Executive Summary
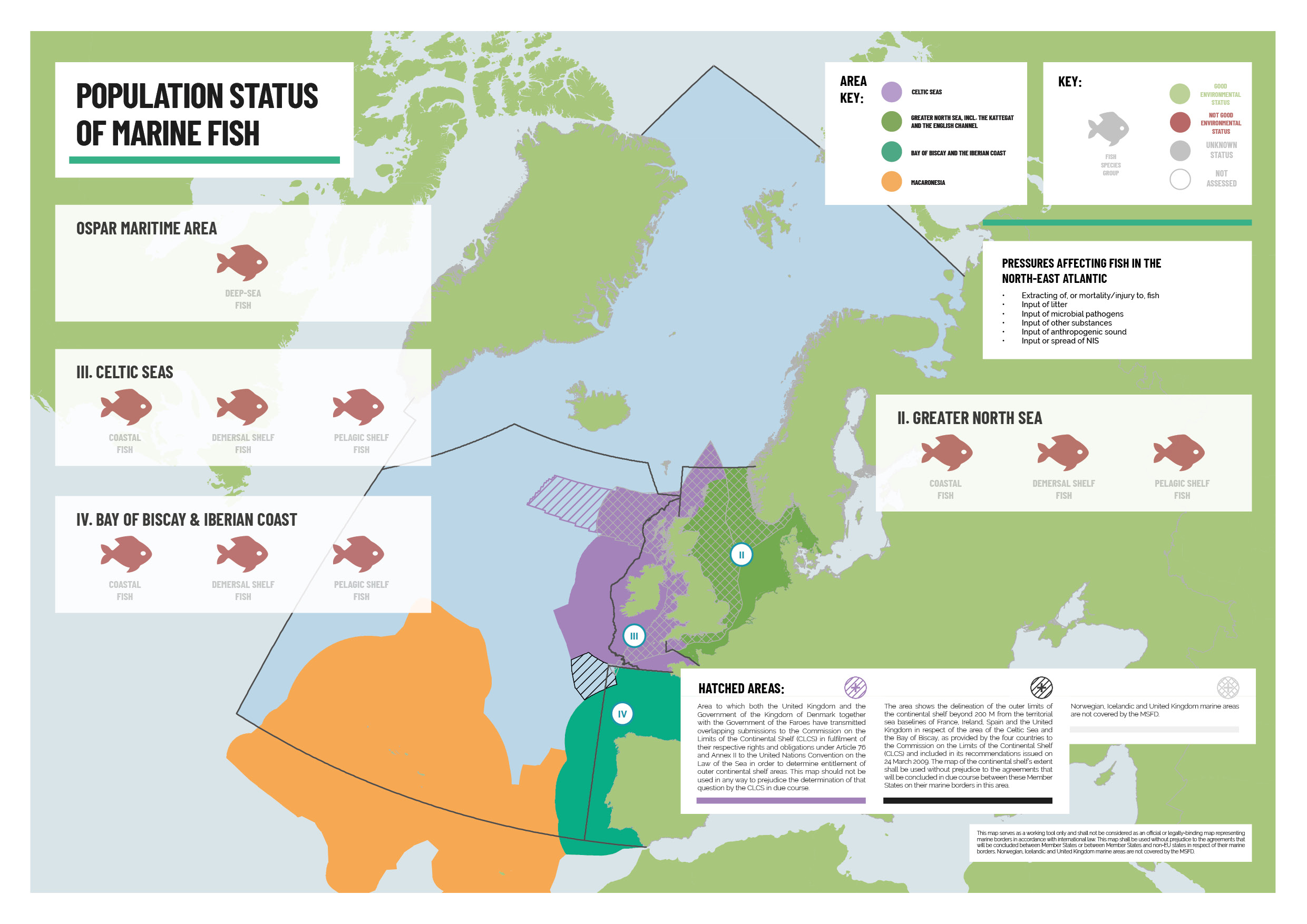
Fish are an essential part of the marine ecosystem, providing many ecosystem functions. Society has a need for fish as a vital source of food. Commercial fishing provides for that societal need. Fisheries management in the North-East Atlantic aims to maintain fish stocks at healthy levels. An integrated assessment of species/stock status found that marine fish were not in good environmental status, despite the improving status of many individual fish stocks over recent years. This finding was common to coastal, demersal and pelagic fish in the Greater North Sea, Celtic Seas, Bay of Biscay and Iberian Coast Regions and for deep-sea fish across the OSPAR Maritime Area. A separate assessment for the Norwegian waters in OSPAR Arctic Waters showed mixed results, with commercial stocks in both good and not good status. The overall status of fish was assessed by integrating the OSPAR common indicator for the recovery of sensitive fish species with third-party assessments of commercial fish stocks produced by the International Council for the Exploration of the Sea (ICES) and the International Commission for the Conservation of Atlantic Tunas (ICCAT). This is the most comprehensive fish status assessment by OSPAR to date, and the first to integrate commercial fish stocks.
