94/6 covers the reduction of inputs from potentially toxic chemicals used in aquaculture. In 2006, OSPAR agreed that, for the time being, implementation reporting on PARCOM Recommendation 94/6 could cease. Since there are now significant developments in the aquaculture industry giving rise to concern about pollution, OSPAR decided in 2020 to initiate a new reporting round on Recommendation 94/6 and compile the results in 2022.
6.5 In addition, managing pressures from aquaculture is relevant to OSPAR action on species and habitats. OSPAR Recommendation 2016/03 on furthering the protection and conservation of the Atlantic salmon (Salmo salar) in Regions I, II, III and IV of the OSPAR Maritime Area covers the need to assess measures to manage threats from escape and accidental release from fish farms, and spread of diseases and parasites. There have been no OSPAR measures specifically on non-indigenous species from aquaculture.
6.6 OSPAR work on litter is dealt with in paragraph 6.36 below.
Cross cutting international and national measures
6.7 Within the EU, key overarching directives have set a framework within which aquaculture operates:
- The Water Framework Directive (WFD), which requires member states to take measures to prevent deterioration of the ecological and chemical status of waters, restore polluted waters, reduce pollution and cease or phase out inputs of hazardous substances5 . Its scope includes coastal waters one nautical mile out to sea and, for chemical status, out to twelve nautical miles. It also contains requirements for monitoring and management of shellfish protected areas;
- The Marine Strategy Framework Directive (MSFD) aims to achieve good environmental status (GES-MSFD) in marine waters by 2020;
- The Environmental Impact Assessment Directive and the Strategic Environment Assessment Directive, which set procedures to ensure that implementation of plans, programmes or projects, including aquaculture, takes account of their likely environmental effects;
- The Birds Directive, which protects wild birds and their habitats, and the Habitats Directive, which protects rare, threatened or endemic animals, plants and habitats;
- The Aquatic Animal Health Directive, covering health requirements and disease prevention and control in aquaculture.
- For EEA countries, some of these directives apply but in other cases, national legislation provides the overarching framework.
6.8 The European Commission has produced guidance on sustainable aquaculture in the context of the Natura 2000 network (European Commission, 2012) and on the application of the WFD and MSFD in relation to aquaculture (European Commission, 2016). These provide information on regulatory good practice and suggestions to national authorities and industry about the requirements of the Directives in relation to aquaculture. More detailed background on sustainable aquaculture in the context of WFD and MSFD can be found in Jeffrey et al (2014).
6.9 The Natura 2000 guidance notes that there are many well-known Natura 2000 areas where aquaculture activities are taking place sustainably, including the Wadden Sea in the Netherlands, Arcachon in France, the Sado Estuary in Portugal, Doñana in Spain, shellfish culture in England and Wales and several lochs in Scotland. It emphasises the need for thorough assessment of the potential impacts of aquaculture, including careful consideration of the location of facilities.
6.10 The guidance on WFD / MSFD covers various pressures including benthic impacts and nutrients; diseases and parasites; chemical discharges; escapees and non-indigenous species; and physical impacts and predator control. The guidance is being built on further by the EU TAPAS project, which runs from 2016 to 2020 and is looking at sustainability issues in aquaculture, including development of an aquaculture sustainability tool box to support planning and licensing decisions (EU TAPAS, 2020).
6.11 The multi-faceted and site-specific nature of the environmental impacts of aquaculture mean that the primary route for managing these impacts is through the assessment, including through modelling, of individual projects or plans under individual countries’ regulatory systems – for example, considering the carrying capacity of the environment, stocking levels, nutrient dispersal, physical impacts on the environment, or the interaction with threatened species. This paper does not seek to go into detail on these mechanisms. There are also technical developments in the management of aquaculture – for example, potential moves towards more use of recirculating aquaculture systems for growing larger fish before stocking sea pens (Jeffrey et al, 2015); again, the paper does not cover these in detail. Some developments in relation to several of the issues highlighted by OSPAR and others are, however, summarised below.
Specific impacts and measures - finfish
6.12 Finfish aquaculture in large net cages, such as those used for salmon, have the potential for multiple environmental effects. This could include accumulation of organic waste, such as excretory products or uneaten food, affecting factors such as sediment chemistry and benthic organisms, and nutrient enrichment through nitrate, ammonia, phosphate and organic carbon, leading to local eutrophication. There may also be physical impacts of cages, or harm to predators attracted by the farmed fish.
