Status Assessment 2020 - European flat oyster and Ostrea edulis beds
| Assessment of status | Distribution | Population size / Extent | Condition | Previous OSPAR status assessment | European flat oyster | Ostrea edulis beds | Status | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
Region | I | ○ | NA | |||||||||
| II | ? | 1,2,5 | ? | 1,2,5 | ? | 1,2,5 | ● | ● | ? | 1,2,5 | ||
| III | ↓ | 1,2,5 | ↓ | 1,2,5 | ↓ | 1,2,5 | ○ | ● | Poor, ↓ | 1,2,5 | ||
| IV | ↓ | 1,2,5 | ↓ | 1,2,5 | ↓ | 1,2,5 | ○ | ● | Poor, ↓ | 1,2,5 | ||
| V | NA | |||||||||||
Explanation to table:
Distribution, Population size, Condition
Trends in status (since the assessment in the background document)
↓ decreasing trend or deterioration of the criterion assessed
↑ increasing trend or improvement in the criterion assessed
←→ no change observed in the criterion assessed
? trend unknown in the criterion assessed
Previous status assessment: if QSR 2010 then entry Regions where species occurs ( ○) and has been recognised by OSPAR to be threatened and/or declining (● ) based on Chapter 10 Table 10.2 and Table 10.3 . If a more recent status assessment is available, then enter ‘poor’/’good’
Status*(overall assessment))
red – poor
green – good
blue cells? - status unknown, insufficient information available
NA - Not Applicable
*applied to assessments of status of the feature or of a criterion, as defined by the assessment values used in the QSR 2023 or by expert judgement.
Key Pressures
↓ key pressures and human activities reducing
↑ key pressures and human activities increasing
←→ no change in key pressures and human activities
? Change in pressure and human activities uncertain
Threat or impacts (overall assessment)
red – significant threat or impact
green –no evidence of a significant threat or impact
blue cells – insufficient information available
NA – not applicable
1 – direct data driven
2 – indirect data driven
3 – third party assessment, close-geographic match
4 – third party assessment, partial-geographic match
5 – expert judgement
| Assessment of key pressures | Removal of (non-) target species (i.e. fisheries) | Introduction of non-indigenous species and translocations | Microbial pathogens | Habitat damage | Hazardous substances | Threat or impact | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Region | I | ||||||||||||||
| II | ←→ | 1,2,4,5 | ↑ | 1,2,3,4,5 | ? | 1,5 | ←→ | 1,2,5 | ? | 5 | ? | 1,2,3,4,5 | |||
| III | ←→ | 5 | ↑ | ←→ | 1,5 | ←→ | 1 | ↑ | 1,2,5 | ? | 3 | ←→ | 1,2,3,5 | ||
| IV | ←→ | 5 | ↑ | ←→ | 1,5 | ←→ | 1 | ↑ | 1,2,5 | ? | 3 | ←→ | 1,2,3,5 | ||
| V | |||||||||||||||
Confidence
Moderate to high confidence in the status assessment because of quantitative indicators for Region III and IV. In Region II, the expert judgement varies considerably among the participating countries, resulting in a poor evidence of status.
Moderate confidence in the threat assessment because some impacts (e.g. by hazardous substances) are hardly investigated or not sufficiently monitored, resulting in lacking data for some threats in certain regions.
Background Information
Year added to OSPAR List: 2003
Decline: Until the 19th century, large coastal and offshore O. edulis beds existed along the coasts in north western European waters, although they probably did not form a continuous zone. They declined or have been largely lost since (OSPAR Commission 2009).
Sensitivity: O. edulis is highly sensitive to substrate loss, smothering, silt deposition, synthetic compound contamination, introduction of microbial pathogens/parasites, introduction of non-native species and direct extraction. Recoverability is considered to be very low, presumably it takes more than twenty years to re-build a reef structure.
Last status assessment: OSPAR (2009) reported no changes in the evaluation against the Texel-Faial selection criteria.