The regulation of fishing activities lies outside of OSPAR’s competence, but a requirement to cooperate with other competent bodies is set out in Article 4, Annex V of the OSPAR Convention and this is an explicit objective of the North-East Atlantic Environment Strategy (NEAES) 2030. Fisheries management regulations have successfully brought the harvesting of some fish stocks to sustainable levels, but many stocks are still overexploited. Measures are now also being taken with an ecosystem perspective, including the introduction of some intended to protect vulnerable habitats and species. However, concerns remain, including in relation to by-catch of sensitive/non-commercial species, the need to integrate concepts of ecosystem function into fisheries management regulation – such as the idea of trophic cascades – and how management regimes can take account of the impact of fisheries on the pelagic habitat and food webs. For its part, OSPAR has focused on 22 fish species that are considered to be under threat and/or in decline in the North-East Atlantic and has taken national and collective actions to protect and conserve almost all of these species. With the exception of some skates and rays, most of the OSPAR listed fish species are assessed as having poor status and there are still gaps in the OSPAR Marine Protected Area (MPA) network for almost all of the fish species on the OSPAR List of Threatened and/or Declining Species and Habitats (OSPAR Agreement 2008-06).
In response to the findings of this assessment, OSPAR will initiate discussions on regional-scale ecosystem-based management, including through the ‘Collective Arrangement’ and in cooperation with fisheries management bodies and other competent organisations; this will include initiatives to minimise, and where possible eliminate, incidental by-catch of fish.
Q1. Identify the problems? Are they the same in all OSPAR Regions?
Fish are an essential part of the marine ecosystem, providing many ecosystem services. This means that the good health of fish communities has a positive effect on the marine food web and, conversely, that poor health will have a negative effect. Society has a need for food, and fish are a vital source of protein, which means that many species are targeted by fishing activity in the North-East Atlantic. The removal of these target species can result in reduced prey for other species, thereby affecting food web dynamics and relationships between species. Mortality through fishing activity can reduce the biomass of fish populations to unsafe levels and affect the age structure of populations by reducing the proportion of older and larger individuals.
A variety of fishing methods are utilised, depending on the target species. While all have the potential to remove non-target species, negatively impacting fish species of conservation concern such as sharks, skates, and rays as well as other species such as marine mammals and seabirds, these impacts vary greatly according to the gear used. Fishing methods which contact the seabed cause physical disturbance which can result in habitat loss, impacting fish species diversity, reducing prey, and affecting fish survival. This can severely impact OSPAR listed threatened and/or declining habitats such as Lophelia pertusa reefs and maerl beds. Bottom-trawl and dredge fisheries also cause sediment re-suspension, which can affect the availability of seabed species to the fish that feed upon them.
Climate change can impact marine fish by altering reproductive output, by changing the distribution patterns of species and by changing growth and mortality.
Society’s need for the infrastructure associated with a range of activities such as energy generation, coastal defence, land claim and aggregate extraction can negatively affect fish habitat and migration routes. The infrastructure needs for energy generation are expected to increase significantly in the next decade.
In the 2010 Quality Status Report (QSR) it was reported that fishing pressure had a considerable impact on marine ecosystems, with many fish stocks reported as still being outside safe biological limits. The 2017 Intermediate Assessment indicated that there were signs of recovery in fish populations.

Trawlers in the North Sea. © Shutterstock
Q2. What has been done?
The dominant pressure on fish populations in the North-East Atlantic is the extraction of biomass through fishing activities which lie outside of OSPAR’s competence to manage, as set out in Article 4, Annex V of the OSPAR Convention; this requires cooperation with other competent bodies, an explicit objective of the NEAES 2030.
OSPAR has listed 22 fish species (mostly non-targeted) that are considered to be under threat and/or in decline in the North-East Atlantic and has taken national and collective actions to protect and conserve almost all these species.
Fisheries management regulations have resulted in the harvesting of more fish stocks at levels considered sustainable for those stocks and in a shift of focus from fish stock management to an ecosystem perspective under which measures to protect vulnerable habitats and species are being introduced. However, concerns remain, including issues relating to by-catch.
There have been instances of Marine Protected Areas being put in place by some Contracting Parties in order to protect vulnerable fish.
Q3. Did it work?
With the exception of some skates and rays, most of the OSPAR listed fish species are assessed as having poor status, and there are still gaps in the OSPAR MPA network for almost all of the fish species on the OSPAR List. The coverage is considered as being ecologically coherent only for houting, long snouted seahorses, allis shad and salmon, among the species for which MPAs may be a useful management tool.