| Source | Type | Impact On | Effect | Control |
|---|---|---|---|---|
| Uneaten food, faeces, pseudofaeces, scales. | Solid organic | Seafloor +/-100m | Enrichment, elimination of fauna. H2S outgassing | Improve feeding, site rotation and harrowing |
| Excreta and food leachate | Soluble organic | Water – generally localised | Eutrophication and toxic ammonia | Site selection and rotation |
| Harvesting and seed dredging | Ecological change Physical | Fish, benthic communities, seabed habitat damage. | Wild and commercial stock viability and habitat destruction | Fisheries and seed management |
| Therapeutants, antifoulants, feed additives, disinfectants, net washings | Chemical contamination | Water, sediments and biota | Toxicity to organisms, water and sediment quality, food chain | Proper usage, good husbandry, site selection and mechanical cleaning |
| Escapees | Ecological change | Wildfish, ecosystem, and habitat. | Disease, sea lice, genetic, competition and displacement | Site and equipment selection, maintenance, marking, recapture, containment |
| Stock | Disease parasites reservoir | Wildfish and wild shellfish | Infections and disturbance | Site selection, management, husbandry, treatment |
| Translocation of stock | Alien species | Ecosystems and habitats | Displacement, competition | Certification of stock, containment and restriction of movement |
| Predation | Behavioural | Birds and mammals | Mortalities, behavioural | Predator nets, scarers |
| Access and onsite activity | Visual disturbance, compaction | Birds, mammals, seabed, | Disturbed feeding and roosting | Limited access (frequency and timing), single access route |
| Space utilisation | Physical Presence | Other Users | Visual impact, navigation, other users | Site selection, spatial planning, marking |
Nutrients
6.13 The nutrient inputs from the largest finfish aquaculture systems in the OSPAR region were referred to in OSPAR’s 2017 report on eutrophication (OSPAR, 2017). In some areas, aquaculture can be a significant source of nutrients. For example, Norway had reported increases in ammonia inputs from aquaculture into the Norwegian Sea, Barents Sea and North Sea. Fish farms in the north and west of Scotland may be a significant source of nutrients in areas where freshwater inputs are low; the report noted that while they may not be available for immediate use by algae or higher forms of plant life, they are likely to make an important contribution to biogeochemical cycling.
6.14 The ICES WGEIA concluded that, in relation to organic waste, the ecological effects on the soft sediment habitats beneath the majority of fish farms are well understood and can be measured and regulated. Trends towards more dispersive, high current sites, including open ocean installations, require more understanding of regional effects and impacts on hard bottom sites. However, the trend towards more dispersive sites should help to mitigate impacts from organic waste from aquaculture (ICES, 2020).
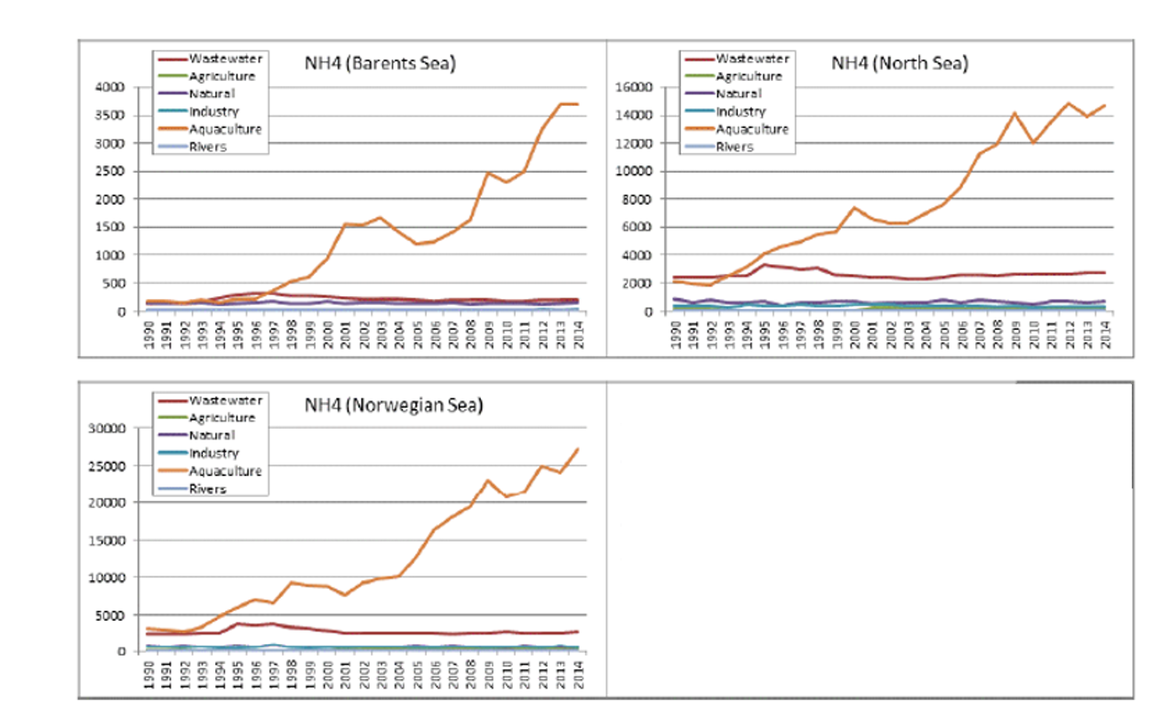
Figure 9: Inputs of ammonia into Norwegian Sea, Barents Sea and North Sea (from OSPAR, 2017)
Unit: Tonne (Source Norderhaug et al, 2016)
Impacts on wild fish – escapes and diseases
6.15 Advice from ICES to OSPAR in 2014 considered the interactions between wild and captive fish stocks, such as the impact of escaped stock on wild fish through interbreeding and competition; transmission of disease; and the ecological consequences of using antibiotics, pesticides and disinfectants, and anaesthetics (ICES, 2014). A report from the North Atlantic Salmon Conservation Organisation concluded that there was growing evidence that salmon farming could affect wild salmon populations, through the impacts of sea lice as well as farm escapees (NASCO, 2016).