Geographical Range and Distribution
The distribution of O. edulis beds is shown in Figure 1. The European flat oyster is native to the North-East Atlantic in the OSPAR area, naturally inhabiting intertidal to subtidal depths from Norway to Morocco in the Atlantic and the North Sea (Lapègue et al. 2006, Pogoda 2019). Depleted remnant oyster populations are present in UK, Ireland, France, the Netherlands, Denmark, Portugal and Spain. Oysters are functionally extinct in the German and Belgian North Sea. At the west coast of Sweden and the coast of southern Norway larger wild populations of O. edulis can still be found.
Historically, extensive offshore native oyster beds existed in the North Sea until the 19th century, probably never covering a continuous zone. Nowadays these extensive oyster beds have been lost to great extent. Owing to substantial restoration efforts in many European countries, remnant oyster beds are supported and oysters will be re-introduced in areas formerly populated by O. edulis.
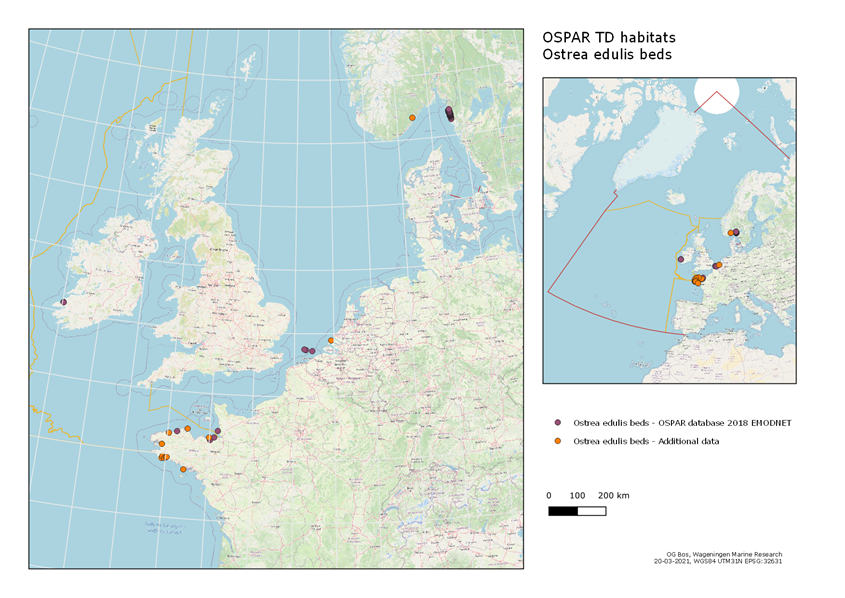
Figure 1: Known distribution of O. edulis beds. Data obtained from OSPAR database, with additional data provided by France and Norway (Please note that UK data points for this habitat are restricted).
Population/Abundance
In some countries of the OSPAR area the European flat oyster is considered virtually extinct (Region II: NL, BE, DE), while in other areas the trends in relative abundance are considered stable or unknown (Region II: NO, SE) or even still decreasing in distribution, extent and condition (Region III & IV: FR).
Oyster abundance varies between countries and even sites, with the French population in northern Brittany occurring in very low abundances (below 1 oyster/m2, Pouvreau et al., 2019), while some Norwegian fjord patches in Agder county show densities greater than 50 oysters/m2 (Bodvin 2011). Commercial harvesting still impacts remaining populations in some countries (e.g. FR, UK, IK), while restoration and protection measures increase populations in other areas (e.g. NL, DE). Overall population estimates of the respective countries are rarely found (for SE: 36,6 ± 16.3 million individuals (total population ± SE), (Thorngren et al. 2019)).