Fisheries management regulations have resulted in many successes in the harvesting of fish stocks at levels considered sustainable, and in rebuilding stocks to healthy levels. However, overall good environmental status has not been achieved for fish population status. Another development has been a shift from fish stock management to an ecosystem perspective, with the introduction of measures to protect vulnerable habitats and species. However, concerns remain, including in relation to by-catch, the need to integrate concepts of ecosystem function into fisheries management regulation – such as the idea of trophic cascades – and how management regimes can take account of the impact of fisheries on the pelagic habitat and food webs. Further work to progress the alignment of fisheries management and environmental protection responses is also required.

Marine Protected Area coverage is considered as being ecologically coherent for long snouted seahorses. © Monterey Bay Aquarium
Q4. How does this field affect the overall quality status?
The overall situation in which fish groups across the various assessed areas are not in good environmental status has wider implications for the marine ecosystem. Fish provide many ecosystem services, and consequently the status of fish populations has implications for the wider ecosystem. Fish play a role in regulating the balance of the food web. The degree to which fish influence the regulation of food web dynamics can vary depending on the physical and climatic conditions to which they are subjected.
The ecosystem service of providing biomass that can be harvested by humans (wild fish and other natural aquatic biomass and related raw materials) can also be affected in ways that lead to shifts in commercial fisheries. Such shifts can include extra fishing pressure on vulnerable components of the marine ecosystem. Reduced fish biomass also impacts feeding opportunities for other ecosystem components such as birds and mammals. Reduced fish population abundance and biomass can lead to a decrease in carbon fixation by the marine ecosystem.
Q5. What do we do next?
Two of the key objectives under the NEAES 2030 are particularly relevant. First, as a cross-cutting issue, OSPAR will initiate discussions on the development of a practical approach for regional-scale ecosystem-based management, including through the ‘Collective Arrangement’ (Agreement 2014-09) and in cooperation with fisheries management bodies and other competent organisations, in order to strengthen ecosystem resilience to climate change and to safeguard the marine environment, its biodiversity and ecosystem services (SX.O2). Also, OSPAR has set out to implement MPAs as well as other effective area-based conservation measures (OECMs) (S5.O1).
In addition, “OSPAR will work with relevant competent authorities and other stakeholders to minimise, and where possible eliminate, incidental by-catch of marine mammals, birds, turtles and fish so that it does not represent a threat to the protection and conservation of these species and will work towards strengthening the evidence base concerning incidental by-catch by 2025” (S7.O6). This will give increased attention to the effects of incidental by-catch, including for protected fish species. OSPAR should also take into consideration relevant by-catch studies in the framework of OSPAR, the European Commission and ICES and their conclusions.
Other areas for potential OSPAR response could include:
- Closing the gaps in the OSPAR MPA network for the fish species on the OSPAR List;
- Progressing the work of understanding the management effectiveness of MPAs, and sharing experience and best practice on management actions that best achieve the conservation objectives as they relate to fish communities;
- Mitigation responses that support job changes in areas where fishing industry has declined;
- The restoration of certain habitats to produce co-benefits for some fish species ( Benthic Habitats Thematic Assessment );
- Examining the implications of removing fish from the wild (wrasse and lumpfish) for use as cleaner fish to manage parasite levels in farmed fish, including the genetic interaction of escaped cleaner fish; this has been identified by ICES as worthy of further investigation (ICES, 2020).
The effective implementation of many of these objectives will depend heavily on national action, which should continue to be reported through, for example, the implementation reporting requirements of OSPAR’s Recommendations on threatened and declining species and habitats (next reporting due in December 2025).
OSPAR recognises the need to increase its focus on identifying and implementing collective actions which add value both to existing national actions and to the efforts of other international organisations. Overall, the 2019 implementation reporting indicates that there is a good level of engagement to implement the national actions within the OSPAR Recommendations, in particular within the areas where the species and habitats are considered to be under threat and/ or in decline. The level of engagement in collective actions is clearly lower, with some of the more complex actions not having been progressed and implemented. Many of these actions focus on monitoring and assessment and relatively few on response, but in both cases there has been only modest progress. OSPAR will therefore develop a series of biodiversity action plans, starting with marine birds and coastal shelf benthic habitats, in order to identify priority response measures which are well-defined, add value and can be delivered within the resources available to the OSPAR Contracting Parties.
The 2023 QSR provides a powerful evidence base for action. OSPAR will strengthen its capacity to use this evidence base, and all future assessments, to support engagement with other international partners. Engagement cannot be an end in itself: the development of a practical approach to ecosystem-based management (EBM) will provide the opportunity and the mechanism to share evidence and common objectives for a more sustainable use of the marine environment. Working with interested partners and drawing on international best practice, OSPAR will design and implement a pilot project on EBM in one of the OSPAR Regions.
Progress against all of these challenging objectives for biodiversity will be tracked through OSPAR’s NEAES 2030 Implementation Plan. A planned review in 2025 will provide an opportunity to adjust OSPAR’s NEAES 2030 and, if necessary, OSPAR will take further action to protect and conserve biodiversity.