6.16 Escapes of farmed fish, resulting from operations or from incidents such as technical defects, accidents or bad weather, is a particular threat from finfish aquaculture, with potential impacts on the genetic structure of wild populations, and on transfer of disease. Climate change could add to the risks of storm events.
6.17 The EU Prevent Escape project, running from 2009 to 2012, looked at the scale of this issue and ways to manage it. One aspect looked at escapes from salmon production in Norway following the introduction of a new technical standard for sea cages in 2006. The number of escaped salmon declined from over 600 000 per year between 2001 and 2006, to less than 300 000 fish per year between 2007 and 2011. Based on this success, it recommended that European policy-makers introduce a technical standard for sea cages, combined with enforcement mechanisms (Prevent Escape, 2013).
6.18 In Norway there is a national monitoring programme for escaped salmon, and fish farmers have to report escapes to the national authorities. Numbers of escapees vary between years, and can be affected substantially by large events. In 2019, nearly 285 000 salmon escaped from Norwegian aquaculture: of this number, nearly 180 000 were from one event involving fish of 0,02 kg, and another 50 000 from an event involving fish of 4 kg (Barentswatch, 2020). Figure 10 is taken from this source.
6.19 In the UK, the Scottish Government introduced new technical standards to help prevent escapes of farmed fish; all equipment had to meet that standard by 2020. It covers factors such as mooring of pens, construction of pens, net design and construction, and feed barges. Escapes of farmed fish are reported to the national authority (Marine Scotland, 2015). The Scottish database on escapes reports over 47 000 escapes of salmon in 2018 and over 60 000 in 2019. Over 73 000 salmon escaped in an incident in January 2020 associated with storm damage (Scotland’s Aquaculture, 2020).
6.20 Although there is substantial knowledge about escapes of farmed salmon and their impacts, ICES noted the scope for improvements in reporting of escapes, defining the cause of escapes, and understanding the impacts on wild populations. It also referred to development of approaches such as the use of sterile salmon and of closed containment systems. Knowledge about the impact of escapes of species other than salmon is limited (ICES, 2020).
6.21 In relation to diseases, ICES advice to OSPAR noted that measures to mitigate disease operate at two levels: area-based (coordinated stocking, harvesting, and fallowing) and farm-based (vaccination, early pathogen detection, veterinary prescribed treatments, and depopulation or early harvest in the event of viral disease). It also referred to problems in managing sea lice transmission due to increasing parasite resistance to chemicals used for control. It suggested establishing management zones incorporating biomass limits and coordinated management (ICES, 2014).
6.22 According to the Norwegian Government, regulations relating to sea lice have been tightened in recent years and there are currently very low levels of sea lice in Norwegian facilities. Increase in salmon and trout production is regulated through a ‘traffic-light’ system relating to sea lice prevalence. Fish farms have to report the levels of sea lice. Nonetheless, sea lice can be a limitation on aquaculture growth: for example, the Norwegian government recently published a new map of areas where aquaculture growth is permitted and others where it is not allowed to expand further, or even has to cut back. In Scotland, if a weekly average adult female sea lice count per fish of two (or above) is recorded on any fish farming site, this must be reported to the authorities within a week and action has to be taken to reduce the level of lice (Marine Scotland, 2019).
6.23 Despite substantial knowledge about sea lice and their impacts, uncertainties remain, such as the potential influence of climate change on lice and on their interaction with wild hosts, population level impacts of lice on wild fish, the fate, persistence and toxicity of chemical treatments for lice, and pest management strategies. The use of cleaner fish (lumpfish and wrasse) as a biological control for sea lice has increased in recent years, with over 60 million used in Norway in 2019. The sustainability of wrasse fisheries, and the genetic interaction of escaped cleaner fish with wild populations, are areas meriting further investigation (ICES, 2020). There are also mechanisms of physical control such as thermolicers, using well boats, lasers and using submersible cages with swim ups to keep salmon below the sea lice belt (Cefas, personal communication).
6.24 Finfish aquaculture can also expose wild fish to viral or bacterial pathogens, although assessing the impact on wild populations is difficult for endemic pathogens. Risk assessments, monitoring and good husbandry practices are tools for preventing and managing disease outbreaks. Data and understanding varies for different pathogens (ICES, 2020).
Antibiotics, pharmaceuticals and other toxic substances
6.25 ICES advice on interactions between aquaculture and wild fish noted that some resistance to antibiotics had been detected in bacteria. For pesticides used in aquaculture, negative impacts on non-target organisms were considered to be minor, but resistance in sea lice to some treatments had led to increased use of flubenzurones and of cleaner fish (wrasse and lumpfish). Flubenzurone residues had been found in wild crustaceans, but the significance of the impacts was not known. ICES described management options to reduce antibiotic use in fish farms, notably vaccine use, better biosecurity, reduction of waste feed, and different diets. The effectiveness of these depended on local circumstances (ICES, 2014). ICES reports that significant knowledge gaps remain relating to the persistence of therapeutic treatments, the impact of long-term or multiple exposures during the life cycle of nontarget species, and the effects of exposure to multiple medicines on non-target species and the ecosystem (ICES, 2020).