Condition
The production of natural flat oyster spat in Europe has decreased almost by 60% in the past ten years (Maneiro et al. 2017). Diseases (mainly bonamiosis, marteiliosis) are found in O. edulis populations along the European Atlantic coast in the North Sea and in the Mediterranean basin. While some stocks already are negatively impacted by Bonamia ostreae (e.g. DK, ES, FR, NL, UK), other waters (SE, NO) are still considered Bonamia-free, screened by a regular monitoring. The parasite Marteilia refringens is reported in flat oyster populations in several countries (e.g. ES, FR, GR, IT) and can be associated with mortality.
Regarding the genetic differentiation of wild flat oyster populations regions three clusters i.e. Spanish, Irish/British/French, and Dutch/Danish cluster have been identified (Vera et al. 2016).
Threats and Impacts
Several factors have determined the decline of wild flat oyster populations down to marginal figures in many European areas: overexploitation of natural beds; introduction of non‐native oysters; adverse effects of climate change and specific epidemic outbreaks (Vera et al. 2019). However, besides these also microplastics are found to impact health and biological functioning of European flat oysters and the associated macrofauna (Green 2016), while alien species could also have negative effects like increased competition, predation or disease transfer. Pressures caused by predators especially Sea Bream, Starfish, Crustaceans, Oyster driller (Pouvreau et al., 2019) pose an additional threat on oyster populations. Habitat destruction and the loss of oyster beds due to bottom fishing are still relevant in some areas. Further, the lack of wild donor populations as well as the lack of suitable substrate hamper the recovery of most oyster populations within the OSPAR area (Pogoda et al. 2019).
Measures that address key pressures from human activities or conserve the species/habitat
The European flat oyster historically was and still is subject to husbandry and cultivation practices (Colsoul et al. 2020) as well as fisheries target, which needs to be considered when developing management measures to mitigate key pressures.
Successful management therefore includes the regulation and control of fisheries (e.g. seasonal restrictions, quotas, minimum landing size, stock surveys), the limitation of the propagation of non-indigenous species, the diminution of pathogen and parasite transmission as well as measures to introduce suitable substrate and to preserve or restore suitable habitat for reproduction and successful spatfall (OSPAR 2009, Pogoda et al. 2019). Other useful measures could include the allocation of broodstock areas, the establishment of marine protected areas in regions with known severe damage/ loss or the restoration of oysters to promote recovery of the habitat (OSPAR 2009). For all these mitigation measures, a comprehensive, suitable long-term monitoring is indispensable to assess habitat quality and the development of the oyster beds over time (Zu Ermgassen et al., 2020).
Conclusion (including management considerations)
Oyster habitats are hot spots of biodiversity and are vital to the health of the surrounding ecosystem. As an ecological keystone species O. edulis offers substrate, spawning ground, food and shelter for many more species. In Europe, native oysters once formed extensive beds and reefs along North Atlantic coastlines and in the North Sea, even in offshore regions of moderate depth. High fishing pressure and poor shell management practices not only resulted in the decline of living oysters, but also in the loss of the most important natural settlement substrate for oyster larvae: oyster shells. In waters with sufficient larval abundances, the lack of suitable substrate can be the limiting factor for the recovery of oyster populations. Throughout much of Europe, a lack of broodstock in sufficiently high density for successful breeding is another reason for low larval abundances. Furthermore, in many European ecoregions the invasive protozoan parasite Bonamia ostreae increases mortality of native oyster populations. The Oslo-Paris-Commission (OSPAR) included O. edulis on the list of threatened and declining species and habitats, for which restoration measures should be developed and the EU Habitats Directive calls for the protection, conservation and restoration of biogenic reefs, such as native oyster reefs.
Against this background, ecological restoration measures are crucial to support the recovery of self-sustaining oyster habitats. Across Europe, a number of restoration projects are in the process of being developed and implemented (see https://noraeurope.eu ). To ensure long-term preservation of the species and the habitat it creates, including the related positive ecological effects, dynamic steps for upscaling oyster restoration have to be undertaken.