Amaral-Zettler, L.A., Zettler, E.R. and Mincer, T.J. (2020). Ecology of the plastisphere. Nature Reviews Microbiology, 18(3):139-151.
Arctic Council. (2005). Arctic Climate Impact Assessment. Arctic Council Secretariat. https://oaarchive.arctic-council.org/handle/11374/2453
Barange, M.; Perry, R.I. (2009). Physical and ecological impacts of climate change relevant to marine and inland capture fisheries and aquaculture. In K. Cochrane, C. De Young, D. Soto and T. Bahri (eds). Climate change implications for fisheries and aquaculture: overview of current scientific knowledge. FAO Fisheries and Aquaculture Technical Paper. No. 530. Rome, FAO. pp. 7–106
Baudron, A.R., Brunel, T., Blanchet, M.A., Hidalgo, M., Chust, G., Brown, E.J., Kleisner, K.M., Millar, C., MacKenzie, B.R., Nikolioudakis, N. and Fernandes, J.A. (2020). Changing fish distributions challenge the effective management of European fisheries. Ecography, 43(4), pp.494-505. https://doi.org/10.1111/ecog.04864
Bentley, J. W., Lundy, M. G., Howell, D., Beggs, S. E., Bundy, A., De Castro, F.and Reid, D. G. (2021). Refining fisheries advice with stock-specific ecosystem information. Frontiers in Marine Science, 8, 602072.
Beukhof, E., Frelat, R., Pecuchet, L., Maureaud, A., Dencker, T. S., Sólmundsson, J. and Lindegren, M. (2019). Marine fish traits follow fast-slow continuum across oceans. Scientific reports, 9(1), 1-9.Bluemel, J. K., Fischer, S. H., Kulka, D. W., Lynam, C. P., & Ellis, J. R. (2022). Decline in Atlantic wolffish Anarhichas lupus in the North Sea: Impacts of fishing pressure and climate change. Journal of Fish Biology, 100(1), 253–267. https://doi.org/10.1111/jfb.14942
Bluemel, J. K., Fischer, S. H., Kulka, D. W., Lynam, C. P., & Ellis, J. R. (2022). Decline in Atlantic wolffish Anarhichas lupus in the North Sea: Impacts of fishing pressure and climate change. Journal of Fish Biology, 100(1), 253-267
Bœuf, G. and Payan, P. (2001). How should salinity influence fish growth?. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 130(4):411-423.
Bosch, N. E., Monk, J., Goetze, J., Wilson, S., Babcock, R. C., Barrett, N., Clough, J., Currey-Randall, L. M., Fairclough, D. V., Fisher, R., Gibbons, B. A., Harasti, D., Harvey, E. S., Heupel, M. R., Hicks, J. L., Holmes, T. H., Huveneers, C., Ierodiaconou, D., Jordan, A., Knott, N. A. and Langlois, T. J. (2022). Effects of human footprint and biophysical factors on the body-size structure of fished marine species. Conservation biology : the journal of the Society for Conservation Biology, 36(2), e13807. https://doi.org/10.1111/cobi.13807
Brander, K. and Mohn, R. (2004). Effect of the North Atlantic Oscillation on recruitment of Atlantic cod (Gadus morhua). Canadian Journal of Fisheries and Aquatic Sciences, 61(9), 1558–1564. https://doi.org/10.1139/f04-087
Brown, C.J.M., Curry, R.A., Gray, M.A. et al. Considering Fish as Recipients of Ecosystem Services Provides a Framework to Formally Link Baseline, Development, and Post-operational Monitoring Programs and Improve Aquatic Impact Assessments for Large Scale Developments. Environmental Management 70, 350–367 (2022).