6.26 The EU TAPAS project has looked at farm-scale modelling tools for the evaluation of the ecotoxicological impacts of potentially toxic substances such as antifouling agents, veterinary medicines and other compounds. It compiled, developed, and tested environmental thresholds for potentially toxic substances used in EU aquaculture, and developed assessment tools for use by farms and regulators (EU TAPAS, 2020).
6.27 Fish feed also contains contaminants such as pesticides and mycotoxins, and heavy metals such as zinc, copper and selenium. While the levels of contaminants in fish feed and in farmed fish are monitored, regulatory limits set for fish feed have been based on fish health and food safety and not the risk of environmental effects in surrounding sediments (Grefsrud et al, 2018). ICES has recently reported that upper limits of metals have been reduced due to environmental concerns, and that regulatory limits are expected to become more stringent in future (ICES, 2020).
6.28 Reporting to OSPAR of riverine inputs and direct discharges indicates steady increases in losses of copper from aquaculture in some countries during the last ten years. Specific sources include antifoulants used in treatment of net pens (the largest contributor) and fish feed (OSPAR, 2020).
Specific impacts and measures – shellfish
6.29 Shellfish culture depends on the natural environment for the supply of feed, and might, depending on the circumstances, make a positive contribution to the provision of ecosystem services, such as through carbon sequestration and nutrient removal. Similar ecosystem service benefits can apply to the cultivation of seaweed (Gentry et al, 2020). Potential pressures do exist, however. For example, suspended shellfish culture (e.g. of mussels) can potentially lead to changes in local sediments or the water column, or, if too intensive, could strip primary production from areas being farmed. Smothering of intertidal areas with detritus could also be a problem. Collection of mussel seed for aquaculture (e.g. poorly managed dredging) can have an impact on wild mussel populations – for example, in the past seed collection was associated with declines in mussel beds in the Wadden Sea (European Commission, 2012; Common Wadden Sea Secretariat, 2010). There is also potential for impacts of shellfish aquaculture on sensitive species of birds or mammals, for example through alteration in ecosystem functioning, disturbance, exclusion, or entanglement (ICES, 2020).
6.30 Aquaculture also has the potential to introduce non-indigenous species which may affect the natural environment – the Pacific oyster, Crassostrea gigas, has established feral populations since its introduction for aquaculture in various countries in the OSPAR region (Miossec et al, 2009). A study for the Netherlands Government reported that shellfish transport had been the route for introduction of several non-indigenous species into north-west Europe (Gittenberger et al, 2017). In Scotland, the invasive sea squirt Didemnum vexillum has been recorded on oyster bags (NatureScot, 2020)6 .
6.31 Knowledge gaps and areas for future research identified by ICES include ecological carrying capacity, the impacts of different methods of bivalve culture in different environments, the use and control of non-native species, unexplained variation in the availability and settlement of seed mussels, and impacts on birds, mammals and sensitive habitats (ICES, 2020).
Recirculating Aquaculture Systems – associated environmental impacts
6.32 Given the basic premise of RAS, which aims to control inputs and outputs and uses its separation from the natural environment to enhance biosecurity and production control, environmental impacts are fewer and managed. In addition, given that a RAS presents a microcosm of the wider external environment, many of the technologies introduce design features to mitigate impacts on the ‘internal environment’ and thereby will also have a mitigating effect on their potential impact on the external environment. In this respect many of the potential impacts on the marine environment as highlighted in paragraphs 6.1 – 6.31 and in table 2 will be considerably mitigated.
6.33 While land-based aquaculture is expected to reduce negative environmental impact from escapes and sea lice, more knowledge and development is needed to reduce effluents. Normal RAS-systems are built with particle filters to reduce particulate waste material, but reduction of dissolved nutrients would incur a high cost. Effluents of dissolved nutrients (nitrogen, phosphorus) will become a major challenge in sensitive rivers, coastal waters and fjords with reduced water circulation, and will put a higher focus on recipient capacity.
6.34 For example, one of the growing benefits seen by the development of RAS systems is the advantage / benefit of near-market production. This results in shorter transportation to market and can also reduce the carbon footprint of the RAS-sourced products in the market. Traditionally, RAS have had a high energy requirement and currently the net benefit on carbon pollution is highly dependent on the nature of the energy source. Such systems are able to run very effectively from renewable sources of energy, which are bringing the balance of carbon use for RAS more in line with other production systems. In this respect, when combined with renewable energy sources (and the use of new feed technology specifically designed for use in RAS systems) RAS production is moving toward having a more sustainable carbon footprint.
6.35 As with all intensive aquaculture systems there are chances to experience significant stock losses, and increasingly aspects of animal welfare are being incorporated in future designs, recognising the importance of this issue. An issue of particular relevance to RAS is the occurrence of fish ‘off flavours’ (or tainting) which can affect the marketability of the product. The emergence over the last 20 years of an increasingly experienced and skilled RAS design and management workforce along with RAS-based research is currently addressing many of these issues.
Litter – a cross-cutting issue
6.36 Aquaculture, whether of finfish or shellfish, is a source of marine litter. OSPAR’s 2014 regional action plan on marine litter included an action to identify options to address key waste items from aquaculture. A scoping study produced in taking forward that action (OSPAR, 2019) noted that there are no good estimates of the amount of litter produced from aquaculture. The study particularly considered litter from shellfish farming, including nets, bags and other plastic equipment. A survey of OSPAR countries showed that regulation relating to waste management was included in permits to farm shellfish or fish. Awareness raising, voluntary initiatives and economic incentives, such as extended producer responsibility, were among areas recommended for further development, including through pilot projects.