Knowledge Gaps
Many fundamental ecological questions (see Zu Ermgassen et al. 2020) like the interactions of O. edulis with predators and competitors need to be investigated more comprehensively, but also research on factors and requirements for successful settlement and recruitment of wild populations (e.g. critical weight, settlement substratum characteristics) is needed (Allison 2019, Colsoul et al. 2020), as well as information on environmental factors influencing these processes.
Further investigations are also needed in the field of marine conservation to better understand factors (environmental parameters, biological interactions) determining the success of restoration measures (Zu Ermgassen et al. 2020). Limitations also exist regarding the monitoring of native oyster beds for many areas, but also on the interaction with non-indigenous species, effects of diseases and infections and the effects of hazardous substances.
Allison S (2019) The endangered European native oyster Ostrea edulis (L) and creation of Marine Conservation Zones: a win – win scenario for fisheries and conservation? University of Essex
Allison S, Hardy M, Hayward K, Cameron TC, Underwood GJC (2020) Strongholds of Ostrea edulis populations in estuaries in Essex, SE England and their association with traditional oyster aquaculture: Evidence to support a MPA designation. J Mar Biol Assoc United Kingdom 100:27–36.
Alvarez Alonso C and Foster S (2019). Strangford Lough Special Area of Conservtion (SAC) condition assessment. Department of Agriculture, Environment and Rural Affairs. Northern Ireland.
Barnard S&, Strong J (2014) Reviewing, refining and identifying optimum aggregation methods for undertaking marine biodiversity status assessments.
BfN (2020) Wiederansiedlung der Europäischen Auster: Erstes Riff in der deutschen Nordsee angelegt (Press release). https://www.bfn.de/presse/pressearchiv/2020/detailseite.html?tx_ttnews%5Btt_news%5D=6966&cHash=eb25bb2c05ccda399e466b04ce56fdb8 (accessed 21 September 2020)
Berthe FCJ, Le Roux F, Adlard RD and Figueras A (2004) Marteiliosis in molluscs: a review. Aquatic Living Resources 17, 433–448.
Bodvin T (2011) Utredning av europeisk flatøsters Ostrea edulis L. – Kunnskapsoversikt med forslag til handlingsplan. UN-utredning 10-2011.
Borja A, Elliott M, Andersen JH, Cardoso AC, Carstensen J, Ferreira JG, Heiskanen A-S, Marques JC, Neto JM, Teixeira H, Uusitalo L, Uyarra MC, Zampoukas N (2013) Good Environmental Status of marine ecosystems: What is it and how do we know when we have attained it? Mar Pollut Bull 76:16–27.
Borja A, Prins TC, Simboura N, Andersen JH, Berg T, Marques JC, Neto JM, Papadopoulou N, Reker J, Teixeira H, Uusitalo L (2014) Tales from a thousand and one ways to integrate marine ecosystem components when assessing the environmental status. Front Mar Sci 1.
Bromley C, McGonigle C, Ashton EC, Roberts D (2016) Bad moves: Pros and cons of moving oysters - A case study of global translocations of Ostrea edulis Linnaeus, 1758 (Mollusca: Bivalvia). Ocean Coast Manag 122:103–115.
Christianen MJA, Lengkeek W, Bergsma JH, Coolen JWP, Didderen K, Dorenbosch M, Driessen FMF, Kamermans P, Reuchlin-Hugenholtz E, Sas H, Smaal A, van den Wijngaard KA, van der Have TM (2018) Return of the native facilitated by the invasive? Population composition, substrate preferences and epibenthic species richness of a recently discovered shellfish reef with native European flat oysters ( Ostrea edulis ) in the North Sea. Mar Biol Res 14:590–597.
Colsoul B, Pouvreau S, Di Poi C, Pouil S, Merk V, Peter C, Boersma M, Pogoda B (2020) Addressing critical limitations of oyster (Ostrea edulis) restoration: identification of nature-based substrates for hatchery production and recruitment in the field. Aquat Conserv Mar Freshw Ecosyst.