Bryndum-Buchholz, A., Tittensor, D. P. and Lotze, H. K. (2021). The status of climate change adaptation in fisheries management: Policy, legislation and implementation. Fish and Fisheries, 22(6), 1248–1273. https://doi.org/10.1111/faf.12586
Cheung, W. W. L., Lam, V. W. Y., Sarmiento, J. L., Kearney, K., Watson, R., Zeller, D. and Pauly, D. (2010). Large-scale redistribution of maximum fisheries catch potential in the global ocean under climate change. Global Change Biology, 16(1), 24–35. https://doi.org/10.1111/j.1365-2486.2009.01995.x
Cormier, R., Elliott, M. andRice, J. (2019). Putting on a Bow-tie to sort out who does what and why in the complex arena of marine policy and management. Science of the Total Environment, 648: 293-305. https://doi.org/10.1016/j.scitotenv.2018.08.168
Cormier, R., Elliott, M., and Kannen, A. (2018). IEC/ISO Bow-tie analysis of marine legislation: A case study of the Marine Strategy Framework Directive. ICES Cooperative Research Report No. 342. 70 pp. https://doi.org/10.17895/ices.pub.4504 http://www.ices.dk/sites/pub/Publication%20Reports/Cooperative%20Research%20Report%20(CRR)/CRR342/CRR342.pdf
Cornacchia, F. (2022) Impacts on Ecosystem Services due to changes in the state of the environment in the North-East Atlantic Ocean. https://open.rws.nl/open-overheid/onderzoeksrapporten/@142922/impacts-on-ecosystem-services-due-to/
Crain, C.M., Halpern, B.S., Beck, M.W. and Kappel, C.V. (2009). Understanding and managing human threats to the coastal marine environment. Annals of the New York Academy of Sciences, 1162(1):39-62
Culhane, F., Teixeira, H., Nogueira, A. J., Borgwardt, F., Trauner, D., Lillebø, A. and Robinson, L. A. (2019). Risk to the supply of ecosystem services across aquatic ecosystems. Science of the total environment, 660, 611-621
Drinkwater, K. F. (2005). The response of Atlantic cod (Gadus morhua) to future climate change. ICES Journal of Marine Science, 62(7), 1327–1337. https://doi.org/10.1016/j.icesjms.2005.05.015
Dulvy, N.K., Rogers, S.I., Jennings, S., Stelzenmüller, V., Dye, S.R. and Skjoldal, H.R. (2008). Climate change and deepening of the North Sea fish assemblage: a biotic indicator of warming seas. Journal of Applied Ecology, 45(4), pp.1029-1039. https://doi.org/10.1111/j.1365-2664.2008.01488.x
FAO. 2023. Implementation of the International Plan of Action to Deter, Prevent and Eliminate Illegal, Unreported and Unregulated Fishing – 1. Methodologies and indicators for the estimation of the magnitude and impact of illegal, unreported and unregulated fishing: 1.1 Principles and approaches. FAO Technical Guidelines for Responsible Fisheries, No. 9, Suppl. 1, Vol. 1. Rome. https://doi.org/10.4060/cc6434en
Gasparatos, A., Doll, C.N., Esteban, M., Ahmed, A. and Olang, T.A. (2017). Renewable energy and biodiversity: Implications for transitioning to a Green Economy. Renewable and Sustainable Energy Reviews, 70:161-184.
Genner, M. J., Sims, D. W., Southward, A. J., Budd, G. C., Masterson, P., Mchugh, M., Rendle, P., Southall, E. J., Wearmouth, V. J. and Hawkins, S. J. (2010). Body size-dependent responses of a marine fish assemblage to climate change and fishing over a century-long scale. Global Change Biology, 16(2), 517–527. https://doi.org/10.1111/j.1365-2486.2009.02027.x
Gestoso, I., Ramalhosa, P. and Canning-Clode, J. (2018). Biotic effects during the settlement process of non-indigenous species in marine benthic communities. Aquatic Invasions, 13(2)
Gill, A.B. (2005). Offshore renewable energy: ecological implications of generating electricity in the coastal zone. Journal of applied ecology, pp.605-615.
Graham, C. T. and Harrod, C. (2009). Implications of climate change for the fishes of the British Isles. Journal of Fish Biology, 74(6), 1143–1205. https://doi.org/10.1111/j.1095-8649.2009.02180.x
Haines-Young, R. and Potschin-Young, M. (2018). Revision of the common international classification for ecosystem services (CICES V5. 1): a policy brief. One Ecosystem, 3, e27108.
Hamilton, P.B., Cowx, I.G., Oleksiak, M.F., Griffiths, A.M., Grahn, M., Stevens, J.R., Carvalho, G.R., Nicol, E. and Tyler, C.R. (2016). Population‐level consequences for wild fish exposed to sublethal concentrations of chemicals–a critical review. Fish and Fisheries, 17(3):545-566.
Hammerschlag, N., Schmitz, O. J., Flecker, A. S., Lafferty, K. D., Sih, A., Atwood, T. B. and Cooke, S. J. (2019). Ecosystem function and services of aquatic predators in the Anthropocene. Trends in ecology & evolution, 34(4), 369-383
Hawkins, A. D. and Popper, A. N. (2017). A sound approach to assessing the impact of underwater noise on marine fishes and invertebrates. ICES Journal of Marine Science, 74(3), 635-651.
Hiddink, J. G., Jennings, S., Kaiser, M. J., Queirós, A. M., Duplisea, D. E. and Piet, G. J. (2006). Cumulative impacts of seabed trawl disturbance on benthic biomass, production, and species richness in different habitats. Canadian journal of fisheries and aquatic sciences, 63(4), 721-736.
Hjerne, O., Hajdu, S., Larsson, U., Downing, A. S., & Winder, M. (2019). Climate driven changes in timing, composition and magnitude of the Baltic Sea phytoplankton spring bloom. Frontiers in Marine Science, 6, 482.