6.37 The EU is supporting the Aqua-Lit project, which is working to better understand the extent of litter from aquaculture, measures to prevent and reduce it, recycling solutions for plastic waste, and associated policy mechanisms (Aqua-Lit, 2020). Approaches to tackling plastic pollution were also discussed in a report for the Aquaculture Stewardship Council (Huntington, 2019).
Conclusions
7.1 Key messages7
- while aquaculture production in the OSPAR Maritime Area has generally stabilised since the early 2010s, there are ambitions for substantial increases, notably in Norway. OSPAR may want to increase its understanding of the potential impacts of future growth, for example in relation to spatial planning, the risk of spread of non-indigenous species or parasites, or contamination of water with hazardous substances, pharmaceuticals, or nutrients;
- OSPAR may wish to consider future engagement with ICES to identify and address knowledge gaps of particular significance for environmental management of aquaculture;
- the implications of new or expanded forms of aquaculture, including development of offshore aquaculture, recirculating aquaculture systems or of new species, should also be considered;
- the viability of some aquaculture systems (e.g. shellfish farming) is dependent on wider environmental quality concerns which fall within OSPAR’s remit.
Distribution and intensity of activity
7.2 Finfish aquaculture in the OSPAR Maritime Area is dominated by salmon production. Norway is by far the largest producer of salmon in the OSPAR area (and the largest producer globally), followed in the OSPAR area by the United Kingdom and the Faroe Islands. Spain and France are globally significant producers of molluscs (mussels and oysters). (Paragraphs 2.5 - 2.7)
Trends
7.3 Production trends have varied according to species and country. Salmon production in Norway rose substantially in the years to 2012, but has stabilised since. Shellfish production fluctuated in the past decade, but there have been recent increases in Spanish mussel production. (Paragraphs 2.5, 2.6, 2.9, 4.4)
7.4 Norway has ambitious plans to increase aquaculture production including land-based production and offshore installations; other salmonid producers also have plans for growth. Some growth is also projected for other countries and aquaculture species. (Paragraphs 4.2 - 4.7)
7.5 There are prospects for aquaculture in new offshore environments and involving new species (e.g. seaweed). (Paragraphs 4.9 - 4.10)
Economic value
7.6 The economic value of the sector is dominated by Norwegian salmon, worth over €6,7 billion in 2018. For some activities, such as shellfish production in Spain and France, the sector has a significant social role as a local employer. (Paragraphs 3.1 - 3.8)
Pressures and impacts
7.7 Pressures from aquaculture can include nutrient enrichment, impacts on wild populations from escaped or introduced fish or shellfish, the transfer of parasites, diseases or non-indigenous species, the impacts of chemicals (including toxic substances from fish feed), litter, and impacts on sensitive species or ecosystems. (Paragraphs 6.1 - 6.37)
Measures
7.8 Site-specific decisions on location and management of aquaculture, via assessment of projects under individual countries’ regulatory systems, are the primary measure for addressing the environmental impacts of aquaculture. Understanding of how to manage impacts has grown over the past decade, although some knowledge gaps remain. (Paragraphs 6.1 - 6.37)
Footnotes
1 Data in figures 1-5 from FAO 2020a, and are for total North East Atlantic, including Baltic Sea
2 FAO global rankings include production in non-OSPAR regions of Spain and France
3 Figures include employment in non-OSPAR regions
4 There are also significant investments globally in recirculating aquaculture systems (RAS) for land-based production. These could feed into increases in production from existing sea sites by use of hybrid (part RAS, part sea) and shortening crop cycles in the sea (Cefas, personal communication).
5 The WFD provides for local mixing zones in relation to aquaculture facilities, where levels of priority substances are allowed to exceed environmental quality standards; this involves defining a boundary beyond which these standards must not be exceeded (European Commission, 2016).