Didderen K, Lengkeek W, Bergsma JH, Dongen U Van (2020) WWF & ARK Borkum Reef Ground oyster pilot. Culemborg.
Didderen K, Lengkeek W, Kamermans P, Deden B, Reuchlin-Hugenholtz E, Bergsma JH, ..., Sas H (2019) Pilot to actively restore native oyster reefs in the North Sea. Bureau Waardenburg, Culemborg. WWF, Zeist.
Eagling LE, Ashton EC, Jensen AC, Sigwart JD, Murray D, Roberts D (2018) Spatial and temporal differences in gonad development, sex ratios and reproductive output, influence the sustainability of exploited populations of the European oyster, Ostrea edulis. Aquat Conserv Mar Freshw Ecosyst 28:270–281.
Ermgassen, 2017. Milford Haven Native Oyster Regeneration Project - Stage One (current status and practicalities). A report commissioned by West Wales Shellfishermans Association Ltd supported by the Milford Haven Native Oyster Regeneration Management Group.Fariñas-Franco JM, Pearce B, Mair JM, Harries DB, MacPherson RC, Porter JS, Reimer PJ, Sanderson WG (2018) Missing native oyster (Ostrea edulis L.) beds in a European Marine Protected Area: Should there be widespread restorative management? Biol Conserv 221:293–311.
Green DS (2016) Effects of microplastics on European flat oysters, Ostrea edulis and their associated benthic communities. Environ Pollut 216:95–103.
Guy C, Smyth D, Roberts D (2019) The importance of population density and inter-individual distance in conserving the European oyster Ostrea edulis. J Mar Biol Assoc United Kingdom 99:587–593.
Harrang E., Heurtebise S., Faury N., Robert M., Arzul I., Lapègue S., 2015. Can survival of European flat oysters following experimental infection with Bonamia ostreae be predicted using QTLs? Aquaculture 448, 521-530.
Hayer S, Bick A, Brandt A, Ewers-Saucedo C, Fiege D, Füting S, Krause-Kyora B, Michalik P, Reinicke G-B, Brandis D (2019) Coming and going – Historical distributions of the European oyster Ostrea edulis Linnaeus, 1758 and the introduced slipper limpet Crepidula fornicata Linnaeus, 1758 in the North Sea. PLoS One 14:e0224249.
Helmer L, Farrell P, Hendy I, Harding S, Robertson M, Preston J (2019) Active management is required to turn the tide for depleted Ostrea edulis stocks from the effects of overfishing, disease and invasive species. PeerJ 2019:e6431.
Jenkins C, Eggleton J, Barry J, O’Connor J (2018) Advances in assessing Sabellaria spinulosa reefs for ongoing monitoring. Ecol Evol 8:7673–7687.
Kamermans P, Walles B, Kraan M, van Duren L, Kleissen F, van der Have T, Smaal A, Poelman M (2018) Offshore Wind Farms as Potential Locations for Flat Oyster (Ostrea edulis) Restoration in the Dutch North Sea. Sustainability 10:3942.
Kerckhof F, Coolen JWP, Rumes B, Degraer S (2018) Recent findings of wild European flat oysters Ostrea edulis (Linnaeus, 1758) in Belgian and Dutch offshore waters: new perspectives for offshore oyster reef restoration in the southern North Sea. Belgian J Zool 148:13–24.
Kregting L, Hayden-Hughes M, Millar R. V,. Joyce P. W.S, Smyth D. M (2020). A first record of intertidal Ostrea edulis 3D structural matrices in Strangford Lough Northern Ireland - An emergent reef?. Journal of Sea Research, Volume 163, 2020, 101927, ISSN 1385-1101Lapègue S, Beaumont A, Boudry P, Goulletquer P (2006) European flat oyster-Ostrea edulis.