Hočevar, S. and Kuparinen, A. (2021). Marine food web perspective to fisheries-induced evolution. Evolutionary Applications, 14, 2378– 2391. Available at: https://doi.org/10.1111/eva.13259
Holmlund, C. M. and Hammer, M. (1999). Ecosystem services generated by fish populations. Ecological economics, 29(2), 253-268.
Howell, D., Schueller, A. M., Bentley, J. W., Buchheister, A., Chagaris, D., Cieri, M. and Townsend, H. (2021). Combining ecosystem and single-species modeling to provide ecosystem-based fisheries management advice within current management systems. Frontiers in Marine Science, 7, 607831.
ICES (2016) Report of the Working Group on Fish Distribution Shifts (WKFISHDISH), 22–25 November 2016. ICES HQ, Copenhagen, Denmark. ICES CM 2016/ACOM: 55, 197 pp.
ICES (2020). Working Group on Environmental Interactions of Aquaculture (WGEIA). ICES Scientific Reports. 2:112. 187 pp. Available at: http://doi.org/10.17895/ices.pub.7619
ICES (2021a): Barents Sea Ecoregion – Fisheries overview. ICES Advice: Fisheries Overviews. Report. Available at: https://doi.org/10.17895/ices.advice.9166
ICES (2021b): Norwegian Sea ecoregion – Fisheries overview. ICES Advice: Fisheries Overviews. Report. https://doi.org/10.17895/ices.advice.9150
ICES(2022) Working Group for the Celtic Seas Ecoregion (WGCSE). Draft report. ICES Scientific Reports. 4:45. 931 pp. http://doi.org/10.17895/ices.pub.19863796
ICES (2022): Norwegian Sea ecoregion – fisheries overview. ICES Advice: Fisheries Overviews. Report. https://doi.org/10.17895/ices.advice.21640826.v1
Ikpewe, I. E., Baudron, A. R., Ponchon, A. and Fernandes, P. G. (2021). Bigger juveniles and smaller adults: Changes in fish size correlate with warming seas. Journal of Applied Ecology, 58(4), 847-856.
IPCC. (2022). Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change.
Jennings, S. and Kaiser, M.J. (1998). The effects of fishing on marine ecosystems. In Advances in marine biology (Vol. 34, pp. 201-352). Academic Press.
Knights, A. M., Piet, G. J., Jongbloed, R. H., Tamis, J. E., White, L., Akoglu, E., Boicenco, L. et al. (2015). An exposure-effect approach for evaluating ecosystem-wide risks from human activities. ICES Journal of Marine Science, 72: 1105–1115. http://academic.oup.com/icesjms/article/72/3/1105/703182/An-exposureeffect-approach-for-evaluating.
Lefort, S., Aumont, O., Bopp, L., Arsouze, T., Gehlen, M. and Maury, O. (2015). Spatial and body-size dependent response of marine pelagic communities to projected global climate change. Global Change Biology, 21(1), 154–164. https://doi.org/10.1111/gcb.12679
Lyashevska, O., Harma, C., Minto, C., Clarke, M. and Brophy, D. (2020). Long-term trends in herring growth primarily linked to temperature by gradient boosting regression trees. Ecological Informatics, 60, 101154. https://doi.org/10.1016/j.ecoinf.2020.101154
Mantzouni, I. and MacKenzie, B. R. (2010). Productivity responses of a widespread marine piscivore, Gadus morhua, to oceanic thermal extremes and trends. Proceedings of the Royal Society B: Biological Sciences, 277(1689), 1867–1874. https://doi.org/10.1098/rspb.2009.1906
Marine Stewardship Council (2019a). Working together for thriving oceans. MSC annual report 2018-19. Available at: https://www.msc.org/docs/default-source/default-document-library/about-the-msc/msc-annual-report-2018-2019.pdf
Marine Stewardship Council (2019b). MSC certificates suspended for all North-East Atlantic mackerel fisheries. Press Release January 31 2019. Available at: https://www.msc.org/media-centre/press-releases/press-release/msc-certificates-suspended-for-all-north-east-atlantic-mackerel-fisheries
Marine Stewardship Council (2019c). North Sea cod to lose sustainability certification. Press release 24 September 2019. Available at: https://www.msc.org/media-centre/press-releases/north-sea-cod-to-lose-sustainability-certification
Marine Stewardship Council (2020). Celebrating and supporting sustainable fisheries. MSC annual report 2019–20. Available at: https://www.msc.org/docs/default-source/default-document-library/about-the-msc/msc-annual-report-2019-2020.pdf
McQueen, K., Marshall, C.T. 2017. Shifts in spawning phenology of cod linked to rising sea temperatures. ICES Journal of Marine Science, 74(6): 1561-1573. https://doi.org/10.1093/icesjms/fsx025
Mészáros, L., Van der Meulen, F., Jongbloed, G. and El Serafy, G. (2021). Climate change induced trends and uncertainties in phytoplankton spring bloom dynamics. Frontiers in Marine Science, p.1067.