6 D. vexillum is also spread by boating movements.
7 The views expressed on key messages are those of the assessor and do not necessarily represent the views of the OSPAR Commission
8 For the delineation of OSPAR regions see https://www.ospar.org/convention/the-north-east-atlantic
9 Low/medium/high
References
Aquaculture Industry Leadership Group 2017. Aquaculture Growth to 2030 - a strategic plan for farming Scotland’s seas. https://aquaculture.scot/wp-content/uploads/2017/11/Aquaculture_Growth_2030.pdf
Aqua-Lit 2020. Working with the aquaculture industry to prevent marine litter from entering the sea. Webpage at https://aqua-lit.eu/
Barentswatch 2020. Webpage on Sustainability in Aquaculture – Escapes Accessed May 2020. https://www.barentswatch.no/en/havbruk/escapes
Brugere, C., Aguilar-Manjarrez, J., Beveridge, M.C.M. & Soto, D. 2018. The ecosystem approach to aquaculture 10 years on – a critical review and consideration of its future role in blue growth. Reviews in Aquaculture Vol 11 Issue 3 pp 493 514. Available at: https://doi.org/10.1111/raq.12242
Campbell, I., Macleod, A., Sahlmann C., Neves, L., Funderud, J., Øverland, M., Hughes, A.D., & Stanley, M. The environmental risks associated with the development of seaweed farming in Europe – prioritizing key knowledge gaps. Frontiers in Marine Science 6:107. Available at: https://doi.org/10.3389/fmars.2019.00107
CERES 2020. CERES case studies. Available at: https://ceresproject.eu/case-studies/
Climefish 2019a. Case study fact sheet on North East Atlantic aquaculture. Horizon 2020 research and innovation programme under grant agreement 677039. Available at: https://climefish.eu/2019/04/10/north-east-atlantic-aquaculture/
Climefish 2019b. Case study fact sheet on Spanish aquaculture (Webpage) Horizon 2020 research and innovation programme under grant agreement 677039. Available at: https://climefish.eu/2019/04/10/spanish-aquaculture/
Collins, C., Bresnan, E., Brown, L., Falconer, L., Guilder, J., Jones, L., Kennerley, A., Malham, S., Murray A. & Stanley, M. 2020. Impacts of climate change on aquaculture. MCCIP Science Review 2020, 482–520. Available at: https://doi.org/10.14465/2020.arc21.aqu
Common Wadden Sea Secretariat, 2010. Wadden Sea Plan 2010. Eleventh Trilateral Governmental Conference on the Protection of the Wadden Sea. Common Wadden Sea Secretariat, Wilhelmshaven, Germany. Available at: https://www.waddensea-worldheritage.org/sites/default/files/2010_Wadden%20Sea%20Plan.pdf
Costello, C., Cao, L., Gelcich, S. et al. The future of food from the sea. Nature (2020). Available at: https://doi.org/10.1038/s41586-020-2616-y
Department of Agriculture, Food and the Marine 2015. National Strategic Plan for Sustainable Aquaculture Development. Available at: https://www.agriculture.gov.ie/media/migration/seafood/marineagenciesandprogrammes/nspa/NationalStrategicPlanSusAquaDevel181215.pdf
European Commission 2012. Guidance Document on Aquaculture and Natura 2000. Available at: https://ec.europa.eu/fisheries/sites/fisheries/files/docs/body/guidance-aquaculture-natura2000.pdf
European Commission 2013. Strategic Guidelines for the sustainable development of EU aquaculture. COM/2013/229 Available at: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52013DC0229&from=EN
European Commission 2016. Commission Staff Working Document on the application of the Water Framework Directive (WFD) and the Marine Strategy Framework Directive (MSFD) in relation to aquaculture. SWD(2016) 178 final. Available at: https://ec.europa.eu/fisheries/sites/fisheries/files/docs/body/swd-2016-178_en.pdf
European Commission 2019. The EU Blue Economy Report. 2019. Publications Office of the European Union. Luxembourg. Available at: https://op.europa.eu/en/publication-detail/-/publication/676bbd4a-7dd9-11e9-9f05-01aa75ed71a1/language-en/
European Commission 2020. The EU Blue Economy Report. 2020. Publications Office of the European Union. Luxembourg. Available at: https://ec.europa.eu/maritimeaffairs/sites/maritimeaffairs/files/2020_06_blueeconomy-2020-ld_final.pdf
European Environment Agency 2019. Aquaculture production in Europe Fig 7. Available at: https://www.eea.europa.eu/data-and-maps/indicators/aquaculture-production-4/assessment
Eurostat 2019. Agriculture, forestry and fishery statistics 2019. Publications office of the European Union Luxembourg. doi 10.2785/798761 Available at: https://ec.europa.eu/eurostat/documents/3217494/10317767/KS-FK-19-001-EN-N.pdf/742d3fd2-961e-68c1-47d0-11cf30b11489
Eurostat 2020. Online data code fish_aq2a Production from aquaculture excluding hatcheries and nurseries (webpage accessed May 2020). Available at: https://appsso.eurostat.ec.europa.eu/nui/show.do?dataset=fish_aq2a&lang=en
EU TAPAS 2020. Tools for assessment and planning of aquaculture sustainability (TAPAS). Funded under EU Horizon 2020 research and innovation programme, grant agreement 678396. Available at: http://tapas-h2020.eu/