Maneiro V, Silva A, Pazos AJ, Sánchez JL, Pérez-Parallé ML (2017) Effects of temperature and photoperiod on the conditioning of the flat oyster ( Ostrea edulis L.) in autumn. Aquac Res 48:4554–4562.
Mortensen, S. H., Skår, C., Sælemyr, L. (2020) Summarizing the screening for Bonamia ostreae in Norwegian populations of flat oysters, Ostrea edulis. Bergen.
Mortensen, Stein. Hald, Fredrik. Klakegg, Øystein. Ronold A (2017) Østers! : fra råvare til servering : den norske østersdyrkingens historie. Vigmostad Bjørke.
Mortensen S, Bodvin T, Strand Å, Holm MW, Dolmer P (2017) Effects of a bio-invasion of the pacific oyster, crassostrea gigas (Thunberg, 1793) in five shallow water habitats in scandinavia. Manag Biol Invasions.
Mortensen, S., Skår, C.K., Sælemyr, L., Bøgwald, M., Jelmert, A. (2019) The surveillance and control programme for bonamiosis and marteiliosis in European flat oysters, Ostrea edulis, and blue mussels, Mytilus sp. in Norway in 2018. Rapport fra havforskningen nr 17, 2019, 16 s.
Mortensen SH (1993) A health survey of selected stocks of commercially exploited Norwegian bivalve molluscs. Dis Aquat Organ.
Nielsen P, Petersen JK (2019) Flat oyster fishery management during a time with fluctuating population size. Aquat Living Resour 32:22.
OIE (2015) Disease information on the infection with Bonamia ostreae in wild molluscs in 2015 (WAHIS Interface). https://www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/Diseaseoutbreakmaps?disease_type_hidden=&disease_id_hidden=&selected_disease_name_hidden=&disease_type=1&disease_id_terrestrial=-999&disease_id_aquatic=167&speciesselect%5B%5D=25&submit1=OK&s
OSPAR Commission (2009) OSPAR Background Document for Ostrea edulis and Ostrea edulis beds. Biodiversity Series. OSPAR, London, UK.
Pogoda B (2019) Current Status of European Oyster Decline and Restoration in Germany. Humanities 8:9.
Pogoda B, Brown J, Hancock B, Nordheim H Von (2017) Berlin oyster recommendation on the future of native oyster restoration in Europe bringing back an ecological key player: principles for native oyster restoration in Europe.
Pogoda B, Brown J, Hancock B, Preston J, Pouvreau S, Kamermans P, Sanderson W, von Nordheim H (2019) The Native Oyster Restoration Alliance (NORA) and the Berlin Oyster Recommendation: bringing back a key ecosystem engineer by developing and supporting best practice in Europe. Aquat Living Resour 32:13.
Pogoda B, Peter C, Merk V, Colsoul B, Holler P, Bartholomä A, Jaklin S, Michaelis R, Kramer M, Pesch R, Prinz K (2020) Site selection for biogenic reef restoration in offshore environments: The Natura 2000 site Borkum Reefground as a case study for native oyster restoration. Aquat Conserv Mar Freshw Ecosyst.
Pouvreau S, Cochet H, Gachelin S, Chaudemanche S, Fabien A, Petton S, Brossin A, Guillet T, Le Roy V, Huber M, Queau I, Di Poï C, Mérou N, Arzul I, Lecadet C, Bouget JF, Cochennec N, Boudry P, Sebaibi : N, Dufeu M, Crc AG, Nord B, Gonzalez : R, Salaun B (2019) Inventaire, diagnostic écologique et restauration des principaux bancs d’huitres plates en Bretagne : le projet FOREVER Rapport intermédiaire 2018 Contribution : Ifremer 1.
Probst WN, Lynam CP (2016) Integrated assessment results depend on aggregation method and framework structure – A case study within the European Marine Strategy Framework Directive. Ecol Indic 61:871–881.