Murgier, J., McLean, M., Maire, A., Mouillot, D., Loiseau, N., Munoz, F. and Auber, A. (2021). Rebound in functional distinctiveness following warming and reduced fishing in the North Sea. Proceedings of the Royal Society B, 288(1942), 20201600.
Naylor, R.L., Goldburg, R.J., Primavera, J.H., Kautsky, N., Beveridge, M., Clay, J., Folke, C., Lubchenco, J., Mooney, H. and Troell, M. (2000). Effect of aquaculture on world fish supplies. Nature, 405(6790):1017-1024.
NEAFC (2018). Statement by the North-East Atlantic Fisheries Commission (NEAFC) regarding the conclusion of the negotiations on the Agreement to Prevent Unregulated High Seas Fisheries in the Central Arctic Ocean. Available at: https://www.neafc.org/system/files/NEAFC-statement_Central-Arctic-Ocean-Agreement.pdf
NEAFC (2020). NEAFC website section on Illegal Fishing. Available at: https://www.neafc.org/mcs/iuu
Neuheimer, A.B., MacKenzie, B.R. and Payne, M.R. (2018) Temperature-dependent adaptation allows fish to meet their food across their species’ range. Science Advances, 4 (7), eaar4349, doi: 10.1126/sciadv.aar4349
Núnez-Riboni, I., Taylor, M. H., Kempf, A., Püts, M. and Mathis, M. (2019). Spatially resolved past and projected changes of the suitable thermal habitat of North Sea cod (Gadus morhua) under climate change. ICES Journal of Marine Science, 76(7), 2389-2403
Oke, K. B., Cunningham, C. J., Westley, P. A. H., Baskett, M. L., Carlson, S. M., Clark, J. and Palkovacs, E. P. (2020). Recent declines in salmon body size impact ecosystems and fisheries. Nature communications, 11(1), 1-13.
Olafsdottir, A.H., Utne, K.R., Jacobsen, J.A., Jansen, T., Óskarsson, G.J., Elvarsson, B.Þ., Broms, C. and Slotte, A. (2019) Geographical expansion of Northeast Atlantic mackerel (Scomber scombrus) in the Nordic Seas from 2007 to 2016 was primarily driven by stock size and constrained by low temperatures. Deep Sea Research Part II: Topical Studies in Oceanography, 159, 152–168. https://doi.org/10.1016/j.dsr2.2018.05.023
Ottersen, G. (2000). Covariability in early growth and year-class strength of Barents Sea cod, haddock, and herring: The environmental link. ICES Journal of Marine Science, 57(2), 339–348. https://doi.org/10.1006/jmsc.1999.0529
Peck, M. and Pinnegar, J.K. (2018) Chapter 5: Climate change impacts, vulnerabilities and adaptations: North Atlantic and Atlantic Arctic marine fisheries. In Impacts of Climate Change on Fisheries and Aquaculture: Synthesis of current knowledge, adaptation and mitigation options. FAO Fisheries and Aquaculture Technical Paper No. 627. FAO, Rome, pp. 87–111.
Pecuchet, L., Lindegren, M., Hidalgo, M., Delgado, M., Esteban, A., Fock, H. O. and Payne, M. R. (2017). From traits to life‐history strategies: Deconstructing fish community composition across European seas. Global Ecology and Biogeography, 26(7), 812-822.
Perry, A.L., Low, P.J., Ellis, J.R. and Reynolds, J.D. (2005) Climate change and distribution shifts in marine fishes. Science, 308, 1912–1915. https://doi.org/10.1126/science.1111322
Pimentel, M.S., Faleiro, F., Margues, T., Bispo, R., Dionísio, G., Faria, A.M., Machado, J. et al. (2016) Foraging behaviour, swimming performance and malformations of early stages of commercially important fishes under ocean acidification and warming. Climatic Change, 137(3–4), 495–509.
Pinnegar, J. K., Jennings, S., O’Brien, C. M. and Polunin, N. V. C. (2002). Long-term changes in the trophic level of the Celtic Sea fish community and fish market price distribution. Journal of Applied Ecology, 39(3), 377–390. https://doi.org/10.1046/j.1365-2664.2002.00723.x
Planque, B. and Frédou, T. (1999). Temperature and the recruitment of Atlantic cod (Gadus morhua). Canadian Journal of Fisheries and Aquatic Sciences, 56(11), 2069–2077. https://doi.org/10.1139/f99-114
Poloczanska, E. S., Brown, C. J., Sydeman, W. J., Kiessling, W., Schoeman, D. S., Moore, P. J., Brander, K., Bruno, J. F., Buckley, L. B., Burrows, M. T., Duarte, C. M., Halpern, B. S., Holding, J., Kappel, C. V., O’Connor, M. I., Pandolfi, J. M.,
Parmesan, C., Schwing, F., Thompson, S. A. and Richardson, A. J. (2013). Global imprint of climate change on marine life. Nature Climate Change, 3(10), 919–925. https://doi.org/10.1038/nclimate1958
Régnier, T., Gibb, F.M. and Wright, P.J. Understanding temperature effects on recruitment in the context of trophic mismatch. Sci Rep 9, 15179 (2019). https://doi.org/10.1038/s41598-019-51296-5
Robinson, L.A., White, L.J., Culhane, F.E. and Knights, A.M. (2013). ODEMM Pressure Assessment Userguide V.2. ODEMM Guidance Document Series No.4. EC FP7 project (244273) ‘Options for Delivering Ecosystem-based Marine Management’. University of Liverpool. ISBN: 978-0-906370-86-5: 14 pp.