FAO 2018. Impacts of climate change on fisheries and aquaculture. ISBN 978-92-5-130607-9. Available at: http://www.fao.org/3/i9705en/i9705en.pdf
FAO 2019. Bivalve production increasing due to positive demand patterns. FAO Globefish Market Report 03/12/2019 (Webpage). Available at: http://www.fao.org/in-action/globefish/market-reports/resource-detail/en/c/1253633/
FAO 2020a. Fisheries Statistical Collections: Global Aquaculture Production (Webpage accessed 17/06/2020). Available at: http://www.fao.org/fishery/statistics/global-aquaculture-production/en
FAO 2020b. The State of World Fisheries and Aquaculture 2020. Sustainability in action. Rome. Available at: https://doi.org/10.4060/ca9229en
FAO 2020c. GLOBEFISH Highlights January 2020 issue with Jan. – Sep. 2019 statistics – a quarterly update on world seafood markets. Globefish Highlights no. 1–2020. Rome. Available at: https://doi.org/10.4060/ca7968en
Froehlich, H. E., Couture, J., Falconer, L., Krause, G., Morris, J. A., Perez, M., Stentiford, G. D., Vehvila¨inen, H., and Halpern, B. S. 2020. Mind the gap between ICES nations’ future seafood consumption and aquaculture production. ICES Journal of Marine Science. Available at: https://doi:10.1093/ icesjms/fsaa066
FutureEUAqua 2020. Future growth in sustainable, resilient and climate friendly organic and conventional European aquaculture. Available at: https://futureeuaqua.eu/
Gittenberger, A., Rensing, M. & Wesdorp, K.H. 2017. Uitheemse mariene soorten in Nederland; GiMaRIS 2017. Available at: https://docplayer.nl/107231806-Uitheemse-mariene-soorten-in-nederland.html
Government of Iceland 2020 . Aquaculture. Ministry of Industries and Innovation. Webpage accessed 22/06/20. Available at: https://www.government.is/topics/business-and-industry/fisheries-in-iceland/aquaculture/
Grefsrud, E.S., Glover, K., Grøsvik, B.E., Husa, V., Karlsen, Ø., Kristiansen, T., Kvamme, B.O., Mortensen, S., Samuelsen, O.B., Stien, L.H., & Svåsand, T. (red.) (2018). Risikorapport norsk fiskeoppdrett 2018. Fisken og havet, særnr. 1-2018. 10137000. Available at: Risikorapport norsk fiskeoppdrett 2018 | Havforskningsinstituttet (hi.no)
Huntington, T. 2019. Marine Litter and Aquaculture Gear – White Paper. Report produced by Poseidon Aquatic Resources Management Ltd for the Aquaculture Stewardship Council. 20 pp plus appendices. Available at: https://www.asc-aqua.org/wp-content/uploads/2019/11/ASC_Marine-Litter-and-Aquaculture-Gear-November-2019.pdf
ICES 2014. Special request July 2014. OSPAR request on interactions between wild and captive fish stocks. Available at: https://www.ices.dk/sites/pub/Publication%20Reports/Advice/2014/Special%20Requests/OSPAR_%20Interactions_of_wild_and_captive_fish_stocks.pdf
ICES 2019 Barents Sea Ecoregion – Ecosystem overview. In Report of the ICES Advisory Committee, 2019. ICES Advice 2019, Section 5.1. Available at: https://doi.org/10.17895/ices.advice.5747
ICES 2020. Working Group on Environmental Interactions of Aquaculture (WGEIA). ICES Scientific Reports. 2:112. 187 pp. Available at: http://doi.org/10.17895/ices.pub.7619
Jeffery, K.R., Vivian, C.M.G., Painting, S.J., Hyder, K., Verner-Jeffreys, D.W., Walker, R.J., Ellis, T., Rae, L.J., Judd, A.D., Collingridge, K.A., Arkell, S., Kershaw, S.R., Kirby, D.R., Watts, S., Kershaw, P.J., & Auchterlonie, N.A. 2014. Background information for sustainable aquaculture development, addressing environmental protection in particular. Available at: https://ec.europa.eu/environment/enveco/water/pdf/SUSAQ%20Final%20Report%20Part%201.pdf
Jeffery, K.R., McPherson, N., Verner-Jeffreys, D., Taylor, N. & Auchterlonie, N. 2015. Modelling of the potential for shortening the pen-based phase of the salmon ongrowing cycle. A study commissioned by the Scottish Aquaculture Research Forum (SARF). Available at: http://www.SARF.org/
Marine Scotland 2015. A Technical Standard for Scottish Finfish Aquaculture. ISBN: 978-1-78544-372-5. Available at: https://www.gov.scot/binaries/content/documents/govscot/publications/advice-and-guidance/2015/06/technical-standard-scottish-finfish-aquaculture/documents/00479005-pdf/00479005-pdf/govscot%3Adocument/00479005.pdf
Marine Scotland 2019. The Regulation of Sea Lice in Scotland. Available at: https://www.gov.scot/binaries/content/documents/govscot/publications/transparency-data/2019/11/fish-health-inspectorate-sea-lice-information/documents/sea-lice-regulation-topic-sheet/sea-lice-regulation-topic-sheet/govscot%3Adocument/sea%2Blice%2Btopic%2Bsheet.pdf
Miossec, L., Le Deuff, R-M. & Goulletquer, P. 2009. Alien species alert: Crassostrea gigas (Pacific oyster). ICES Cooperative Research Report No, 299, 42pp. Available at: https://www.ices.dk/sites/pub/Publication%20Reports/Cooperative%20Research%20Report%20(CRR)/CRR299.pdf
NASCO 2016. Addressing impacts of salmon farming on wild Atlantic salmon: Challenges to, and developments supporting, achievement of NASCO’s international goals. 2016. Report of a Theme-based Special Session of the Council of NASCO. NASCO Council document CNL(16)60. 196pp. Available at: http://www.nasco.int/pdf/reports_other/2016ThemeBasedSession.pdf