Ramilo A, González M, Carballal MJ, Darriba S, Abollo E, Villalba A (2014) Oyster parasites Bonamia ostreae and B. exitiosa co-occur in Galicia (NW Spain): spatial distribution and infection dynamics. Dis Aquat Organ 110:123–133.
Rowley AF, Cross ME, Culloty SC, Lynch SA, Mackenzie CL, Morgan E, O’Riordan RM, Robins PE, Smith AL, Thrupp TJ, Wootton EC, Malham SK (2014) The potential impact of climate change on the infectious diseases of commercially important shellfish populations in the Irish Sea - A review. ICES J Mar Sci 71:741–759.
Sawusdee A, Jensen AC, Collins KJ, Hauton C (2015) Improvements in the physiological performance of European flat oysters Ostrea edulis (Linnaeus, 1758) cultured on elevated reef structures: Implications for oyster restoration. Aquaculture 444:41–48.
Smyth D, Mahon AM, Roberts D, Kregting L (2018) Settlement of Ostrea edulis is determined by the availability of hard substrata rather than by its nature: Implications for stock recovery and restoration of the European oyster. Aquat Conserv Mar Freshw Ecosyst 28:662–671.
Smyth D, Roberts D, Browne L (2009) Impacts of unregulated harvesting on a recovering stock of native oysters (Ostrea edulis). Mar Pollut Bull 58:916–922.
Strand Å, Blanda E, Bodvin T, Davids JK, Jensen LF, Holm-Hansen TH, Jelmert A, Lindegarth S, Mortensen S, Moy FE, Nielsen P, Norling P, Nyberg C, Christensen HT, Vismann B, Holm MW, Hansen BW, Dolmer P (2012) Impact of an icy winter on the Pacific oyster (Crassostrea gigas Thunberg, 1793) populations in Scandinavia. Aquat Invasions.
Thorngren L, Bergström P, Dunér Holthuis T, Lindegarth M (2019) Assessment of the population of Ostrea edulis in Sweden: A marginal population of significance? Ecol Evol 9:13877–13888.
Vera M, Carlsson J, Carlsson J El, Cross T, Lynch S, Kamermans P, Villalba A, Culloty S, Martinez P (2016) Current genetic status, temporal stability and structure of the remnant wild European flat oyster populations: conservation and restoring implications. Mar Biol 163:239.
Vera M, Pardo BG, Cao A, Vilas R, Fernández C, Blanco A, Gutierrez AP, Bean TP, Houston RD, Villalba A, Martínez P (2019) Signatures of selection for bonamiosis resistance in European flat oyster ( Ostrea edulis ): New genomic tools for breeding programs and management of natural resources. Evol Appl 12:1781–1796.
Zapata-Restrepo LM, Hauton C, Williams ID, Jensen AC, Hudson MD (2019) Effects of the interaction between temperature and steroid hormones on gametogenesis and sex ratio in the European flat oyster (Ostrea edulis). Comp Biochem Physiol Part A Mol Integr Physiol 236:110523.
zu Ermgassen P, Bonacic K, Boudry P, Bromley C, Cameron T, Colsoul B, Coolen JWP, Frankic A, Hanckock B, van der Have TM, Holbrooke Z, Kamermans P, Laugen A, Nevejan N, Pogoda B, Pouvreau S, Preston J, Ranger C, Sanderson W, Sas H, Strand A, Sutherland W (2020) Forty questions of importance to the policy and practice of oyster restoration in Europe. Aquat Conserv Mar Freshw Ecosyst Manuscr.
Method used
Main source of information:
1. Assessment carried out by Wageningen Marine Research (Bos, Tamis) and Alfred Wegener Institute Helmholtz Centre for Polar and Marine Research (Pogoda, Groß), based upon:
a) a literature review and;
b) a questionaire send to experts from all relevant contracting parties, identified and contacted via the Native Oyster Restoration Alliance (NORA).
Sheet reference:
BDC2020/ European flat oyster and Ostrea edulis beds