Rutterford, L. A., Simpson, S. D., Jennings, S., Johnson, M. P., Blanchard, J. L., Schön, P. J. and Genner, M. J. (2015). Future fish distributions constrained by depth in warming seas. Nature Climate Change, 5(6), 569-573.
Tavares, D. C., Moura, J. F., Acevedo-Trejos, E. and Merico, A. (2019). Traits shared by marine megafauna and their relationships with ecosystem functions and services. Frontiers in Marine Science, 6, 262.
Tekwa, E.W., Watson, J.R. and Pinsky, M.L. (2022). Body size and food–web interactions mediate species range shifts under warming. Proceedings of the Royal Society B, 289(1972), p.20212755. https://doi.org/10.1098/rspb.2021.2755
Thrush, S.F. and Dayton, P.K. (2002). Disturbance to marine benthic habitats by trawling and dredging: implications for marine biodiversity. Annual review of ecology and systematics, 33(1):449-473.
Thrush, S.F. and Dayton, P.K. (2010). What can ecology contribute to ecosystem-based management?. Annual Review of Marine Science, 2:419-441.
Travers-Trolet, M., Bourdaud, P., Genu, M., Velez, L. and Vermard, Y. (2020). The Risky Decrease of Fishing Reference Points Under Climate Change. Frontiers in Marine Science, 7, 568232. https://doi.org/10.3389/fmars.2020.568232
van Overzee, H.M.J. and Rijnsdorp, A.D. Effects of fishing during the spawning period: implications for sustainable management. (2015) Reviews in Fish Biology and Fisheries 25, 65–83. https://doi.org/10.1007/s11160-014-9370-x
Violle, C., M. L. Navas, D. Vile, E. Kazakou, C. Fortunel, I. Hummel, and E. Garnier. (2007). Let the concept of trait be functional! Oikos 116:882– 892.
Watling, L. and Norse, E.A. (1998). Disturbance of the seabed by mobile fishing gear: a comparison to forest clearcutting. Conservation biology, 12(6):1180-1197.
Wells, S.R., Bresnan, E., Cook, K., Eerkes-Medrano, D., Machairopoulou, M., Mayor, D.J., Rabe, B. and Wright, P.J. (2022). Environmental drivers of a decline in a coastal zooplankton community. ICES Journal of Marine Science, 79(3), pp.844-854.
Woods, J.S., Veltman, K., Huijbregts, M.A., Verones, F. and Hertwich, E.G. (2016). Towards a meaningful assessment of marine ecological impacts in life cycle assessment (LCA). Environment international, 89:48-61
Fish Assessments
Contributors
Lead authors: Maurice Clarke, Joanna Bluemel, Ilaria Coscia, Nis Sand Jacobsen, Chris Lynam, Wolfgang Nikolas Probst, Daniel Wood, Bee Beryx, Terence Ilott, Federico Cornacchia, Rob van der Veeren, Emily Corcoran, Ailbhe Kavanagh, Dafne Eerkes Medrano, Ruth Kelly.
Supporting authors: Anna Rindorf, Damian Delaunay, Gro van der Meeren, Claudia Junge, Anik Brin’damour, Anthony Acou, Patricia Goncalves, Håkon Wennhage, Francisco Velasco, GerJan Piet, Vanda Carmo, Els Torelle, Janos Hennicke, and Lena Avellan.
Supported by: OSPAR Fish Expert Group, Intersessional Correspondence Group on the Coordination of Biodiversity Assessment and Monitoring (ICG-COBAM), OSPAR Biodiversity Committee (BDC), Intersessional Correspondence Group on the Quality Status Report (ICG-QSR), Intersessional Correspondence Group on Ecosystem Assessment Outlook (ICG-EcoC), Intersessional Correspondence Group on Economic and Social Analysis (ICG-ESA), Climate Change Expert Group (CCEG), and OSPAR Commission Secretariat
Citation
OSPAR, 2023. Fish Thematic Assessment. In: OSPAR, 2023: Quality Status Report 2023. OSPAR Commission, London. Available at: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/thematic-assessments/fish/