NatureScot 2020. Monitoring for the possible spread of Didemnum vexillum into the subtidal environment of Loch Creran. NatureScot Research Report 1155. Available at: https://www.nature.scot/naturescot-research-report-1155-monitoring-possible-spread-didemnum-vexillum-subtidal-environment
Norwegian Ministry of Trade, Industry and Fisheries/Norwegian Ministry of Petroleum and Energy 2017. New Growth, Proud History: The Norwegian Government’s Ocean Strategy. Available at: https://www.regjeringen.no/contentassets/00f5d674cb684873844bf3c0b19e0511/the-norwegian-governments-ocean-strategy---new-growth-proud-history.pdf
Norwegian Ministries 2019. Blue Opportunities: The Norwegian Government’s Updated Ocean Strategy. Available at: https://www.regjeringen.no/globalassets/departementene/nfd/dokumenter/strategier/w-0026-e-blue-opportunities_uu.pdf
Norwegian Ministry of Trade, Industry and Fisheries 2019. Webpage article on Farmed Salmon. Updated November 2019. Available at: https://www.regjeringen.no/en/topics/food-fisheries-and-agriculture/fishing-and-aquaculture/1/farmed-salmon/id495344/
O’Hagan, A.M., Corner, R.A., Aguilar-Manjarrez, J. Gault, J., Ferreira, R.G., Ferreira, J.G., O’Higgins, T., Soto, D., Massa, F., Bacher, K., Chapela, R. & Fezzardi, D. 2017. Regional review of Policy Management Issues in Marine and Freshwater Aquaculture. Report produced as part of the Horizon 2020 AquaSpace project. 170pp. Available at: http://www.aquaspace-h2020.eu/wp-content/uploads/2017/10/Regional-Review-of-Policy-Management-Issues-in-Marine-and-Freshwater-Aquaculture.pdf
OSPAR 2009. Assessment of Impacts of Mariculture. ISBN 978-1-906840-82-2 Publication Number: 442/2009. Available at: https://qsr2010.ospar.org/media/assessments/p00442_Impacts_of_Mariculture.pdf
OSPAR 2016. OSPAR Recommendation 2016/03 on furthering the protection and conservation of the Atlantic salmon (Salmo salar) in Regions I, II, III and IV of the OSPAR maritime area. Available at: https://www.ospar.org/convention/agreements?q=salmon&t=&a=&s=
OSPAR 2017. Third Integrated Report on the Eutrophication Status of the OSPAR Maritime Area. ISBN: 978-1-911458-34-0 Publication Number: 694/2017. Available at: https://www.ospar.org/documents?v=37502.
OSPAR 2018. Guidelines for Harmonised Quantification and Reporting Procedures for Nutrients (HARP-NUT). Guideline 2: Quantification and Reporting of Nitrogen and Phosphorus Discharges/losses from Aquaculture Plants. Available at: https://www.ospar.org/convention/agreements?q=aquaculture&t=&a=&s=
OSPAR 2019. Scoping study to identify key waste items from the fishing industry and aquaculture. ISBN 978-1-911458-73-9 Publication Number: 733/2019. Available at: https://www.ospar.org/documents?v=41242
OSPAR (2020). Comprehensive Study on Riverine Inputs and Direct Discharges (RID). Available at: https://oap.ospar.org/en/ospar-assessments/committee-assessments/hazardous-substances-and-eutrophication/input/riverine-inputs-and-direct-discharges/ospar-contracting-parties-rid-data-report/riverine-inputs-and-direct-discharges-2018/
Peck M.A., Catalán I.A., Damalas D., Elliott M., Ferreira J.G., Hamon K.G., Kamermans P., Kay S., Kreiß C.M., Pinnegar J.K., Sailley S.F. & Taylor N.G.H. 2020. Climate Change and European Fisheries and Aquaculture: ‘CERES' Project Synthesis Report. Hamburg. Available at https//doi.org/10.25592/uhhfdm.804
Prevent Escape 2013. Final Report Summary - Assessing the causes and developing measures to prevent the escape of fish from sea-cage aquaculture. Grant agreement ID 226885 Funded under FP7-KBBE. Available at: https://cordis.europa.eu/project/rcn/90974/brief/en
Scientific, Technical and Economic Committee for Fisheries (STECF)(2018). Economic Report of the EU Aquaculture sector. Publications Office of the European Union, Luxembourg, 2018, ISBN 978-92-79-79402-5. doi:10.2760/45076, JRC114801. Available at: https://op.europa.eu/en/publication-detail/-/publication/7f9c98f0-0fe4-11e9-81b4-01aa75ed71a1
Scotland’s Aquaculture 2020. Data on fish escapes. Website accessed 27 August 2020. Available at: http://aquaculture.scotland.gov.uk/data/fish_escapes.aspx
Seafish 2018. Seafood 2040 A strategic framework for England. Available at: https://www.seafish.org/document/?id=98F10916-276C-414C-84E7-F6870F9CD417
Statistics Norway 2020 .Aquaculture Webpage accessed 03/09/2020. Available at: https://www.ssb.no/en/jord-skog-jakt-og-fiskeri/statistikker/fiskeoppdrett
Wallhead, P. Ciavatta, S. Kay, S. Palmer, S. Barillé, L. Gernez, P. & Tsiaras, K. 2020. Future scenarios maps and documentation for WP8. Deliverable 6.4 Report. 53 pp Part of EU Tools for Assessment and Planning for Aquaculture Sustainability (TAPAS). tapas-h2020.eu/results/deliverable-6-4-future-scenarios-maps-and-documentation-for-toolbox/



